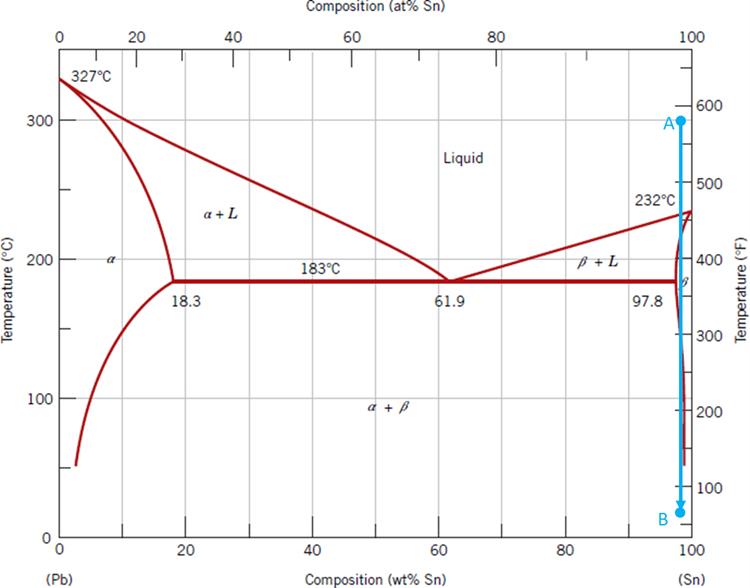
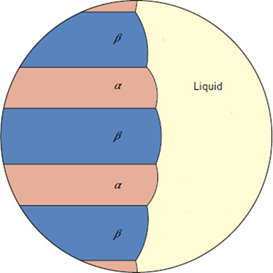
wing three phases are at equilibrium at the eutectic point. Which phase will have the highest solubility of Sn? Group of answer choices Beta Phase
Q: How do you call in binary phase diagram, the line which is described as the locus of all system…
A:
Q: (b) Explain with the aid of neatly drawn binary phase diagrams the following terms? i. Isormophous…
A: i) In isormophous system of alloys the two elements added will exhibit complete solubility in both…
Q: A hypothetical A-B alloy of composition 54 wt% B-46 wt% A at some temperature is found to consist of…
A: Given data as per the question Percentage of A =54% Percentage of B = 46% mass fraction =0.5
Q: Which of the following is true about Alloy in general? a. is it a pure metal with only one element…
A: Which of the following is true about Alloy in general? a. is it a pure metal with only one element…
Q: Consider an A-B phase diagram. At what temperature do we observe the maximum L L+a L+B solubility of…
A: As per given question We have to choose correct options and explain
Q: For a Cu-Ag alley of composition 30 wt% Ag 70 wt% Cu and at 775°C do the following: (a) Determine…
A: Above problem can be solved by using lever rule , composition can be taken from phase diagram of Cu…
Q: a binary phase diagram ( pressure omitted) what is the maximum number of phases that can coexist for…
A: The two components system are called binary system. The restriction on constant temperature and…
Q: 2) Consider a copper-nickel (Cu-Ni) alloy composed of 45 wt% Ni:55 wt% Cu that is at equilibrium at…
A: Given: The wt % of nickel in copper = 45wt%Ni and 55wt%Co The temperature, T= 1200 C
Q: Conducting the inverse lever rule on a phase diagram helps determine a) The amounts of phases b) The…
A: Conducting the inverse lever rule on a phase diagram helps determine the - The amounts of…
Q: 9.18 A magnesium-lead alloy of mass 5.5 kg consists of a solid a phase that has a composition that…
A:
Q: b) At a temperature just above the isotherm at 1830C, for the 30 wt% Sn Alloy, calculate the…
A: Given data: The temperature of the composition at 183°C. The composition weight% of Sn alloy is,…
Q: A) Identify all congruent reactions and their coordinates in the phase diagram B) Identify all…
A: As the question contains multiple sub-questions so solving only C1.C2 and C3 If want any specified…
Q: What are the equilibrium phases, compositions, and phase fractions in a sample with overall…
A: Given data To determine the phase, Compositions and phase fractions in a sample of 40%wtCu at 6000C
Q: 12) If you have an alloy with a composition of 50 wt% Ag at a temperature of 778.9° C, identify: a)…
A: Given for a Cu-Ag phase wt% of Ag is 50% Temperature = 778.9 degree C Eutectoid Temperature = 779…
Q: For a Pb-30% Sn alloy, determine the phases 400 present, their amounts, and their compositions at…
A: Data given- Pb - 30 % Sn alloy Temprature = 300°C ,200°C , 0°C
Q: 1.b. By using the Iron-Carbon (Fe-C) phase diagram provided herein, answer the following questions.…
A: Since you have posted a question with multiple sub-parts, we will solve first three sub- parts for…
Q: Arrange in a table the number of phases that can be observed at R.T in the range starting from zero…
A: Hello. Since you have posted multiple questions and not specified which question needs to be solved,…
Q: c) The Cu-Zn phase diagram is shown in Figure 2. i) For a sample of composition 40 wt% Zn and 60 wt%…
A:
Q: 1-Pointed all the phases for this type of phase equilibrium diagram. 2-Draw the cooling carves for…
A:
Q: By referring to Pb-Sb phase diagram, which equation refers to the eutectic reaction? liquid → alpha…
A: Option is correct Liquid----- alpha+beta
Q: The microstructure of a steel consists of 100% coarse perlite. According to this information, it is…
A: A phase diagram of an alloy is a graphical representation of all the phases present in the alloy…
Q: Construct the hypothetical phase diagram for metal A and B between temperature of 600 C° and 1000 C°…
A: according to the given question
Q: Choose the corect answer -A vertical line in the phase diagram represents a.an intermetallic…
A: Phase Diagram :- A phase diagram in material science is a type of chart which shows material…
Q: Complete the following table for water T, °F P, psia U, Btu/lbm phase description 300…
A: From Saturated Water-Temperature table at 300oF we get the specific internal energy values as,…
Q: a) Indicate hypooeutenic, hypereutenic ranges and eutenic composition in wt%Sn. Use the phase…
A: a) Micro structure of alloy is given as,
Q: Give an example of phase equilibrium and chemical equilibrium?
A: Phase equilibrium The phase equilibrium comes in the scenario when a substance is having more than…
Q: 10.15) A magnesium-lead alloy of mass 5.5 ka consists of a solid a phase that has a compo- sition…
A: Given, Mass of alloy = 5.5 kg
Q: Material science The figure given below shows the Pb-Sn binary phase diagram. a) Cite the phases…
A: According to the information we know at 250C Ts%W In the above graph, we can see that at 250 C it…
Q: Vhich is NOT a way to change the number of phases in a phase diagram? a) Change composition b)…
A: Correct option is (d) Change time duration.
Q: What is the maximum number of phases that can be in mutual equilibrium in a four-component system (…
A:
Q: Determine the composition of the phases;
A: Phase Equilibrium: Equilibrium is the lowest energy state for a given T, P, and composition (i.e.,…
Q: Describe the different phases in the Fe-Fe-C diagram?
A: This diagram is known as iron carbon phase diagram.
Q: c) The Cu-Zn phase diagram is shown in Figure 2. i) For a sample of composition 40 wt% Zn and 60 wt%…
A:
Q: A) Draw the Iron Carbon Phase Equilibrium Diagram? B) Define the Arrest points?
A: A)The Iron Carbon Phase Equilibrium Diagram
Q: What do you call the diagram in which the solid-liquid and vapor co-exist in equilibrium? a. water…
A: Given data To select the correct option
Q: An alloy system consists of two metals A&B which are soluble in each other in all proportions in the…
A:
Q: An alloy of Component A and Component B, which do not react but are miscible in all proportions in…
A: Phase Diagrams: It is an equilibrium diagram which represents the composition of constituent…
Q: An alloy of 30 wt % Pb and 70 wt % Sn is slowly cooled from 250°C to 27°C: a) Is this alloy…
A: As per our company guidelines we are supposed to answer only first 3 sub-parts. Kindly repost other…
Q: composition of the phase boundary (or solubility limit) f
A: Given: Alloy composition 60 wt% A-40 wt% B with alpha phase fraction 0.55 and beta phase fraction…
Q: Using the Mg-Pb phase diagram below, if you start at point A and cool down very slowly to room…
A: As we are using the graph for determine the phase and weight % and weight And Drawn the…
Q: Consider the binary alloy phase diagram shown schematically below. The left-hand side denotes phase…
A:
Q: (Solution Needed) An unknown compound is at 200°C and 1 MPa. What is the maximum number of phases…
A: GIVEN DATA- A COMPUND GIVEN AT PRESSURE 1MPa AND 200'C WE HAVE TO SELECT MAXIMUM NO. OF PHASES…
Q: Material science Assuming this system forms a laminar type eutectic, determine the volume proportion…
A:
Q: Phases that exist on the right side of eutectic point of an invariant reaction line are called:…
A: Option d is correct.
Q: A hypothetical eutectic phase diagram for metals A and B shows that cooling a liquid melt with a…
A:
Q: What is neutral equilibrium?
A: given: What is the neutral equilibrium?
Q: An intermetallic compound is represented by what on a phase diagram? a) Horizontal line b) Vertical…
A: Correct option is (b) by vertical line .
Q: (a) What is the difference between the states of phase equilibrium and metastability?
A: Solution: Note: Dear Student! As per our guidelines, we are only allowed to answer a single…
Assume that the following three phases are at equilibrium at the eutectic point. Which phase will have the highest solubility of Sn?
Step by step
Solved in 2 steps with 1 images

- A forensic chemist is given a white solid that is suspected of being pure cocaine (C17H21NO4 , molar mass = 303.35 g/mol). She dissolves 1.22 ± 0.01 g of the solid in 15.60 ± 0.01 g benzene. The freezing point is lowered by 1.32 ± 0.04°C.a. What is the molar mass of the substance? Assuming that the percent uncertainty in the calculated molar mass is the same as the percent uncertainty in the temperature change, calculate the uncertainty in the molar mass.b. Could the chemist unequivocally state that the substance is cocaine? For example, is the uncertainty small enough to distinguish cocaine from codeine (C18H21NO3, molar mass = 299.36 g/mol)?c. Assuming that the absolute uncertainties in the measurements of temperature and mass remain unchanged, how could the chemist improve the precision of her results?Given the Ultimate analysis of Alabama coal: Sulfur = 1.26% Hydrogen = 5.29% Ash =5.19% Carbon = 79.71% Nitrogen = 1.42% Oxygen = 7.13% Moisture = 4.80% Calculate the following: a) The ash and moisture free analysis b) Actual A/F ratio at 20% excess air c) Gravimetric Analysis of wet gas d) Volumetric Analysis of dry gas e) LHV of the fuel in kJ/kgA certain coal has the following ultimate analysis: Carbon = 69% Nitrogen = 5% Hydrogen = 2.5% Sulfur = 7% Determine the amount of oxygen in decimal form if the heating value of fuel is 26963.56 kJ/kg.
- Sling Psychrometer Readings (use the table on the next page to find the Relative Humidity) Trial Outside conditions Wet Bulb Temperature Dry Bulb Temperature Relative Humidity % 1 Mostly Sunny 14 C 20 C 2 Partly cloudy 16 C 20 C 3 Sunny 10 C 22 C 4 Cloudy 22 C 22 C PART A: Using the data above, if the Dry and Wet Bulb Temperatures are close to each other, what does that mean about the Relative Humidity? What does the Dry Bulb Temperature relate to? On a day that it is sunny outside, would you expect the wet and dry bulb temperature to be close to, or far from each other? PART B: If the Dewpoint Temperature and Temperature are far from each other, what does that mean about the relative humidity? On a day that it is raining, would you expect the Dewpoint Temperature and Temperature to be close to each other, or far from each other?Pls. answer thank you! A fuel oil C12H26 is used in an internal combustion engine and the Orsat analysis are as follows: CO2 = 12.896: O2 = 3.5%; CO = 0.2% and N2 = 83.5%. Determine the percent excess air.Pls answer thank you! The following is the ultimate analysis of a sample of petrol by weight :C = 84.2 %; H = 15.8 %. Calculate the ratio of air to petrol consumption by weight if the volumetric analysis of the dry exhaust gas is :CO2 = 11.07 %: C0 = 1.23 %: O2 = 3.72 %; N2 = 83.97 %.
- In an environment, the temperature suddenly increased from 100°C to 200°C. In order for a thermoelement in the environment to show 199.4°C in 8 seconds, how many seconds must the time constant of the thermoelement be? a. 1.919 b. 1.609 c. 1.630 d. 1.563 e. 1.955The materials laboratory that you direct received the following data on the tensile strength in MPa of 15 samples of Aluminum Nitride. The data obtained for each sample are: Sample Endurance (Mpa) SAMPLE (STRENGTH) MPa 1 190 2 233 3 268 4 160 5 257 6 300 7 310 8 262 9 185 10 205 11 275 12 245 13 220 14 290 15 228 You are asked to calculate the Weibull modulus, m, considering that the probability of failure is n/16, where n is the number of specimens that failed at a given stress. You can use Excel to speed up the calculations.a) At an engine speed of 1000 rpm, what is the approximate penetration depth of the temperature fluctuations in (a) a cast iron block and (b) an aluminum engine block? b) Measurements of the exhaust gases of a hydrogen-fueled engine indicate a composition of 71.0% N2, 25.0% O2, and 3.9% H2O. At what equivalence ratio was the engine operated?
- Macro farms Ltd can purchase two types of fertilizer which contain the following percentage Nitrate Phosphate Potash Type X 18 5 2 Type Y 3 2 5 For certain type of crop, the following minimum quantities (kg) are required Nitrate Phosphate Potash 100 50 40 Type X cost GHS10 per kg and type Y cost GHS5 per kg. The company currently buys 1,000 kg of each type and wishes to minimize it expenditure on fertilizers.Required 1. What is the minimum expenditure that can be incurred on fertilizer? 2. Find the savings the company can make by switching from it current policy to your recommendation.Material T, oC P, kPa v, m3/kg x Phase description Water 60 0.25 Water 180 100 R-134a 24 0.004 Fill out this table by using thermodynamic tables and show all details, calcuations and tables used to find the missing values.Suppose we have a piece of a candy bar that has an initial mass of 28g. If we ignite the piece of candy bar (literally burn it), and it increases the temperature of 285.23g of water from 18.95°C to 77.43°C, how many calories per gram of energy did the candy bar provide if the final mass of the marshmallow is 4.45? Note: 1.00 cal = 4.184 J. Give your answer in units of cal/g.

