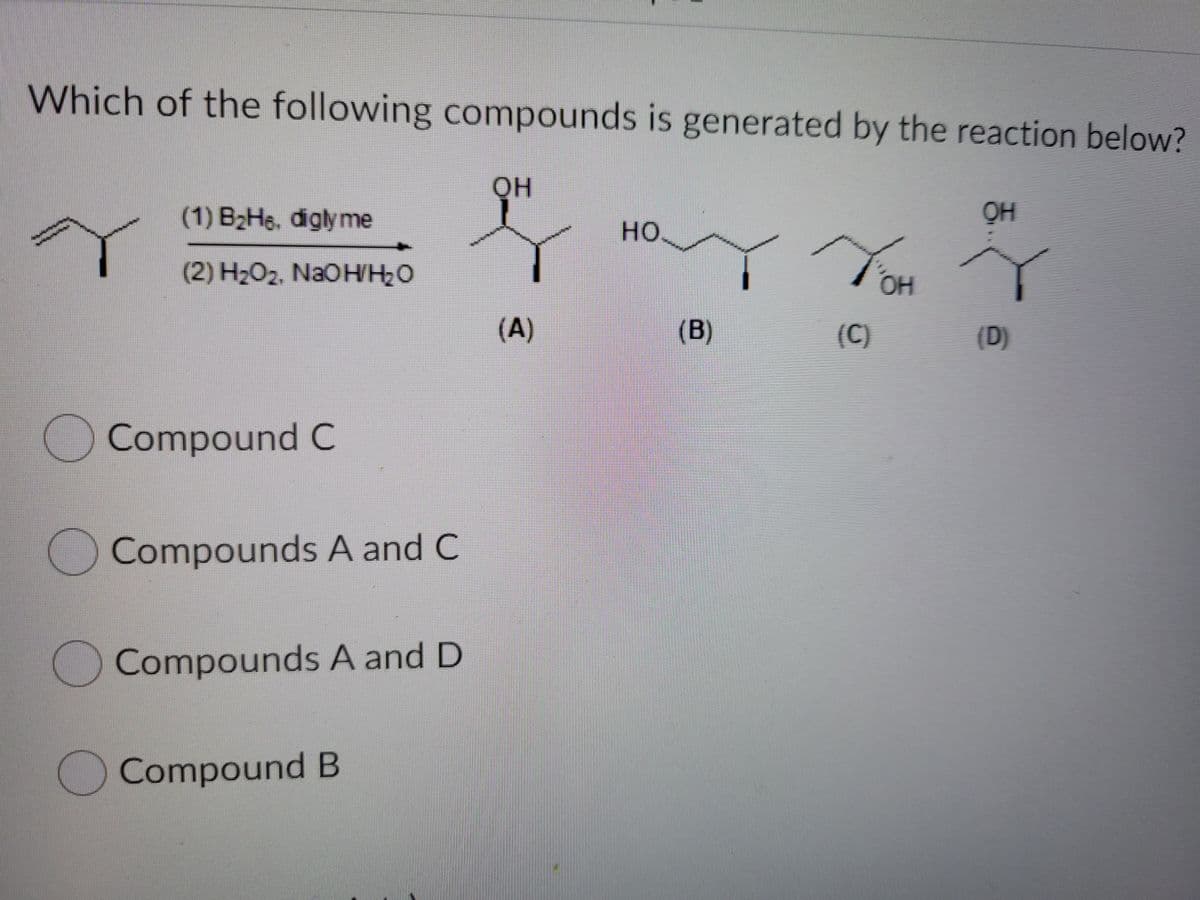
Which of the following compounds is generated by the reaction belo QH (1) B2H6, diglyme OH но. (2) H2O2, NaOHH2O OH (A) (B) (C) (D) Compound C Compounds A and C Compounds A and D Compound B
Q: o form H3PO4(aq) according to the following equation,
A: When one mole of P4O10(s) react with 6mol of H2O(l) then energy is evolved NOTE : When energy is…
Q: GIVE THE IUPAC
A:
Q: Compare the acidity of carboxylic acids with other common acids, and explain howtheir acidity varies…
A: INTRODUCTION: Carboxylic acids is defined as the organic compound that contain carboxyl group which…
Q: Complete the following reactions: Нeat C-N-CH, + NaOH a. CH, Heat b. C-N-CH, + H,O+ HCl CH,
A: Ans : A) : If N,N - Dimethylbenzamide is heated with sodium hydroxide solution, ammonia…
Q: коНӀaд) + НaSO4 (aq) —
A: Neutralization reactions are those which an acid reacts with base to form a salt and H2O. In…
Q: Consider NaHCO3(aq).
A: NaHCO3 . It can undergoes hydrolysis to give OH- and also it can give H+.
Q: a) describe with chemical equation how to prepare the following compound
A: Chemical equation: 2-phenyl-1-pyrroline was selectively dehydrogenated to 2-phenylpyrrole using…
Q: Acid Names IUPAC HCN(aq) Classical Acid Names IUPAC HCIO2(aq) Classical Acid Name IUPAC CH3COOH(aq)…
A: IUPAC name is the systematic name given to a chemical compound International Union of Pure and…
Q: B) O2N- CO D) H3C- -COOH
A: As we know, Carboxylic acid (-COOH) is more acidic than Alcohol (-OH). Now, In organic compound, if…
Q: If the Keq - 680, calculate the SO3 concentration at 100°C.
A:
Q: what is the conjugate acid of C6H5NH2?
A: Bronsted lowry concept of acid and base: An acid is defined as a substance that donates the hydrogen…
Q: Oxalic acid is a poisonous solid. It gives a yellow precipitate when treated with NH4OH and CaCl2. *…
A: Oxalic acid is a poisonous, colorless substance. Oxalate ion is a celating agent that forms…
Q: Which is the stronger base, (CH3)3N or H2BO3− and why?
A: Bronsted-Lowry acid-base theory: The Bronsted-Lowry acid-base theory states that the acid is a…
Q: Predict the major c Select Draw 1. NaOEt/E1OH 2. E1OCOCH,Br 3. dilute NaOH, heat 4. H30* 5. Heat но
A: Active methylene compounds are very acidic proton. Active methylene compounds have two acidic…
Q: How to prepare the compound (Pentyl Propanoate)
A: Pentyl propanoate can be synthesized in two ways.
Q: O 17. 1. CH;CO;H 2. H;O"
A:
Q: Write a complete balanced equation for the combustion of ethanol, the combustion of aspirin, and the…
A: A balanced equation is the equation in which the numberof atoms of each element are equal at both…
Q: What is the ionisable group of nalidixic acid? With illustrations
A:
Q: A substance which can be used with sulfuric acid to oxidize ethyl alcohol is O LIAIH4 O K2Cr207 O…
A: The structure of ethyl alcohol is as shown below.
Q: Rank the following groups in order of decreasing priority. −COOH, −H, −NH2, −OH
A: While assigning R and S configuration, the species with highest molar mass is given highest…
Q: name and draw the structural formulas for the 4 carboxylic acids with the molecular formula C5H10O2
A: 1.)The very first structure can be of a straight chain. Since the five carbon containing carboxylic…
Q: The reaction of C2O42- with H2O to H2C2O4 and OH-
A: The reactants given are 1) C2O42- 2) H2O And the products forming are given as, 1) H2C2O4 2) OH-…
Q: KMNO4, OH" cold and dilute cis pent-2ene + peroxyacetic acid in water
A: The products of given reaction are drawn as,
Q: Which one of the following is NOT true about chloramine? O a) HCIO and CIO- are the primary…
A: HClO and ClO⁻ are oxidizers, and the primary disinfection agents of chlorine solutions. Chloramines…
Q: state the Reaction of +NO with 2° Amines ?
A: Nitrous acid is a test used for the detection of primary, secondary and tertiary amines. It gives…
Q: Give three general preparation methods of binary hydrogen compounds. Give the general reaction…
A: The compounds containing only hydrogen atom bonded to another elements are known as binary hydrogen…
Q: Acetic acid to Terephthalic acid
A: In modern technologies : The terephthalic acid is prepared by catalytic…
Q: Write the basic equilibrium equation for a) (CH₃)₂NH b) C₆H₅O⁻
A:
Q: Phenol, C6H5OH, methanol, and CH3OH both have O-H groups, but phenol is a stronger acid. Draw the…
A:
Q: What is Terephthalic acid and how important it is?
A:
Q: ve three general preparation methods of binary hydrogen compounds. Give the general reaction…
A: The first element of the periodic table is hydrogen. Its atomic number and atomic mass is 1. The…
Q: 6. _(6a)_+ 20CUSO4. + 4NAOH → CH:COOH +_(6b)__ + 2NazSO4 + H2O
A: Following is the balanced chemical equation of the given chemical reaction.
Q: Provide the name of the product (besides water) that is formed in the following reaction:
A:
Q: КОН NAOH NH H2O 4. A В
A: 4. We have to predict the product for the following given reaction as follows in step 2 and
Q: Describe with chemical equations the preparation of the following compounds A B
A: Answer A: Retrosynthesis: Retrosynthesis is the method of "deconstruction" of a target molecule…
Q: Which of the follwoing compounds is responsible for the mass sepctrum and why? (a) octanol,…
A: Mass spectrometry is an analytical tool useful for measuring the mass-to-charge ratio (m/z) of one…
Q: i. NaH, DMF ii. CH3CH₂Br iii. NaOH, H₂O iv. H₂O*, heat ?
A:
Q: State the number of stereoisomers possible for this compound.
A: Given, Megatomoic acid is the sex attractant of the female black carpet beetle, has the following…
Q: The acid-catalyzed hydrolysis of an ester converts an ester into a carboxylic acid. Although there…
A: Esters are the organic compounds having to oxygen atoms (RCOOR), one is linked to carbon by a double…
Q: Question 3 of 19 Write a balanced chemical equation based on the following description: butanoic…
A: The balanced chemical equation when butanoic acid burns in air has to be given.
Q: Give an IUPAC
A: The IUPAC name of given compound is identified as,
Q: он (1) Hg(OAc)2, H2O OH но. (2) NaBH4lethanol OH (A) (B) (C) (D) Compounds A, C and D Compound B…
A: In this question, we want to determine the Products for this reaction. How we can determine, you…
Q: H3PO4 (A) (B) (C) (D) Compound B Compound C Compound D
A:
Q: Draw the structure of salicylic acid and methyl salicylate and circle all the acidic hydrogen atoms.
A: Given compounds are, salicylic acid and methyl salicylate
Q: Write the structural formulas of trichloroacetic acid and acetic acid and explain why…
A: Acetic acid is an organic compound containing a (-COOH) group present in it. The molecular formula…
Q: Draw and name carboxylic acidsand dicarboxylic acids, and usespectral information to determinetheir…
A: Acetic acid and oxalic acid are the example for the mon-carboxylic acid and dicarboxylic acid.…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Starting with acetylene and ethylene oxide as the only sources of carbon atoms, show how to prepare the compound Q.)HexanedialAn unknown hydrocarbon A with the formula C6H12 reacts with 1 molar equivalent of H2 over a palladium catalyst. Hydrocarbon A also reacts with OsO4 to give diol B. When oxidized with KMnO4 in acidic solution, A gives two fragments. One fragment is propanoic acid, CH3CH2CO2H, and the other fragment is ketone C. What are the structures of A, B, and C? Write all reactions, and show your reasoning.Compound A (C6H12O2) reacts with water, acid, and heatto yield compound B (C5H10O2) and compound C (CH4O).Compound B is acidic. Deduce possible structures of compounds A, B, and C
- An unknown hydrocarbon A with the formula C6H12 reacts with 1 molar equivalent ofH2 over a palladium catalyst. Hydrocarbon A also reacts with OsO4 to give diol B. When oxidized with KMnO4 in acidic solution, A gives two fragments. One fragmentis propanoic acid, CH3CH2CO2H, and the other fragment is ketone C. What are thestructures of A, B, and C? Write all reactions and show your reasoning.2-bromo-2-methylbutane undergoes hydrolysis reaction with water, H2O toform compound W. Compound X and compound Y are produced when 2-bromo-2-methylbutane undergoes elimination reaction with alcoholic ofsodium hydroxide, NaOH. (i) Draw the structural formula of compounds W, X and YHow do you decompose KHS2O3
- Borazole, B3N3H6, is an unusually stable cyclic compound. Propose a structure forborazole, and explain why it is aromatic.Compound AA has a molecular formula of C3H6O and gives a positiveresult using Tollen’s reagent. The reaction of compound AA with hotacidified potassium permanganate, KMnO4 gives compound BB. Thecatalytic hydrogenation of compound AA with nickel, Ni producedcompound CC. The reaction of compound BB with ethanamine,CH3CH2NH2 produces compound DD I) Draw the structural formula of compounds AA, BB, CC and DD. 2)Name the type of chemical reaction for the formation of compound CC.When alkyne A is treated with NaNH2 followed by CH3I, a product havingmolecular formula C6H10O is formed, but it is not compound B. What isthe structure of the product, and why is it formed?
- Rank the following acids in decreasing acidity. Explain your answer CH2ClCOOH, CHCl2COOH, CH3COOHHaving one mole of carbon suboxide (C2O3) means that you haveAn unknown hydrocarbon A with the formula C6H12 reacts with 1 molar equivalent of H2 over a palladium catalyst to give hydrocarbon B. Hydrocarbon A also reacts with OsO4 to give the glycol C. When oxidized with KMnO4 in acidic solution, A gives two fragments. One fragment is propanoic acid, CH3CH2COOH, and the other fragment is ketone D (R2C=O). What are the structures of A, B, C and D? Write all reactions.

