Which of the following alkene will form the most stable carbocation when reacts with HBr? Select one a cyclohexene b 2 heptene Ci methyleyclohexene. 1-nonane
Which of the following alkene will form the most stable carbocation when reacts with HBr? Select one a cyclohexene b 2 heptene Ci methyleyclohexene. 1-nonane
Chapter30: Orbitals And Organic Chemistry: Pericyclic Reactions
Section30.SE: Something Extra
Problem 36AP
Related questions
Question
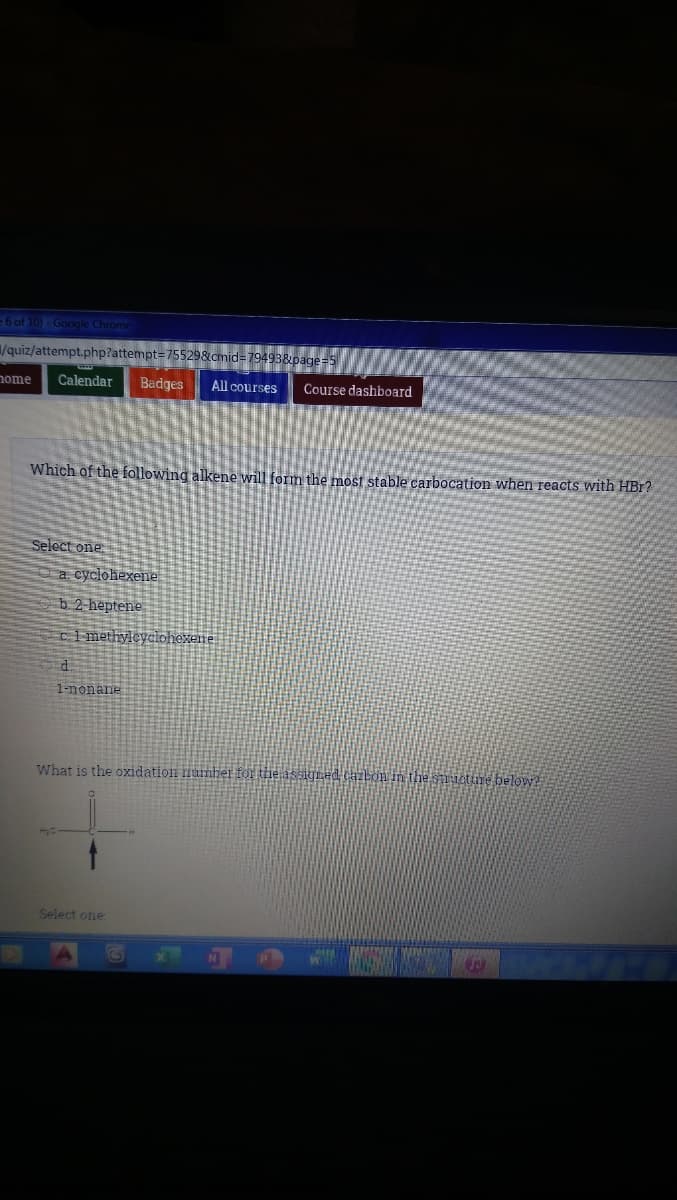
Transcribed Image Text:=6 of 10)- Google Chrome
/quiz/attempt.php?attempt=755298cmid=79493&page
Calendar
Badges
All courses
home
Course dashboard
Which of the following alkene will form the most stable carbocation when reacts with HBr?
Select one
a. cyclohexene
b 2 heptene
ci methyleyclohexene
1-nonane
What is the oxidation lumber fOr the assigned caibon in the snuetume below?
Select one
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

