Q: Using Markovnikov's' rule, where would the "X" of the "HX" compound bond in the addition reaction…
A:
Q: 54. is used in embalming, tanning. preparing glues, and polymeric products, as germicides,…
A: Carbonyl compounds: These are two types 1. Aldehydes 2. Ketone Aldehyde and ketones both…
Q: what is Anti-Markovnikoff's Rule ?
A: A Russian chemist “Vladimir Markovnikov” formulated a rule to identify the outcome of the some…
Q: What is Markovnikov’s rule?
A: ANSWERE IS GIVEN BELOW ;
Q: Describe the factors that lead to Markovnikov's Rule. Choose a reaction example that follows…
A: The Markovnikov's Rule States that, When a compound HX is added to an unsymmetrical alkene, the…
Q: Measured values at present indicate that the chain. . of polyethylene can be higher than 280 GPa.
A: Measured values at present indicate that the chain modulus of polyethylene is certainly higher than…
Q: Which group contains only examples of addition polymers? a. polyester, polystyrene,…
A: In this question we will see that the groups which are making by addition Polymerzation. You can…
Q: Markovnikov's rule can best be explained by considering; A. the number of hydrogen atoms attached to…
A:
Q: Sketch the repeat structure for each of the following alternating copolymers. a)…
A:
Q: In reference to cloth or fiber, the term acetate usually means cellulose acetate, a semisynthetic…
A:
Q: What is the basis or reason behind Markovnikov's Rule?
A: Statement : Markovnikov's rule states that When a monoprotic acid (HX) is added to an asymmetric…
Q: Which of the following three compounds: - polyethylene [essentially no vapor pressure at 1 atm] -…
A: Sublimation: Sublimation is a process in which the solid state of a matter is directly changed into…
Q: What is the reason behind Markovnikov's Rule? Please explain briefly and concisely.
A: The reason behind Markonikov's rule is the stability of carbo-cation formed during the reaction.
Q: What is a BH3 anti-markovnikov reaction in organic chemistry?
A:
Q: ·5'-G-G-A-T- T-A-C- T-A-T-G-C-3' Complimentary DNA sequence? e • How many hydrogen bonds would form…
A:
Q: A sample of polyethylene glycol is best measured by: a- ester value b- hydroxyl value c- acid…
A: Polyethylene glycol also referred as PEg is used in drugs in combination with PVAs that is polyvinyl…
Q: end groups contribute insignificant to the physical/chemical properties of polymers
A: According to the question we have to clearify the types of polymers end can contribute relatively…
Q: Upon heating with acid, salicylic acid can form a polymer. What is its structure likely to be?
A:
Q: What fragments are formed by dehydration of butan-2-ol?
A:
Q: Cellulose is a polymer of O amino acid fat alkene O alkynes O glucose QUESTION 8 The molecule below…
A: Cellulose is the major constituent of plant cell walls
Q: 1- Please find the typical molecular weight of the below common polymers. Compare them based on the…
A: 1.PET- molecular wt Between 8000 to 31000 2.PC-50000-300000g/mol 3.HDPE-200,000 -500,000g/mol…
Q: Polymers: 1. List the following group of monomers in decreasing order of the ability to undergo…
A: In cationic polymerization carbocation is formed which is stabilised by the attaching electron…
Q: Explain why styrene (CH2=CHPh) can be polymerized to polystyrene by all three methods of…
A: Chain-growth polymerization is a process in which the high molecular weight polymer is formed early…
Q: The tea leaves that contain the caffeine to be isolated are predominantly composed of cellulose, a…
A: Stability of following confirmation depends up on substitution location . Please follow 2nd…
Q: Is toluene reactive or not in Br2 in CH2Cl2: -with the presence of light -Without the presence of…
A: 1- With the presence of light : In the presence of light, toluene compound react easily with Br2…
Q: which involve .9 large number of small molecules called monomers to from molecule .high molecule…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Determine the Anti-Markovnikov Product prop-1-ene + H2O—> _________ (For Alkene and Alkyne…
A: In accordance with the Anti-Markovnikov rule, the anionic part of the attacking reagent will go to…
Q: Does Markovnikov's rule apply to the addition of HX to vinyl halides ?
A: Is Markovnikov's rule is applicable for the addition of HX to vinyl halide has to be given.
Q: Markovnikov's rule applies to; O addition of HBr to 1-butene O addition of Br2 to 1-butene O…
A: It discusses one of the fundamental rule associated with the addition reaction of olefins.…
Q: Which of the following statement is wrong for polymer additives? O a. It will not influence chemical…
A: Polymer additives are used to improve the usefulness of polymer and to protect polymers for long…
Q: According to Markovnikov's rule when HI (hydrogen iodide) adds to H3C-CH2-HC=CH2,, the main product…
A:
Q: What is the structure/linkage of sucrose?
A: Sucrose is a disaccharide which on hydrolysis gives the corresponding glucose and fructose. The…
Q: 7. A typical polymer may have hundreds to hundreds of thousands of repeating units called…
A: The given statements are A typical polymer may have hundreds to hundreds of thousands of repeating…
Q: Explain the Markovnikov's Rule ?
A: According to Markovnikov's rule, when a protic acid (HCl ,HBr) is added to an unsymmetrical alkene ,…
Q: Which is the structure for the product POLY? n H3C H₂C CH H3C CH3 C-C CH3 Br ~||||||~* JUDI H Br Br…
A: Here we have to write the structural formula of polymer formed in the following given polymerization…
Q: Draw a 6-monomer chain of polystyrene with "head to head" orientation.
A: Styrene is the monomer of the polystyrene. Polystyrene is formed by the free radical vinyl…
Q: What is the Reasoning behind Markovnikov's Rule
A: The result of some chemical addition reactions can be defined using Markovnikov's Law, also known as…
Q: (a) Differentiate between copolymerization and homopolymerization. Give one example of each.(b) What…
A: Polymerization is the process of combining smaller molecular units to form chains of molecules known…
Q: Explain why pKa of acetylene is 25, while that of ethene is 44
A: Relation of pKa and Ka:
Q: Q9: Polystyrene is soluble in benzene while poly (vinyl alcohol) is soluble in water. how will a…
A:
Q: How to apply the Markovnikov's Rule and predict the product(s) of an electrophilic addition reaction…
A: We have to Give how can we predict the product of the electrophilic addition reaction of an alkene.…
Q: Which of the following polymer structure the repeating units have no regular stereochemical…
A: Polymers that are formed by free-radical mechanisms such as polyvinyl chloride are usually atactic.…
Q: Why do some organic chemistry addition reactions require heat or refluxing while others can proceed…
A: Answer - According to the question - some organic chemistry addition reactions require heat or…
Q: What is the degree of polymerization of each of the following polymers with molar mass 100,000…
A: Degree of polymerization :- It is the ratio of molar mass of polymer to the molar mass of monomer…
Q: In reference to cloth or fiber, the term acetate usually means cellulose acetate, a semisynthetic…
A: The structure of Cellulose acetate :
Q: Q2/ What is the secondary bonds of the following polymers ? [ 15 Mark] i-Poly(methyl acrylate) ii-…
A: Secondary bonds are bonds of a different kinds to primary ones, they are weaker in nature and…
Q: Which is more reactive, monohydroxyl alcohol or polyhydroxyl alcohol? Why?
A: Alcohols , on the basis of number of hydroxyl groups attached , can be divided in two categories.…
Which of the following reactions is/are consistent with Markovnikov’s rule?
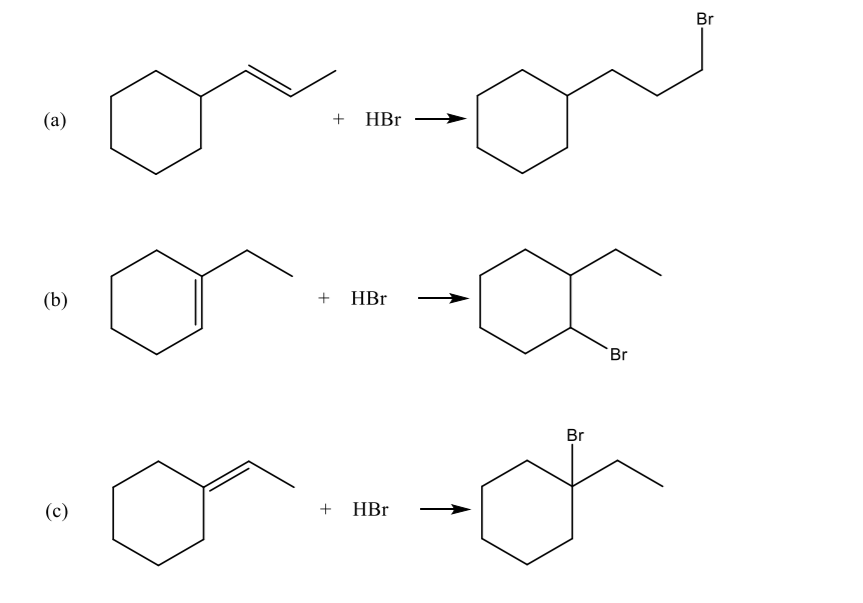
Step by step
Solved in 2 steps with 2 images

- Chlorine gas is used in the production of many other chemicals, including carbon tetrachloride, polyvinyl chloride plastic, and hydrochloric acid. It is produced from the electrolysis of molten sodium chloride. 2 NaCl(l) → 2 Na(l) + Cl2(g) What minimum mass of sodium chloride in megagrams is necessary to make 5.9 ✕ 105 L of chlorine gas at standard temperature and pressure, STP?what total volume of 20.6 % m/v sodium phosphate is needed to have a total of 69.5 g of sodium phosphateComplete and balance the following equations:(gas evolution) HCl + Na2SO3 ---->
- Vanadium could be oxidized to vanadium (II) ions thru the action of zinc powder and an acid True or false?is kalburo (calcium carbide) can be mix in fermented cow dung to generate fire?How many grams of potassium hydroxide, KOH , are required to make a 4.77 %(w/v) aqueous solution in a 250. mL volumetric flask? g

