For Hydrogen D, could the splitting pattern be more complex such as doublet of doublet or doublet of triplets, etc.? We are not supposed to say multiplet, even though the image is blurry we cannot say multiplet. Could you please go over all the splitting patterns for the different hydrogens. Please also indicate which peaks are relevant in the ir, using the table below for the ir spectra that was given earlier.
For Hydrogen D, could the splitting pattern be more complex such as doublet of doublet or doublet of triplets, etc.? We are not supposed to say multiplet, even though the image is blurry we cannot say multiplet. Could you please go over all the splitting patterns for the different hydrogens. Please also indicate which peaks are relevant in the ir, using the table below for the ir spectra that was given earlier.
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter1: Introduction To Chemistry
Section: Chapter Questions
Problem 1.83PAE: 1.83 What type of transition is represented in the following molecular-scale illustration?
Related questions
Question
For Hydrogen D, could the splitting pattern be more complex such as doublet of doublet or doublet of triplets, etc.? We are not supposed to say multiplet, even though the image is blurry we cannot say multiplet.
Could you please go over all the splitting patterns for the different hydrogens. Please also indicate which peaks are relevant in the ir, using the table below for the ir spectra that was given earlier.
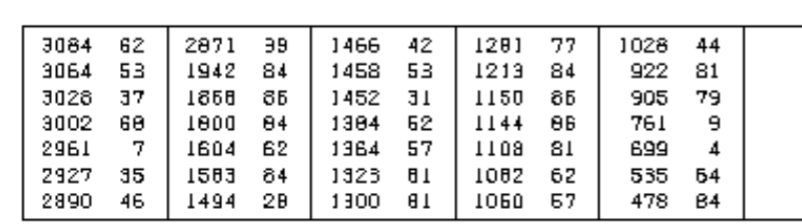
Transcribed Image Text:3084 62
3064 53
3028 37
3002 68
2961
7
2927 35
2890 46
2871 39
1942 84
166666
1800 84
1604 62
1583 84
1494 28
1466 42
1458 53
1452 31
1384 62
1364 57
1923 81
1300 81
1281 77
1213
84
1150
65
1144 86
1108 81
1082 62
1060
63
44
922 81
1028
905
79
761 9
699
4
535
478
54
B4
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 6 images

Follow-up Questions
Read through expert solutions to related follow-up questions below.
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning