Q: 7.5 g of acetic acid (C2H4O2) are diluted to 150 mL with water. What is the molarity of the acetic…
A: Answer. molarity is number of moles of solute dissolved per litre of solution. Unit of molarity : M…
Q: 4. How would you prepare 25.00 mL of a 0.500 M solution using a. Nitric acid from 15.8 M…
A: The question is based on the concept of solution preparation. we have been given requirements of the…
Q: what volume (L) of 0.404 M potassium hydroxide solution would just neutralize 60.6 mL of 0.388…
A:
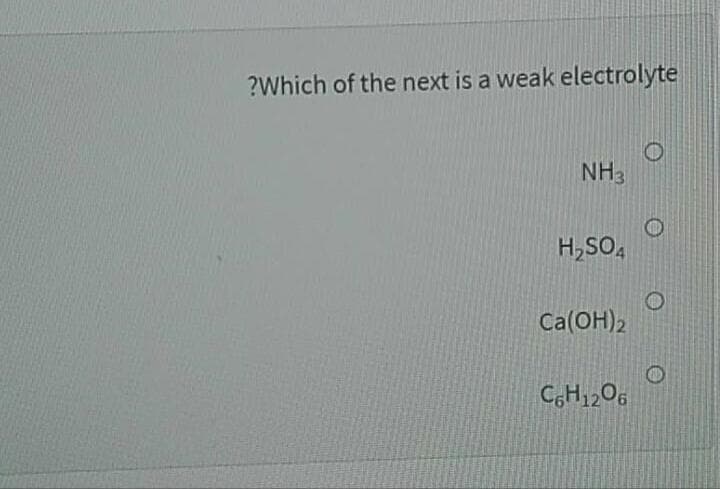
Q: Which of the next is a weak electrolyte? ealso explain it a) NH3 b) H2SO4 c) Ca(OH)2 d) C6H12O6
A: The weak electrolyte has to be predicted and the reason has to be explained.
Q: If you want to know the molarity of the solution of H2SO4 is accurate, What information do you…
A: Number of moles of solute present in 1 liter of solution is known as molarity of the solution.
Q: How would you prepare 275 mL of 0.600 M K,PO4 solution? * O I would add 25.0 g K,PO, to enough water…
A: The molarity is equal to the number of moles of solute dissolved per liter of solution. It is…
Q: 6. The mass of K3PO4 needed to prepare 250.0mL of an aqueous solution in which P0,3 concentration is…
A:
Q: Cu(C2H302)2 WOULD BE AN EXAMPLE OF A
A: Ionic solids that are 100 % soluble and break into corresponding ions along with conducting…
Q: 2. What volume in mL of 4.35 M HCI solution is needed to react with 18.5 g of Ca(OH), ?
A:
Q: اجابة قصيرة A12 (SO4) 3 at a N, What is the pare a solution n of 0.04 M in a i distilled water
A: Relation between normality vs Molarity Normality = n • Molarity Where : n is the n-factor for…
Q: What mass of sodium hydroxide should Lucy expect in 25 mL of a 3.25 M solution?
A:
Q: How would you prepare 275 mL of 0.600 M NazPO, solution? * O I would add 0.165 g Na,PO, to enough…
A: Given: Volume = 275 mL= 0.275 L Molarity = 0.600M To find: How would you prepare 275 mL of 0.600M…
Q: Which is classified as a weak electrolyte? A. F2 B. HF C. OBr2 D. CaF2 E. HBr
A: The substance which dissociate partially in solution to produce ions are known as weak electrolytes.…
Q: What volume of 1.2M potassium hydroxide is needed to neutralize 150mL of 3.2M sulfuric acid? Write a…
A: The balanced chemical reaction between potassium hydroxide (KOH) and sulfuric acid (H2SO4) is, 2KOH…
Q: 5) What volume of 1.00M NaOH is required to neutralize 35.0 ml of 0.102M hydroiodic acid, HI?
A: The interaction between an acid and a base is known as a neutralization reaction, and it results in…
Q: You have 125.0mL of 4.10M aqueous Mg(CIO3)2. Which of the following statements are true? O Mg(CIO3)2…
A:
Q: 1. Which of the following solutions would not be expected to exist? a. NaCl in CCI4 b. NaCl in water…
A: A solution is a homogeneous mixture of one or more solutes dissolved in a solvent.
Q: What volume (V) of o.20 M HNO, is needed to neutralize 150 mL of o.6 M NaOH?
A: The ratio of number of moles to the volume of solution in liters is called as molarity. Molarity…
Q: What volume (L) of 0.238 M potassium hydroxide solution would just neutralize 44.3 mL of 0.167 M…
A: Given: Concentration of KOH = 0.238 M Concentration of H2SO4 = 0.167 M Volume of H2SO4 = 44.3 mL…
Q: How would you prepare the molar solutions of followings? 1 mM NaOH IN H3PO4 i) ii) iii) 0.15 m KMNO4
A: The solutions are given below -
Q: molecule/not making ionsin water) is dissolved in 50 g of water (Kf of water = 1.86 °C). If the…
A: Mass of sample(w) = 2.154 g Assume molar mass is M Mass of water = 50 g = 0.050 Kg Molality(m) =…
Q: What is the molarity of an NaOH solution when 9.75 g of NaOH dissolves in 50 mL of H2O?
A:
Q: Calculate the mass (m/m) percent of sodium iodide in solution prepared by dissolving 6.43 g of NaI…
A:
Q: When preparing a 1.5M solution of Ammonium Sulfate (NH.)2SO, in 500mL, how many grams are needed of…
A: Ph commonly used in biochemistry experiments is 7.4 (Option A).
Q: What is the molality of ions in a 0.425 m solution of (NH₄)₃PO₄ assuming the compound dissociates…
A: (NH4)3PO4 -> 3NH4+ + PO43-
Q: 2. How many moles of HCI can you get from 55mL of a 0.10M flask of HCI? 3. What is the concentration…
A:
Q: What is the molarity of Q Solution preppared 25g 7e4aSの
A: Here we have to calculate molarity of solution-
Q: on 2 Which of the following substances would be a poor choice for making conductive solution O NAOH,…
A:
Q: The volume of 0.10N HNO3 which required to neutralize 50 ml of a 0.15N * :Ba(OH)2 solution is 50 ml…
A:
Q: Which one of the following can be classified as a strong electrolyte? A) C6H12O6 B) CH;OH C) NH,NO3…
A: A strong electrolyte is the one that readily dissociates into ions in solution.
Q: 3) Suppose that 15.0 ml of 0.0250M aqueous H SO, is required to neutralize 10.0 ml of an aqueous…
A: The amount of a substance that is dissolved in a given volume of the solvent to furnish the solution…
Q: 3) Suppose that 15.0 ml of 0.0250M aqueous H,SO4 is required to neutralize 10.0 ml of an aqueous…
A: Given: M1 = 0.0250 M V1 = 15.0 mL V2 = 10.0 mL
Q: 1. How does the increase in solute concentration affect the conductivity of that solution?
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: In an aqueous solution at 25°C, if [H;O*] = 2.5 × 10-* M, then [OH]] is: %3D
A: The equilibrium expression is shown below: Kw = [H3O+] [OH-] Where; Kw is the ionic product constant…
Q: What is the molarity of the solution prepared by concentrating, by evaporation of solvent, 755 mL of…
A: Molarity can be defined as number of moles of solute dissolved per liter of the solution That is…
Q: What volume (L) of 0.236 M potassium hydroxide solution would just neutralize 49.3 mL of 0.217 M…
A: The reaction between H2SO4 and KOH is given as H2SO4 + 2 KOH ---> K2SO4 + 2 H2O Hence from the…
Q: 2. You take 38mL of a 0.45M solution ofH,PO, and react it with 22mL of Ba(OH), Calculate the…
A: 2H3PO4 + 3Ba(OH)2 ---> 6H2O + Ba3(PO4)2 Number of moles of Ba(OH)2 = 0.038 L H3PO4 × ( 1 mole…
Q: Which of the diagrams in the figure best represents an aqueous solution of MgCl2?
A: MgCl2 is an inorganic salt consisting of cation Mg2+ and two Cl-. In terms of atomic size, the size…
Q: How many mL of solution can be prepared from b. 1.23 Kg of BaCl2 if a 6.0M solution is needed?
A:
Q: What is the molality of lithium ions in a 0.302m solution of li3po4 assuming the compound…
A: Given: Molality of Li3PO4 = 0.302 m
Q: How many carbonate ions are present in 30.0 mL of 0.600 M K 2CO3 solution?
A:
Q: If 22.22mL of a 1.50 M Ni(NO3)2 solution is diluted to exactly 250.0mL, what will be the…
A: We have to calculate the concentration of dilute solution.
Q: 3) Assuming the density of the phosphoric acid is 1.07, what is the % H3PO4 of the solution? mL
A: Solution - According to the question - Given value - Density of the phosphoric acid = 1.07 g/ml…
Q: What is the molar concentration of a Solution mae by, dissolnng 0.235 grams of MgSO4 in enough Water…
A: Given : Mass of MgSO4 = 0.235 g Volume of solution = 250.0 mL = 0.250 L…
Q: Calculate the normality of a) 0.85 M H,C,H,O, b) 1.20 M Ba(OH),.
A:
Q: What is the final molarity of a solution prepared by diluting 35mL of 12.0M HCl to a final volume of…
A:
Q: Tuhy is [tooJ-CCH-J=1.0xl0 17 not fessible in wirter? IShat word hoppen it wetred to malke sueh a…
A: Since the water has an equilibrium with H3O+ and OH- ions as per the reaction 2 H2O (l)…
Q: What volume (L) of 0.318 M potassium hydroxide solution would just neutralize 82.4 mL of 0.201 M…
A: As per the reaction written below, 1 mole of H2SO4 reacts with 2 moles of KOH.
Q: MgBry solution. K water 1.86 'Cim K, water 0.512 "Cim O 9.9 "C 5. 6'66 O 102.1 "C 101.5 C O 101.1 C
A:
Step by step
Solved in 2 steps

- Which of the next is a weak electrolyte? ealso explain it a) NH3 b) H2SO4 c) Ca(OH)2 d) C6H12O6Which of the following schematic drawings best describes asolution of Li2SO4 in water (water molecules not shown forsimplicity)? [Section 4.1]what volume (L) of 0.404 M potassium hydroxide solution would just neutralize 60.6 mL of 0.388 MH2SO4 solution?
- 3) Suppose that 15.0 ml of 0.0250M aqueous H SO, is required to neutralize 10.0 ml of an aqueous solution of KOH What is the molarity of the KOH solution?The concentration of magnesium ions in seawater is approximately 0.0541 M. To the solution in part 3 above with MgSO4•7H2O (Epsoms Salt), how many more grams of MgCl2•6H2O will you need to use to make 1.0 L of water with the same concentration of magnesium ions as seawater? (I am reasking this question since the one I posted was never answered, how do I cancel the previous, duplicate question please?)Draw the Lewis structures for each of the species below, then State the molecular arrangement and shape of each species.1.KrF22.BrCl53. PO4-3 (Give clear handwritten solution)

