Which of the following statements is true regarding the proton signals for H₂ and H₂ in the ¹H NMR spectrum of compound A? Ha A Ha and Hb would not couple to each other, as they are chemically equivalent via the symmetry of compound A The coupling constant for the Ha to Hb coupling (Jab) would be larger than for a typical linear chain, due their relative orientation in the most stable conformation of A Ha and Hb would both appear in the spectrum with a complex doublet of triplet of quartets splitting pattern The chemical shifts for H₂ and Hb would be greater than 2 ppm apart, due to their different relative distances from the t-butyl group H₂
Which of the following statements is true regarding the proton signals for H₂ and H₂ in the ¹H NMR spectrum of compound A? Ha A Ha and Hb would not couple to each other, as they are chemically equivalent via the symmetry of compound A The coupling constant for the Ha to Hb coupling (Jab) would be larger than for a typical linear chain, due their relative orientation in the most stable conformation of A Ha and Hb would both appear in the spectrum with a complex doublet of triplet of quartets splitting pattern The chemical shifts for H₂ and Hb would be greater than 2 ppm apart, due to their different relative distances from the t-butyl group H₂
Chapter95: Nuclear Magnetic Resonance Spectroscopy (proton Nmr)
Section: Chapter Questions
Problem 8P
Related questions
Question
The answer to the first one is not A or C.
The answer to the second one is not 5 or 8
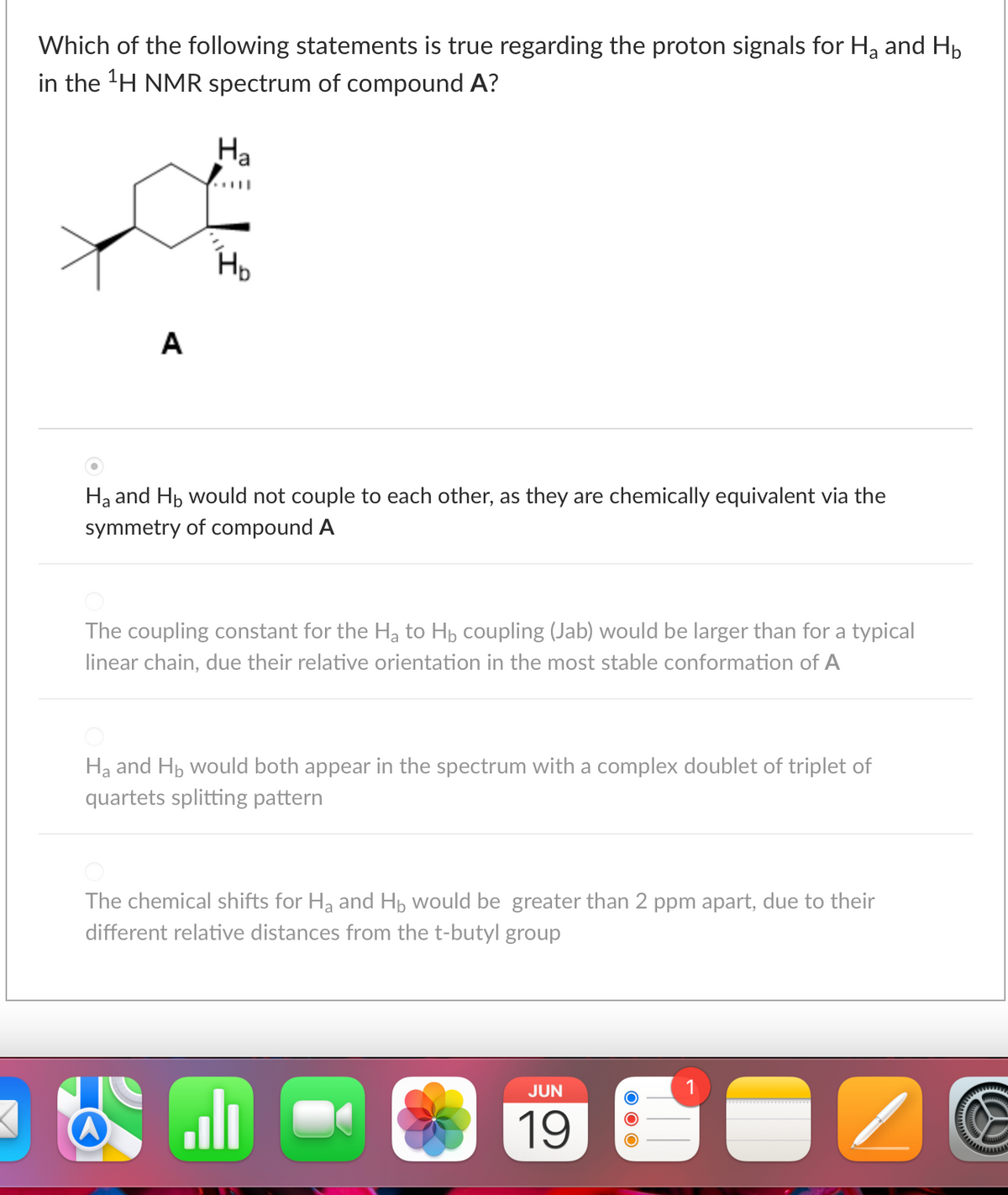
Transcribed Image Text:Which of the following statements is true regarding the proton signals for H₂ and H₁
in the ¹H NMR spectrum of compound A?
Ha
A
Ha and Hb would not couple to each other, as they are chemically equivalent via the
symmetry of compound A
The coupling constant for the H₂ to Hb coupling (Jab) would be larger than for a typical
linear chain, due their relative orientation in the most stable conformation of A
Ha and Hb would both appear in the spectrum with a complex doublet of triplet of
quartets splitting pattern
The chemical shifts for H₂ and Hb would be greater than 2 ppm apart, due to their
different relative distances from the t-butyl group
JUN
19
2
H₂
اله
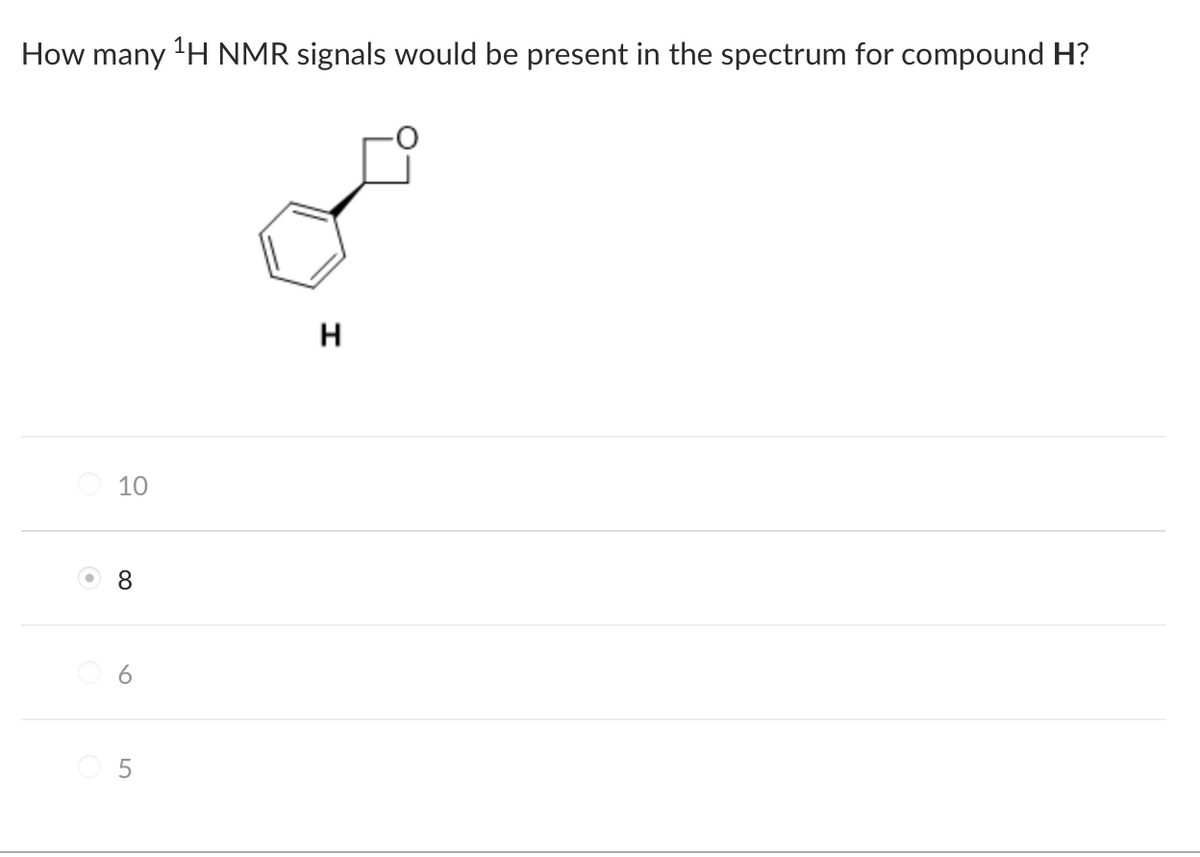
Transcribed Image Text:How many ¹H NMR signals would be present in the spectrum for compound H?
H
10
8
10
5
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT