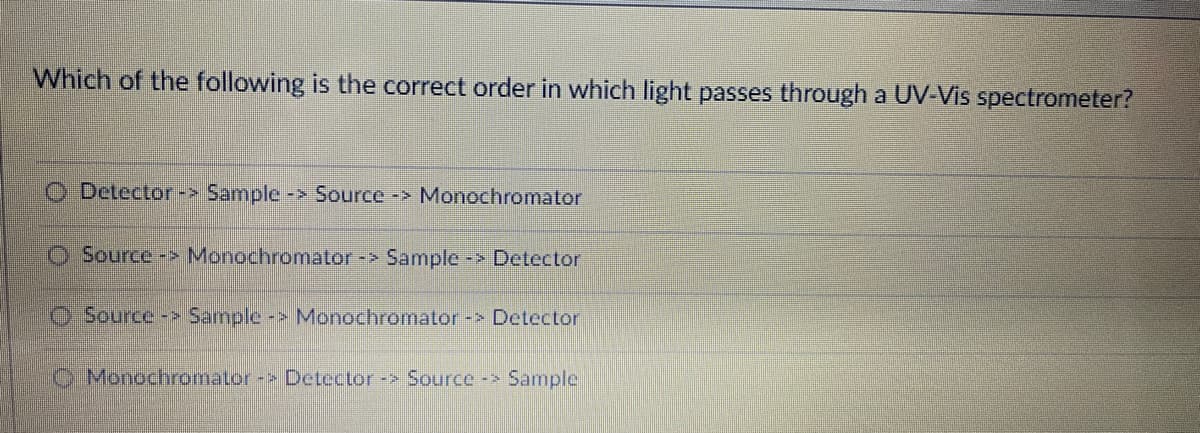
Which of the following is the correct order in which light passes through a UV-Vis spectrometer? O Detector -> Sample -> Source -> Monochromator O Source-> Monochromator -> Sample -> Detector O Source-> Sample -> Monochromator -> Detector O Monochromator -> Detector -> Source -> Sample
Q: Which of the following is the best Lewis structure? :0=Si -ö: o=o=si ESi= A B All have equal…
A:
Q: 1.Does O3+2H++2e-> O2+2H2O release more heat in comparison to O2+4H++4e-> 2H2O. Explain in great…
A: #1: The standard reduction potential for the two reduction half-reactions are: O3(g) + 2H+ + 2e-…
Q: 4. There are two isomers corresponding to the chemical formula C2H6O. One is an alcohol, and the…
A: The compounds with the same molecular formula but the bond connectivity is different, known as…
Q: Which of the following compounds is most acidic? OH II || II IV
A: Acid is the species which gives H+ ion in any solvent.
Q: 10. For the Tollowing, uSE UND a) Write out 6 completely different reactions of 3-pentanone…
A: Here the key functionality is ketone and the carbon atom present in it is very much facile towards…
Q: Calculate the enthalpy of the net reaction from the reactions given. 1) H2(g) + Cl½(g) → 2HCI(g) AH,…
A:
Q: The addition of 1.270 g of zinc metal (65.38 g/mol) to 0.100 L of 0.1942 M HCl in a coffee-cup…
A: We first need to decide on the limiting reagent, and then we can proceed to solve the reaction- The…
Q: What is the total number of valence electrons in the following strcuture? H. ö: || c-H H H. O 22 O…
A: In this question we have to tell the total number of valence electrons in the given structure.
Q: Glucose generally exists in ring (cyclic) form. A Haworth projection shows the orientations of the…
A: Blood sugar or glucose is the main sugar found in your blood. It comes from the food you eat, and is…
Q: Give the oxidation number of each element in the polyatomic ion below: sulfate nitrate carbonate…
A: Since you have asked multiple questions, we will solve the first question for you as per our…
Q: The table below shows the most commonly useful fusion reactions of the hydrogen isotopes deuterium…
A:
Q: 1. How many grams of P are present in 1.98 grams of tetraphosphorus decaoxide ? grams P. 2. How many…
A: Number of moles of a substance = Amount of substance/ molar mass of substance
Q: B) In finite potential barrier, how far a particle penetrate in to the barrier? What is the effect…
A:
Q: What volume of 0.150 M NaOH is needed to neutralize 35.00mL of 0.250 M H2S
A: Given data : For NaOH --> Molarity(M1) = 0.15M Volume(V1) =? For H2S ----> Molarity(M2) =…
Q: Label the structures to show the monosaccharide units produced by hydrolysis of the disaccharide.
A: Monosaccharides are the simplest carbohydrates that cannot be further hydrolyzed.
Q: The change in Gibbs' free energy is also considered a measure of the additional work that a system…
A: Gibb's free energy the measure of maximum non expansion work system can perform at constant…
Q: Avidin is commonly found in uncooked egg white. Consuming excessive amount of uncooked egg whites is…
A: Egg whites contain high levels of avidin, a protein that binds biotin strongly. When cooked, avidin…
Q: The Lewis structure of (NO+) ion is shown below. What is the hybridization and formal charge on…
A:
Q: What are the configurations of carbons 1 and 2 in the structure below? OH Br Et CH3 1S, 2R 1R 25 O…
A:
Q: Question 8 of 20 The AHvap of a certain compound is 38.49 kJ mol- and its ASap is 72.33 J- mol-.…
A: Given that dHvap = 38.49 KJ/mol dSvap = 72.33 J/molK = (72.33J×1KJ/1000J)/mol.K = 0.07233 KJ/mol.K…
Q: 2. The decomposition of N205 (2 N2O5 → 4 NO2 + O2) was carried out at several different…
A: Recall the given reaction, 2 N2O5 → 4 NO2 + O2We have to calculate the activation…
Q: 25. Provide the reagents necessary for the following transformation. OH ??? CI А. SOCI2, pyridine В.…
A:
Q: What is the percentage of ionization of 0.1 mo of formic acid (HCOOH)^5 . If you know that the value…
A: Given: Concentration of HCOOH = 0.10 M
Q: Consider the reaction described by the chemical equation shown. 3C,H, (g) → C,H,(1) AHan = -633.1 kJ…
A: ∆S°reaction = standard entropy of reaction = ∆S°product - ∆S°reactant ∆Sproduct = Standard molar…
Q: The change in Gibbs' free energy is also considered a measure of the additional work that a system…
A: Gibbs free energy is defined as: The amount of energy in a system that is available to do work. So…
Q: A tank contains 6OL of water contaminated with Mercury at a concentration of 0.03 mg/L. Clean water…
A:
Q: What is the osmolarity of a 2.50 M Mg3(PO4)2 solution ? The rx is given so you can get th conversion…
A:
Q: 4. Determine which classes of reactions would fit into each of the following categories for the…
A: Chemical Reaction : It is the process where one or more compound /substance (technically called…
Q: Guide Questions: Chemical Reaction: ZnCl2 + NaOH – NaCl + Zn(OH)2 Q1. What are the reactants and…
A: Here we are required to fin
Q: Which of the following bonds is the STRONGEST (NO) bond? (1) N=O (2) N=O (3) N-0 O 1 O 3 All have…
A: 1) Given :- The given bond are N≡O, N=O and N-O ----------------------------------
Q: Acidic solution such as 10-8 M acetic acid and 10-8 M HCI contains H* that is supplied mostly by a.…
A: Solution: ⇒We are given with the acidic solution of 10-8 M acetic acid and 10-8 HCl. Since its a…
Q: Question 4 of 23 > Monosaccharides Disaccharides Polysaccharides Answer Bank CH,OH CH OH HOCH2 OH…
A: Carbohydrates are optically active polyhydroxy aldehydes or ketones or such type of substances which…
Q: Question 4 of 20 The change in distribution of six gaseous particles between three interconnected…
A:
Q: a) A Beckman Type E glass electrode was used to measure the pH of a solution containing 1M Na* with…
A: In electrochemistry, glass electrodes are used to determine the pH of an unknown solution. The main…
Q: A biochemist completely digests a glycerophospholipid with a mixture of phospholipases A and D. HPLC…
A: A glycerophospholipid is a lipid where the Glyceral group is attached to a saturated fatty acid, an…
Q: De Broglie Hypothesis (Dual Narute of matter And Radi ohou) Ex plovn weu along with detailed…
A: Since you have multiple questions so I will solved first question for you please post separately…
Q: conductometric titration of strong acid with the strong base and weak acid with strong base the…
A: We have to have idea about conductometric titration. As we know when stong acid react with strong…
Q: When lead (II) nitrate reacts with sodium iodide, sodium nitrate and lead (II) iodide are formed.…
A:
Q: 5. When calcium metal is placed in water, a single-displacement reaction occurs. Write a balanced…
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: Determine whether each of the carbohydrates is a monosaccharide, disaccharide, or polysaccharide.…
A: Carbohydrates are optically active polyhydroxy aldehydes or ketones or those Substances which gives…
Q: c) Write out 3 preparations of 2-methylpropanoic acid, a different starting material for each one.…
A: Preperation of 2-methylpropanoic acid is given in three different ways -
Q: The unknown compound that gives the following IR and NMR spectra has the formula CaH1,0. Based on…
A:
Q: Ammonia would be considered a base under which of the following definitions? a. Arrhenius b.…
A: Dear student , As you have asked multiple question but according to guidelines we will answer first…
Q: Can you please help show how to use the given reactant in order to synthesize this molecule?
A: Alcohols react with alkyl halides to give an ether. Alkene undergo epoxidation followed by…
Q: CHM 131 Measurements Lab ent to the coned Part 1 • In lab, the following data was recorded.…
A: Given: Measurement Measurements Estimated digit location Number of sig figures Volume of salt…
Q: How much energy (in kJ) is required to heat 100.0 g H2O from a liquid at 76°C to a gas at 132°C?
A:
Q: For a chemical reaction, the rate constant at 43.2 °C is 0.003760 s1, while the rate constant at…
A:
Q: 10) Name the reaction and draw the product (do not draw the mechanism): NaOH, NaOCI CH,
A: The complete reaction has been given in the following step.
Q: b) Write out 3 preparations of 1-pentanol, a different starting material for each one. You may use…
A: Given, Compound = 1-Pentanol Write out 3 preparations of 1-Pentanol, a different starting material…
Q: 20. Provide the product resulting from the following Williamson Ether synthesis reaction. Br Br- Na…
A: Organic compound can be defined as the compound containing carbon and hydrogen atoms in a compound.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- which of the following statements is incorrect? A. a conventional mass spectrometer does not use a spectrophotometric detectorB. a conventional mass spectrometer does not always require high purity samplesC. a mass spectrum shows no signals due to uncharged speciesD. a conventional mass spectrometer uses high energy UV radiationWhich part of the instrument perform the given function introduction of a micro amount of sample converts sample components into gaseous ions orts the ions according to m/z converts m/z values to electrical signals converts signal/results into a mass spectrum A. mass analyzer B. Vacuum system C. Ion source D. Processor E. inlet system F. detectorsThe validation process comprises four main steps that are : hardware validation, software validation, method validation and system suitability. To evaluate a spectrophotometer’s accuracy by measuring absorbance, a solution of 60.06 mg/L K2Cr2O7 in 0.0050M H2SO4 was carefully prepared together with a 0.0050 M H2SO4 as a reagent blank. If the spectrometer’s signal is properly calibrated, it has been reported that this solution should provide an absorbance of 0.640 +- 0.010 at wavelength 350 nm. Which one of the validations mentioned above had been involved in this process? Explain your choice
- Where can I know the spectral properties of my sample? UV vis spectro, fluorescence spectro, Portable NIRTo determine the content of Pb in a canned sausage, 5.00 g were weighed and after adequate treatment they were gauged to a volume of 100 mL, from there an aliquot of 2 mL was taken and brought to a volume of 250 mL. The readings of a series and of the sample were taken in an Atomic Absorption spectrophotometer, recording the following data: mg Pb/mL 0 1 2 3 4 Sample Absorbance 0.00 0.15 0.29 0.44 0.58 0.38 What is the% Pb in canned sausage?1. An unknown sample of Cu2+ gave an absorbance of 0.262 in an atomic absorption analysis. Then 1.00 mL of solution containing 100.0 ppm Cu2+ was mixed with 95.0 mL of unknown, and the mixture was diluted to 100.0 mL in a volumetric flask. The absorbance of the new solution was 0.500. a) Denoting the initial unknown concentration as [Cu2+]i, write an expression for the final concentration [Cu2+]f, after dilution. Units of concentration are ppm. b) In a similar manner, write the final concentration of added standard Cu2+ designated as [S]f. c.) Find the [Cu2+]I in the unknown
- how the principle of spectrophotometric experiments? Explain plz thank youA solution with a known concentration equal to 3.4 mM gave an absorbance reading of 0.820 in a spectrophotometer. Assuming that there isa linear relationship between concentration and absorbance up to a concentration of 4.0 mM, what would the absorbance reading be for asolution with a concentration 1.5 mM?If a TOF mass spectrometer needs to scan an m/z of 600, what is the speed of this ion if it is accelerated through 4000 volts? How long will this ion (at the speed calculated) take to drift 2.00 m through the spectrometer? How many mass spectra can be taken each second if a new cycle begins each time this ion reaches the detector?
- . A sample of cobalt is to be determined using absorption spectrum analysis. A 0.286 g sampleof a cobalt ore is crushed and digested in 1M HNO3. The stock solution is then diluted to 250.0mL in a volumetric flask (assuming the solution has a density = 1.00 g/mL). The analysis thenproceeds as follows: Sample Solution Std Co solution (11.2 ppm) Final Volume Transmittance 15.0 mL 0.00 mL 100.0 mL 54.1% 15.0 mL 5.00 mL 100.0 mL 19.9%Calculate the percent cobalt in the original ore sample.A solution containing two different fluorescent compounds, Ben and Jerry, were analyzed for their individual concentrations in the mixture. Standards of pure Ben and pure Jerry were prepared at a concentration of 500.0 mM and were run in a UV-Vis Spectrophotometer to determine their absorption properties. Absorbance Wavelength Compound Ben 500 mM Compound Jerry 500 mM 400 nm 0.137 0.136 450 nm 0.312 0.113 500 nm 0.154 0.078 550 nm 0.076 0.079 600 nm 0.227 0.148 650 nm 0.230 0.230 700 nm 0.151 0.357 750 nm 0.157 0.246 800 nm 0.154 0.154 A standard curve of the standards was also prepared to help determine the concentration of each component in the solution. The solution produced an absorbance reading of 0.486 at the λmax of Ben, and 0.463 at the λmax of Jerry. STD CURVE BEN Λmax Ben Λmax Jerry STD CURVE JERRY Λmax Ben Λmax Jerry CONC (mM) ABS ABS CONC (mM)…A solution containing two different fluorescent compounds, Ben and Jerry, were analyzed for their individual concentrations in the mixture. Standards of pure Ben and pure Jerry were prepared at a concentration of 500.0 mM and were run in a UV-Vis Spectrophotometer to determine their absorption properties. Absorbance Wavelength Compound Ben 500 mM Compound Jerry 500 mM 400 nm 0.137 0.136 450 nm 0.312 0.113 500 nm 0.154 0.078 550 nm 0.076 0.079 600 nm 0.227 0.148 650 nm 0.230 0.230 700 nm 0.151 0.357 750 nm 0.157 0.246 800 nm 0.154 0.154 A standard curve of the standards was also prepared to help determine the concentration of each component in the solution. The solution produced an absorbance reading of 0.486 at the λmax of Ben, and 0.463 at the λmax of Jerry. STD CURVE BEN Λmax Ben Λmax Jerry STD CURVE JERRY Λmax Ben Λmax Jerry CONC (mM) ABS ABS CONC (mM)…



