WORKSHEET 1. Classify then name the following substances. Referto your flowchart. Classification Name Chemical Formula SUIFUK dioxide 1odine SO2 moeculaR 12 moceculaR lonic acid SUIFURIC acid Potarrium SUIFate H2SO4 ionic/acid K2SO4 copper (11) Chkomate Cobalt CI) chRomate CuCrO4 1onic Co2(CrO4)3 moecuia R CH;CH3 ethane
WORKSHEET 1. Classify then name the following substances. Referto your flowchart. Classification Name Chemical Formula SUIFUK dioxide 1odine SO2 moeculaR 12 moceculaR lonic acid SUIFURIC acid Potarrium SUIFate H2SO4 ionic/acid K2SO4 copper (11) Chkomate Cobalt CI) chRomate CuCrO4 1onic Co2(CrO4)3 moecuia R CH;CH3 ethane
Chapter12: Gravimetric Methods Of Analysis
Section: Chapter Questions
Problem 12.20QAP
Related questions
Question
I just need to know if these are all correct and I need help with just the blank ones.
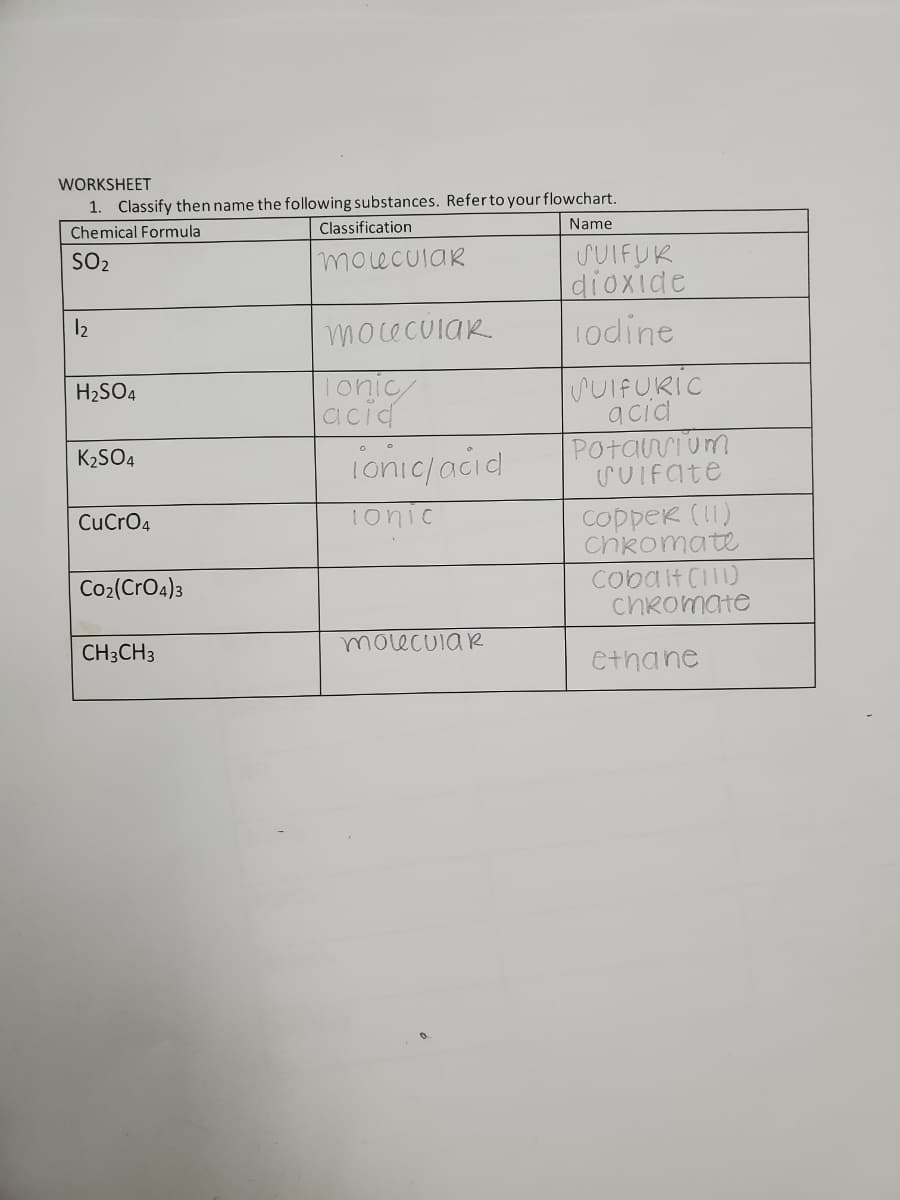
Transcribed Image Text:WORKSHEET
1. Classify then name the following substances. Referto your flowchart.
Classification
Name
Chemical Formula
SUIFUK
dioxide
1odine
SO2
molecular
12
moceculaR.
lonic
acid
SUIFURIC
H2SO4
acid
POtarium
SUIFate
ionic/aciel
K2SO4
Tonic
copper (11)
ChRomate
Cobalt Ci)
chRomate
CuCrO4
Co2(CrO4)3
molecuia R
CH3CH3
ethane
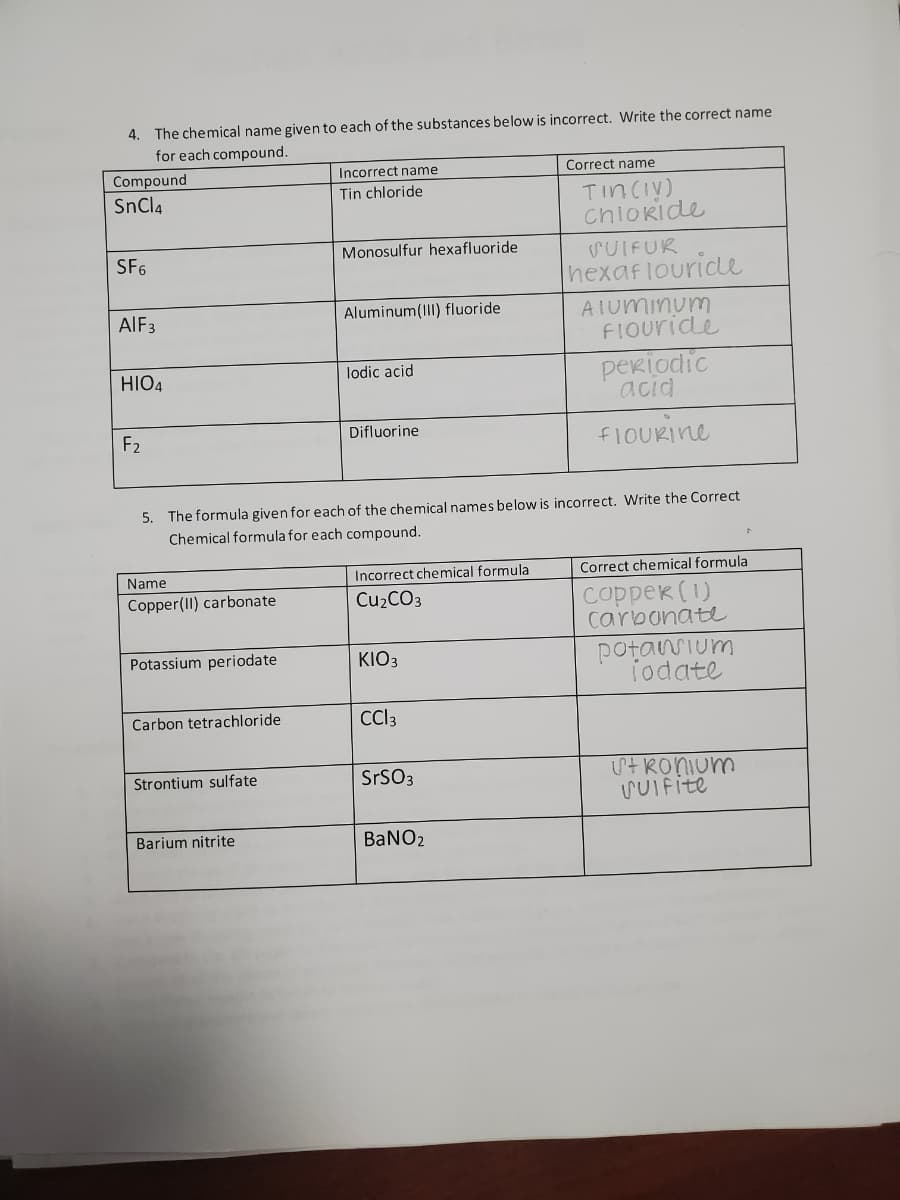
Transcribed Image Text:4. The chemical name given to each of the substances below is incorrect. Write the correct name
for each compound.
Incorrect name
Correct name
Compound
SnCl4
Tin chloride
TinCIy)
chlokide
SF6
Monosulfur hexafluoride
SUIFUR
hexaflouride
Aluminum(III) fluoride
AIuminum
FIOuride
AIF3
periodic
acid
HIO4
lodic acid
F2
Difluorine
FIOURINE
5. The formula given for each of the chemical names below is incorrect. Write the Correct
Chemical formula for each compound.
Name
Incorrect chemical formula
Correct chemical formula
соppек (1)
carbonate
Copper(II) carbonate
Cu2CO3
potanium
iodate
Potassium periodate
KIO3
Carbon tetrachloride
Cl3
itRonium
SUIFite
Strontium sulfate
SrSO3
Barium nitrite
BANO2
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

