You have been asked to compound a 50 ml suspension containing 1 mg/ml of hydrochlorothiazide. Your pharmacy Has tablets that contain 25 mg of hydrochlorothiazide per tablet. Your pharmacy also has a suspension that contains 5 mg/ml hydrochlorothiazide. Which formulation will you choose for the compounding and why?
You have been asked to compound a 50 ml suspension containing 1 mg/ml of hydrochlorothiazide. Your pharmacy Has tablets that contain 25 mg of hydrochlorothiazide per tablet. Your pharmacy also has a suspension that contains 5 mg/ml hydrochlorothiazide. Which formulation will you choose for the compounding and why?
Chapter17: Drugs And Medicine: Healing, Helping, And Hurting
Section: Chapter Questions
Problem 1SC
Related questions
Question
i need the answer quickly
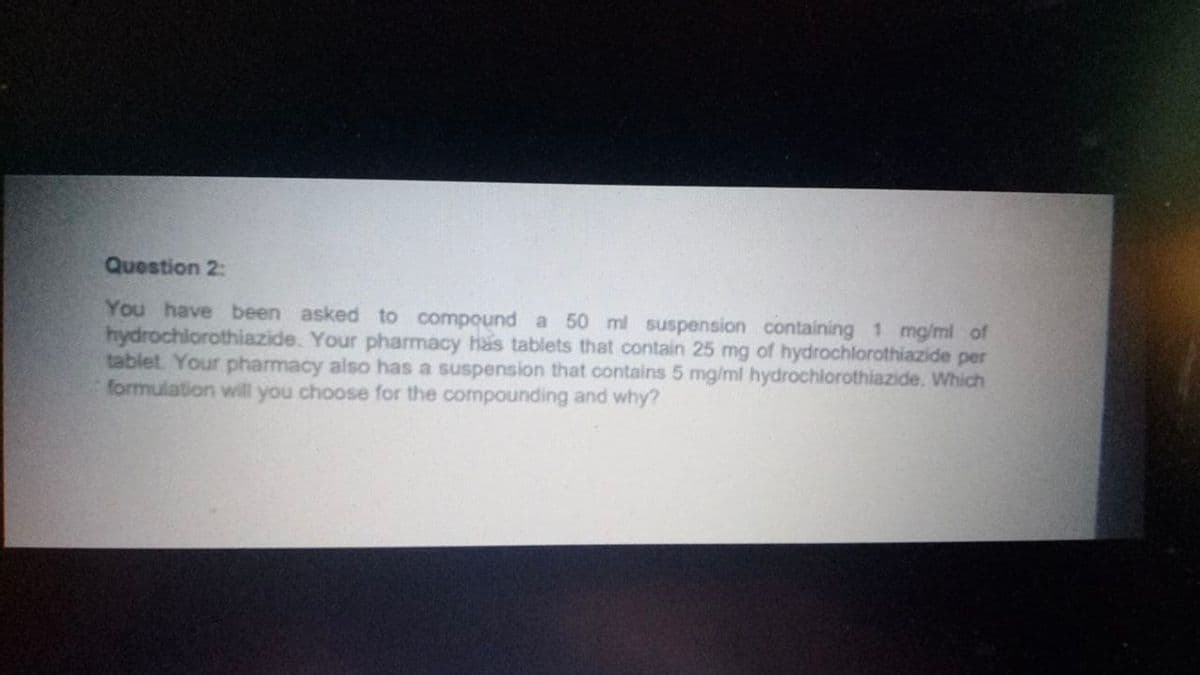
Transcribed Image Text:Question 2:
You have been asked to compound a 50 ml suspension containing 1 mg/ml of
hydrochlorothiazide. Your pharmacy has tablets that contain 25 mg of hydrochlorothiazide per
tablet. Your pharmacy also has a suspension that contains 5 mg/ml hydrochlorothiazide. Which
formulation will you choose for the compounding and why?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 3 images

Recommended textbooks for you

