X bMy Questions | bartleby 101 Chem 101 X X app.101edu.co Submit Question 17 of 43 Which one of the following compounds in aqueous solution would be best described by the picture below? Hint: look at the ratio of red to blue species in solution A) NH4NO B) LIOH C) CH3OH D) CaBr2 ++ 7:08 PM е OType here to search 11/7/2019 +
X bMy Questions | bartleby 101 Chem 101 X X app.101edu.co Submit Question 17 of 43 Which one of the following compounds in aqueous solution would be best described by the picture below? Hint: look at the ratio of red to blue species in solution A) NH4NO B) LIOH C) CH3OH D) CaBr2 ++ 7:08 PM е OType here to search 11/7/2019 +
Chemistry by OpenStax (2015-05-04)
1st Edition
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Chapter15: Equilibria Of Other Reaction Classes
Section: Chapter Questions
Problem 11E: The Handbook of Chemistry and Physics (http://openstaxcollege.org/l/16Handbook) gives solubilities...
Related questions
Question
Transcribed Image Text:X
bMy Questions | bartleby
101 Chem 101
X
X
app.101edu.co
Submit
Question 17 of 43
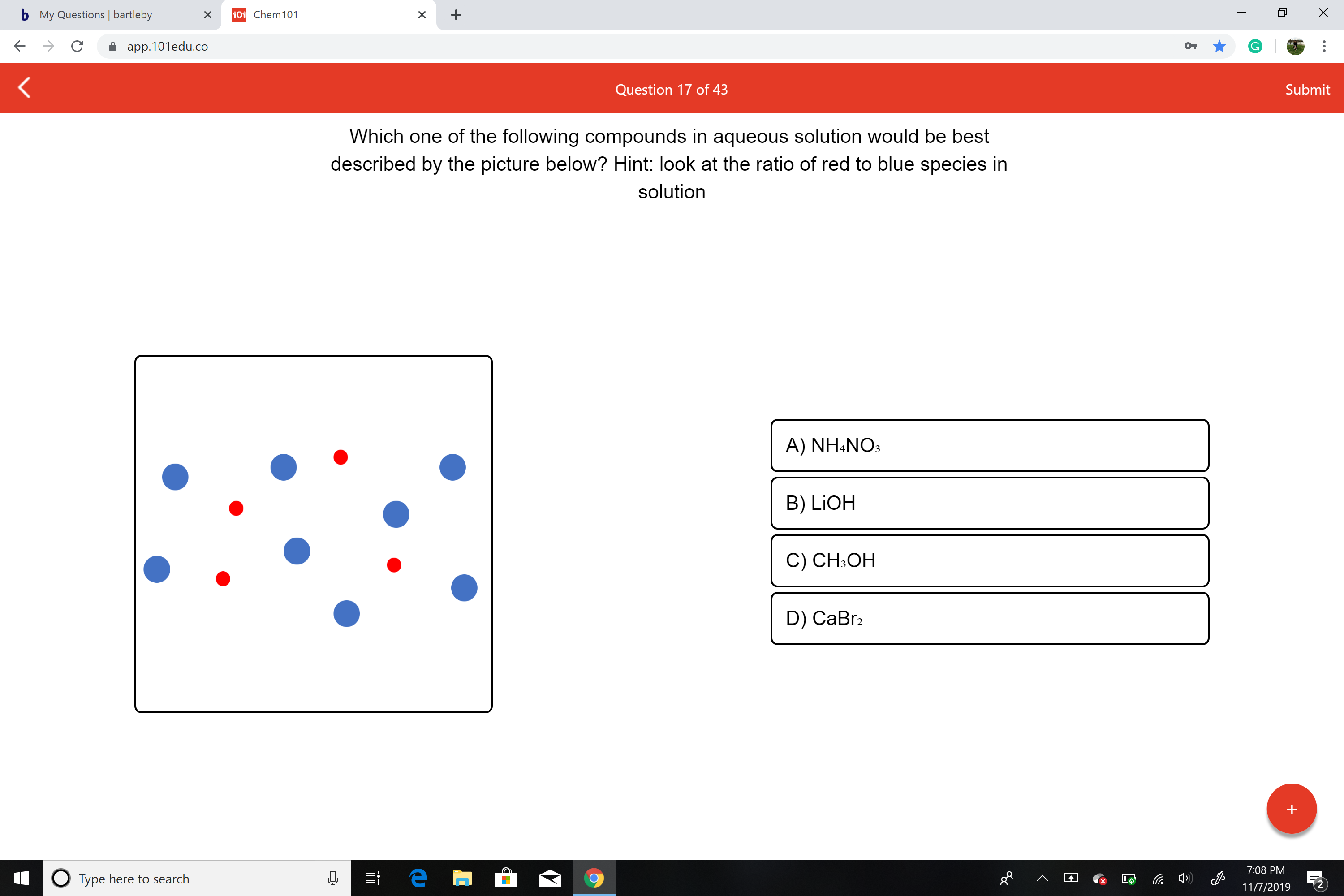
Which one of the following compounds in aqueous solution would be best
described by the picture below? Hint: look at the ratio of red to blue species in
solution
A) NH4NO
B) LIOH
C) CH3OH
D) CaBr2
++
7:08 PM
е
OType here to search
11/7/2019
+
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax