You have 0.5 ml of a stock solution of 1 mg/ml BSA. You need at least 200 µl of the solutions listed below. How do you make them up? 25 ug /ml 50 ug /ml 75 µg /ml 100 ug /ml 150 ug/ml 200 ug/ml
Q: You are to reconstitute 1 g of powder with a volume of 2.8 mL to make a final solution of 50 mg/mL.…
A: The solution is as follows:
Q: A pharmacist receives an order for 60 mL o an oral solution containing memantine hydrochloride…
A: Required = 60 mL of an oral solution containing memantine hydrochloride (NAMENDA) with concentration…
Q: How many grams of drug does a 100mL of 3 % solution contain? Group of answer choices 30 grams 0.03…
A: The mass of solute in gram present in 100 mL of solution is known as the mass percent concentration…
Q: A student wants 51.0 mL of a 1.50% saltwater solution. They only have a 5.00% stock solution. How…
A: The student require 51.0 ml of solution with concentration 1.5%. The concentration of stock is 5%…
Q: What volume (in mL) of 0.0887 M MgF2 solution is needed to make 275.0 mL of 0.0224 M MgF2 solution?
A: We are known with Molarity equation as- M1V1 = M2V2 here, M1 will be 0.0887M and V1 has to…
Q: Theresa Morgan, a chemist, has a 25% hydrochloric acid solution and a 70% hydrochloric acid…
A: The volume of each of the given acid solutions required to prepare the given solution can be…
Q: 170.0 mL of 0.12 M C2 H5NH2 with 285.0 mL of 0.20 M C2 H5 NH3 Cl
A: The pH of solution can be finding out by using Henderson Hasselbalch equation as: The given data…
Q: How many mL of 3.50 M H2SO4 solution is needed to prepare 250 mL of 1.25 M H2SO4 solution
A: Given data: Initial molarity of H2SO4 (M1) is 3.50 M Final volume of H2SO4 (V2) is 250 mL. Final…
Q: In the graph below, at what point does the equivalence point occur? 100 150 200 volume NaOH added…
A: Equivalence point It is a point at which chemically equivalent amount of reactants are mixed. We…
Q: 2. If the patient is to be given 120 mcg of levothyroxine via IV infusion and the vial contains 0.4…
A: There are various units of mass used for any system- 1- microgram = mcg 2- milligram = mg 3- gram =…
Q: Calculate the ppm of 40.5 mg calcium in 3.50 grams of pill. O 1.16 x10“ ppm 86.4 ppm O 8.64 x107 ppm…
A: Given : Mass of calcium i.e solute = 40.5 mg And mass of pill i.e solvent = 3.50 g
Q: To make a calibration curve you would first make stock solution Determine the mass of copper (ll)…
A:
Q: Calculate the volume in milliliters of 8.3 % (v/v)% ethanol solution that contains 33.2 mL of…
A:
Q: How many grams of MgCl2 in 30.00 mL of a 25.5 ppt solution ? (30.00 mL)(- %3D mL а. 25.5 i. 18.0 b.…
A: All the necessary calculations have been done in the following step.
Q: 7. Graduated cylinders can be used if an experiment calls for specific volumes but without the level…
A: All graduated glassware can be read with one uncertain digit, i.e. the uncertainty if +/- 0.1 is the…
Q: H.W: 1- A solution of KCI (its volume =500 mL) contains 7.45 ppm KCl. Calculate its folarity and…
A: Since you have posted multiple questions, we will solve only first question for you. If you want any…
Q: A dilution of benzene in methanol is made by placing 1.5 mL benzene (density of benzene is 0.879…
A: We know that, density is defined as the mass per unit volume. i.e. density =…
Q: If you start with 4.00M HCl how can you make up 200 ml of 0.200M HCl? a) 40ml 0f HCl and 40 ml water…
A:
Q: What is the pHpH when 5.3 gg of sodium acetate, NaC2H3O2NaC2H3O2, is dissolved in 150.0 mLmL of…
A: pH is used to determine the concentration of hydronium ion.
Q: A student wants 35.0 mL of a 2.19% sugar water solution. They only have a 17.0% stock solution. How…
A: Let 'y' milliliters of the 17.0% stock solution is used to make the 2.19% sugar water solution. 17%…
Q: what is the weight in grams of 300 ml of alcohol with a specific gravity of 0.80
A: Specific density may be defined as the ratio of density of substance with the density of…
Q: Today the only form of liquid acetaminophen on the market has a concentration of 160 mg…
A: Given: Mass of acetaminophen in 5.0 mL = 160 mg. And mass of acetaminophen needs to be given = 40…
Q: ppm mean that 100 100 kg per liter. 100 g per liter. 100 mg per liter. O
A: ppm refers to parts per million concentration.
Q: The theoretical weight of 10 tablets is 9.35g. Each tablet contains 425mg of the active ingredient.…
A:
Q: How many grams of solution will contain 30.5 g of NaCl if the solution has a concentration of 8.3 %…
A:
Q: The volume dispensed from the buret amounts to 32 33 32.25 ml O 33.75 mL O 33.70 ml O 32.20 mL
A: Given,
Q: • Stock solution: 1000 mg/L Target sample concentrations: 50-400 mg/L • Create a 5-point standard…
A: Given that, The concentration of the stock solution is C1 = 1000 mg/L. The volume of the final…
Q: If a prescription order requires 25 g of concentrated HCl (density, 1.18 g/mL), what volume should…
A: Density :- It is the ratio of mass of a substance to volume of substance Density (d) = mass (m) /…
Q: 10. Concentrated nitric acid, HNO3(aq), has a concentration of 15.7 M. What volume of concentrated…
A:
Q: 2. An 88-pound patient will be receiving aztreonam 30 mg/kg (MAX 1 gram) in 100 mL D10W. MWaztreonam…
A:
Q: How many mL of a 5.00 ppb solution are needed to have 1.00 g ? The concentration will always be the…
A: Ppb or part per billion is one of the ways to express the concentration of solution
Q: A mother liquor of a NaCl solution having a concentration of 5.00 M is taken two milliliters (2.00…
A: A 2.0 mL aliquot of 5.0 M NaC solution is taken out and diluted to a volume of 10.00 mL. We are…
Q: The following five beakers, each containing a solution of sodium chloride (NaCl, also known as table…
A: Molarity = number of moles of solute/volume of solution (in L) Number of moles = mass/molar mass…
Q: In lab, you will be given a stock solution that has a glucose concentration of 60 mg/dL. You need to…
A:
Q: concentrated form that is 37.0 % HCl by mass and has a density of 1.20 g/mL. Describe exactly how to…
A: We have to find how much concentrated solution must be taken such that stock solution has 0.5M for…
Q: mL Alcohol 85% ad 240 mL Sig. For the scalp. How milliliters each of 95% v/v alcohol and water…
A: The questions asks the volume of alcohol and water to be mixed in order to prepare 240 ml of 85%…
Q: Vodka is typically 80 proof, or 40% ethanol. The density of ethanol is 0.789 g/ml, and the standard…
A: Volume of ethanol = 44.36 mL Density of ethanol = 0.789g/mL A standard shot contains 40% ethanol So,…
Q: If you purchase 1 bottle of 120 tablets of 650. mg sodium bicarbonate in the pharmacy aisle (the…
A:
Q: 500 ml of 2000 ppm of sodium chloride is equal to 1g False True
A: ppm means parts per million. ppm is also expressed in (mg/L). In the case of solute (solid)…
Q: Consider the following prescription: Promethazine HCl 62.5 mg NaCl Nacl sb 50 mL SWFI ad Make…
A: Given, Promethazine HCl = 62.5 mg NaCl = qs SWFI ad = 50 mL Make isotonic solution E-value of…
Q: The following five beakers, each containing a solution of sodium chloride (NaCI, also known as table…
A: Answer:- This question is answered by using the simple concept of calculation of molarity of each…
Q: he following five beakers, each containing a solution of sodium chloride (NaClNaCl, also known as…
A: The concentration of NaCl in beaker 3 and 4 are; 1.0 L=1000.0 mLMNaCl3=mNaCl3MMNaCl×V3=25.5 g58.44…
Q: The protocol for a given lab experiment specifies that you should prepare a diluted solution by…
A:
Q: If an epinephrine injection contains 120 mg in each 15 ml to be administered as 10 mg, how many…
A: The amount of a compound is calculated based on the unitary method. In this method, the amount of a…
Q: You are to reconstitute 3 g of powder with 11.8 mL of diluent to make a final solution of 200 mg/mL.…
A: The solution is as follows:
Q: Acetic acid is commonly sold commercially in a 17 M concentration. What volume (mL) of the…
A: Dilution The dilution process is the process to change (decrease) the concentration of solution in…
Q: hat mass of Na2CO3 is present in 0.500L of a 0.417M solution? Group of answer choices 44.2 grams…
A:
Q: How many milliliters of 2.0 M sulfuric acid are needed to make 400.0 mL of 1.00 M H 2SO 4? 0.00500…
A: Given M1 = 2 M V1 = ? M2 = 1 M V2 = 400 ml
Q: You are asked to prepare a 10% solution using 10 g of a powder that occupies 14 mL. How much diluent…
A: Answer :- (b) 86 mL To prepare 10% solution using 10 g powder that occupies 14 mL we add 86 mL…
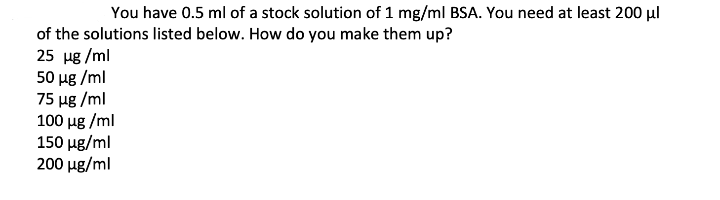
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- A pharmacist receives an order for 60 mL o an oral solution containing memantine hydrochloride (NAMENDA) 1.5 mg/mL. She has on hand a 360-mL bottle of oral solution containing memantine hydrochloride, 10 mg/5 mL, and a diluent of sorbitol solution. How many milliliters each of the available oral solution and sorbitol solution may be used to ill the order? Answer: 45 mL memantine oral solution 15 mL sorbitol solution Solution of the given answer is needed. Thank you so much and God bless. Happy new yearDENSITY SPECIFIC GRAVITY, SPECIFIC VOLUME. Show your solutions If a prescription order requires 25 g of concentrated HCl (density, 1.18 g/mL), what volume should the pharmacist measure? Which of the following will float in water? The density of water is 1 g/mL Object 1: m = 5 g, and v = 2 mL Object 2: m = 3 g, and v= 4mL Object 3: m = 3 g, and v = 1 mL Object 4: m = 4 g, and v = 3 mLA solution contains 500 mg of potassium acetate, 500 mg of potassium bicarbonate and 500 mg of potassium citrate per teaspoon. How many millimoles of potassium acetate, potassium bicarbonate and potassium citrate. are contained in a 25 ml vial? *Type in your answer in 2 decimal places. Do not forget the unit. Potassium acetate (MW-98 g/n) = Potassium bicarbonate (MW-100 g/n) = Potassium citrate (MW-306 g/n) =
- The protocol for a given lab experiment specifies that you should prepare a diluted solution by combining 1.0 mL of a stock solution (one that is more concentrated) and 9.0 mL of deionized water. Which piece(s) of laboratory equipment should you use to carry out the dilution? A. 10 mL volumetric pipet B. 1 mL volumetric pipet C. 10 mL graduated cylinder and a 2 mL serological pipet D. 10 mL seological pipet E. 10 mL graduated cylinder NOTE: the answer is not 10 mL volumetric pipet nor is it 1 mL volumetric pipet.1. If one fluidounce of a cough syrup contains 10 gr of sodium citrate, how many milligrams are contained in 5 mL? 2. A pharmacist received a prescription calling for 30 capsules, each to contain 1/200 gr of nitroglycerin. How many 0.4 mg of nitroglycerin tablets would supply the amount required? 3. If a child accidentally swallowed 2 fluidounces of Feosol elixir containing 2/3 gr of ferrous sulphate per 5 mL, how many milligrams of ferrous sulphate did the child ingest? PLEASE SHOW YOUR COMPLETE SOLUTION & THE CANCELLATION OF THE UNITS OF MEASUREMENT. Thank youa. If 55mg of CaC2O4 is placed in a 250mL pack of H2O nanopure at 25 celcius, how much (in mg) of CaC2O4 remains undesolved? b. If 100mg of CaC2O4 is placed in a 500mL pack of HCl at 25 celcius, how much (in mg) of CaC2O4 remains undesolved? FM: 128g/mol Pksp= 7.90
- How many milliliters of 2.5 M sulfuric acid are needed to make 100.0 mL of 0.40 M H 2SO 4? 16.0 mL 39.3 mL 0.0160 mL 0.0625 mL 62.5 mLStock strength of a drug is 50mg/2ml a client weight is administered 25mg/ kg what volume of drug should be given?How many microliters of 0.200 M Fe(NO3)3 do you need to make 200 microliters of a 2.50 mM solution?How many microliters of 0.200 M Fe(NO3)3 do you need to make 200 microliters of a 2.50 mM solution?
- Sources of error: Method 1 Equipment:About ten grams of NaHCo3. 10 ml distilled waterSmall beaker Electronic balanceGlass dome10 ml graduated cylinderGlass rod Safety instructions :Wear safety goggles Wear a smockBe deilcate with materials Method:Measure out 10 ml of distilled water using the graduated cylinder. Transfer the water to the small beaker.Weigh out the total amount of sodium chloride available for the experiment.Weigh out 0.5 g NaHCo3. Add NaHCo3 to beaker containing water. Stir solution with glass rod until dissolved.Repeat step 3 until it becomes more difficult to dissolve the NaHCo3. Note the number of times 0.5 g NaHCo3 is added.Continue adding one grain of NaHCo3 at a time until it is no longer possible to dissolve it.Once saturation is reached, NaHCo3 crystals form at the bottom of the beaker.Measure the mass of NaHCo3 not added to determine the total amount of NaHCo3 that has exceeded the solubility limit.6) 100.01 mg Cu(NO3)2 is diluted to 100.00 mL with distilled water. Calculate the ppm of the resulting solution and also calculate the uncertainty of the result. Report the result with the suitable number of significant figures (the uncertainty of 100.00 mL capacity volumetric flask is 0.07 mL, the uncertainty coming from the weighing is 0.05 mg).15.0 mL of KOH 100.0 mL of KOH 35.0 mL of KOH 125.0 mL of KOH

