Which statement best describes the bond length(s) in the ozone molecule (shown)? The bond length of O-O is 148 pm and O=O is 121 pm. Ö= .0 0. O O There are two bond lengths that oscillate between 148 pm and 121 pm The bond lengths are equally 269 pm One bond is 148 pm and the other is 121 pm O The bond lengths are equally 127 pm.
Which statement best describes the bond length(s) in the ozone molecule (shown)? The bond length of O-O is 148 pm and O=O is 121 pm. Ö= .0 0. O O There are two bond lengths that oscillate between 148 pm and 121 pm The bond lengths are equally 269 pm One bond is 148 pm and the other is 121 pm O The bond lengths are equally 127 pm.
Chemistry: An Atoms First Approach
2nd Edition
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Steven S. Zumdahl, Susan A. Zumdahl
Chapter3: Bonding: General Concepts
Section: Chapter Questions
Problem 92E: Consider the following bond lengths: CO143pmC9O123 pmC:O109 pm In the Co32 ion, all three C8O bonds...
Related questions
Question
59
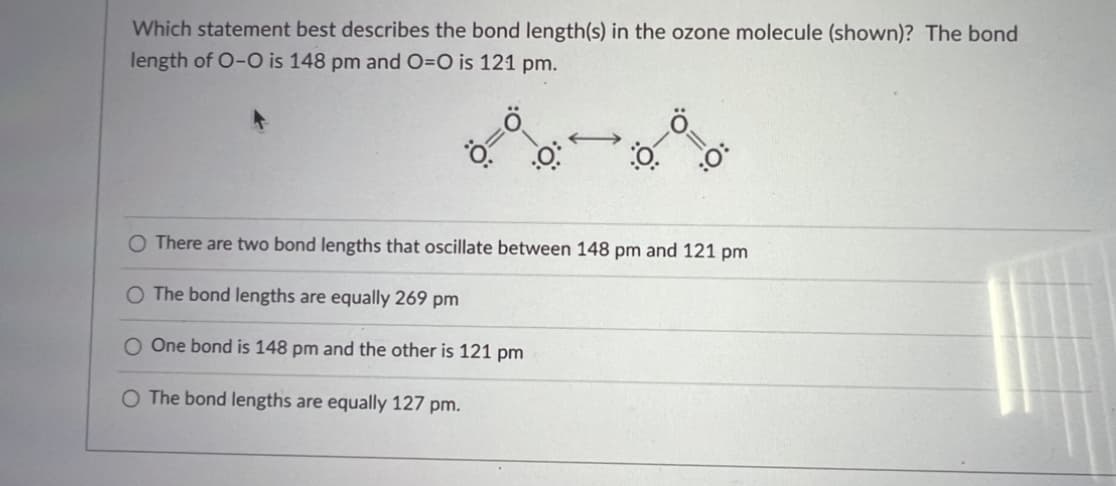
Transcribed Image Text:Which statement best describes the bond length(s) in the ozone molecule (shown)? The bond
length of O-O is 148 pm and O=O is 121 pm.
Ö=
.0
←
:0-0
:O:
O There are two bond lengths that oscillate between 148 pm and 121 pm
O The bond lengths are equally 269 pm
O One bond is 148 pm and the other is 121 pm
O The bond lengths are equally 127 pm.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning