The given dipeptide is Ala-Ser. Indicate the charge state for each structure at the given pH by adding the + or - the appropriate functional groups and adding or removing hydrogen atoms where appropriate. charges to pH = 1 pH = 7 Select Draw Rings More Erase Select Draw Rings More Erase H. %3D H C C -0- H H. -C- N C C -0- H -CI N- H. CH, CH, CH, CH, он он
The given dipeptide is Ala-Ser. Indicate the charge state for each structure at the given pH by adding the + or - the appropriate functional groups and adding or removing hydrogen atoms where appropriate. charges to pH = 1 pH = 7 Select Draw Rings More Erase Select Draw Rings More Erase H. %3D H C C -0- H H. -C- N C C -0- H -CI N- H. CH, CH, CH, CH, он он
Biochemistry
9th Edition
ISBN:9781305961135
Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Publisher:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Chapter2: Water: The Solvent For Biochemical Reactions
Section: Chapter Questions
Problem 40RE: MATHEMATICAL A catalog in the lab has a recipe for preparing 1 L of a TRIS buffer at 0.0500 M and...
Related questions
Question
All one question
Transcribed Image Text:Assignment Score:
82.2%
K Give Up?
O Hint
Check Answer
Resources
Question 6 of 23
Attempt 2
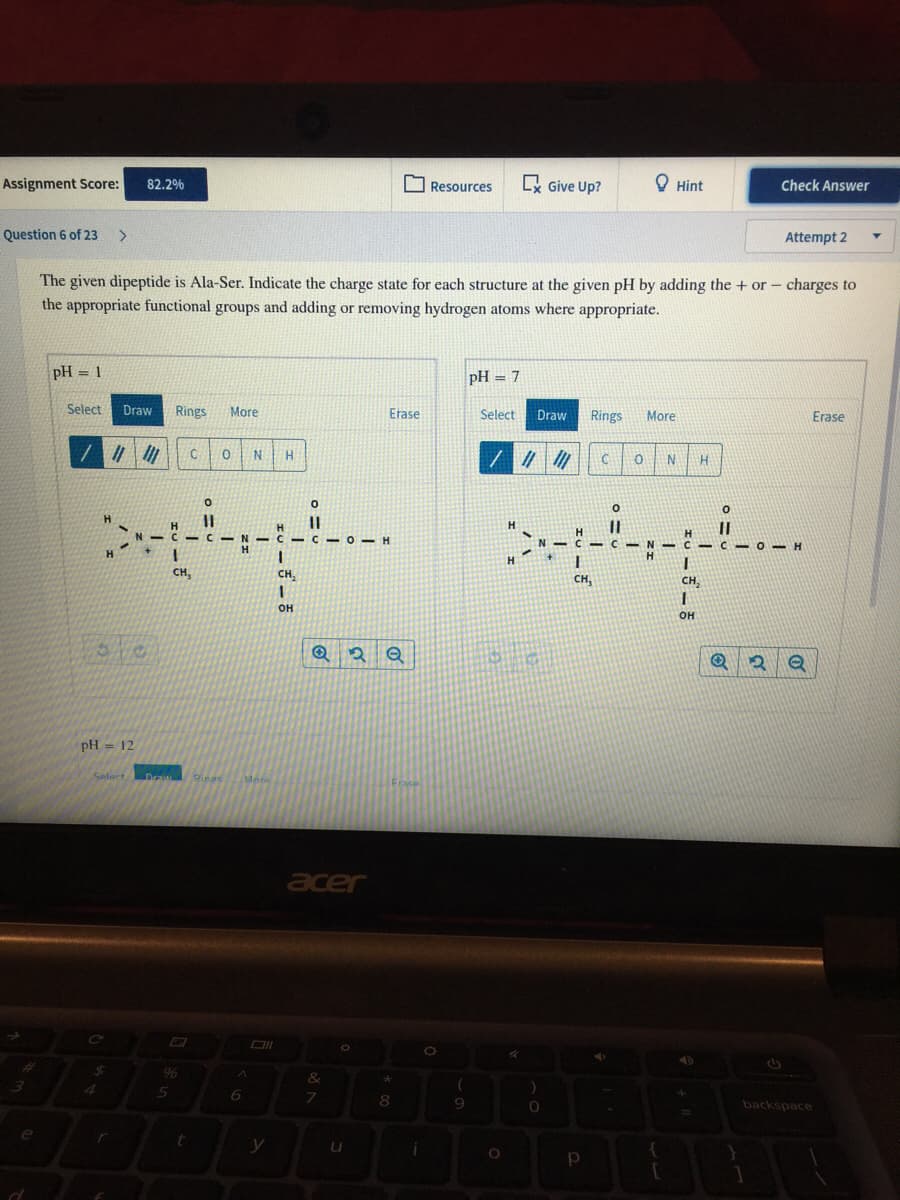
The given dipeptide is Ala-Ser. Indicate the charge state for each structure at the given pH by adding the + or- charges to
the appropriate functional groups and adding or removing hydrogen atoms where appropriate.
pH = 1
pH = 7
%3D
Select
Draw
Rings
More
Erase
Select
Draw
Rings
More
Erase
C
N
%3D
-C- N -C- C-O- H
2N-
N - C C-O
H
H.
CH,
CH,
CH,
CH,
он
он
pH = 12
Seloct
Draw
Rings
Mee
Frace
acer
%24
96
8.
backspace
y
Transcribed Image Text:O Resources
Lx Give Up?
O Hint
82.2%
Assignment Score:
Question 6 of 23
>
он
OH
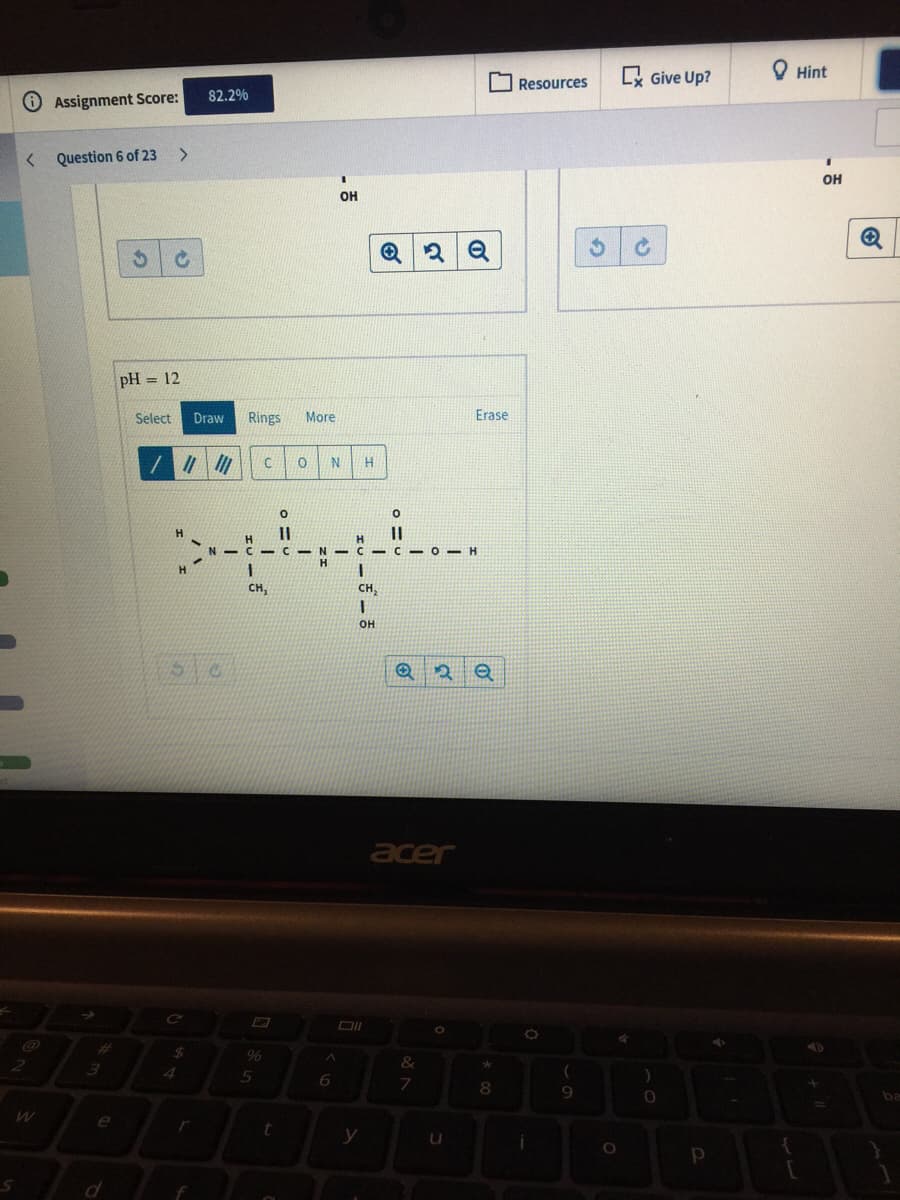
pH = 12
Select
Draw
Rings
More
Erase
H.
H.
H.
- C - N -C - C -0 - H
H.
CH,
CH,
он
acer
%23
%24
96
&
3
4
be
e
r
у
| O
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Biochemistry
Biochemistry
ISBN:
9781305961135
Author:
Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Publisher:
Cengage Learning

Biochemistry
Biochemistry
ISBN:
9781305961135
Author:
Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Publisher:
Cengage Learning