Q: Which of the following molecules gives a condensation reaction with benzaldehyde? a)…
A: Which of the following molecules gives a condensation reaction with benzaldehyde has to be given,
Q: Which of the following CANNOT exhibit intramolecular H-bonding? A. none of the choices B.…
A: H- bonding will form when H is attached to more electronegative atom e.g. O , F etc and other…
Q: P. Which of the following compounds gives ketone by oxidation? (a) HsC-CH (CHs)-CH2-OH (Ь) OH (C) OH…
A: Secondary alcohol gives ketones upon oxidation .
Q: Which of the following reagents can oxidize an aldehyde to a carboxylic acid? O H2CrO4 MCPBA Ozone O…
A:
Q: Which of the following pairs of reagents are used to synthesize the ester below? ethanol + ethanoic…
A:
Q: Which of the following compounds is an ester? CH3CH2COOH. CH3CH2CHO CH3CH2CH20H CH3CH2CH2OCH3…
A: We are given molecules with different functional groups, and we have to identify which one is an…
Q: Which of the following compounds decarboxylates when heated?
A: SOLUTION: Step 1: The carboxylic group can be easily removed when it is bonded on the α- position of…
Q: exees Ethyl ester (P). CH MgBr The product (P) will be (a) (b) H,Ca (e) OH H,Ca C,H, (d) H,C OH
A:
Q: Predict the products formed when cyclohexanone reacts with the following reagents.) hydroxylamine…
A: Given reaction,
Q: Predict the products formed when cyclohexanone reacts with the following reagents.ethylene glycol…
A: The protecting group that is used for the carbonyl compound will be the cyclic acetals and the…
Q: What ester is formed when each carboxylic acid is treated with ethanol (CH 3CH 2OH) in the presence…
A: Esters are formed by the reaction of carboxylic acid with alcohol in the presence of catalytic…
Q: Which of the following compounds will form a yellow solid when dissolved in a basic, aqueous…
A: Iodoform test: The Ketones and aldehydes which have a CH3CO group can react with iodine in a basic…
Q: Which of the following will readily undergo decarboxylation upon heating?
A:
Q: 4. Predict the esters formed in the following reactions. A catalytic amount of acid is added in each…
A: In this reaction Ester is formed. One molecule of water eliminate.Hydrogen from alcohol and OH from…
Q: Compounds that contain both a hydroxyl group (OH) and a carboxyl group (COOH) can undergo an…
A: Esterification reaction is the reaction between carboxylic acid and alcohol with presence of acid to…
Q: What products are formed when each ester is hydrolyzed with water and H 2SO 4?
A: Hydrolysis of esters in the presence of acid H2SO4leads to the formation of carboxylic acid and…
Q: The reaction of an aldehyde or a ketone with a hydroxylamine produces which of the following…
A: When aldehyde or, ketone is treated with hydroxylamine then corresponding oxime compound is formed.…
Q: What product is formed when diethylketone is treated with [Ag(NH3)]* ? (A Propanone and ethanoic…
A: there will be no any reaction. because ketone will will not react with tollens reagent.
Q: Which part of taxol is a ketone? D) E) C) о Он B) `NH Q G) F) ОН A)
A: In this question, we will select the one letter which is represent ketone group in taxol compound.…
Q: Show how each of the following esters could be prepared using a carboxylic acid as one of the…
A: In fischer esterification process esters are prepared from carboxylic acid and alkyl halide in…
Q: What is the product of methyl magnesium bromide, with the following ester excess CHMgBr then HO он…
A: In CH3MgBr, there is a CH3- nucleophile. Hence it will reduce the ester into alcohol as CH3MgBr is…
Q: 30. Which of the following reactions are ways to synthesize carboxylic acids? 1. Oxidation of an…
A: In the given question we have to choose the correct ways to synthesize carboxylic acid from the…
Q: Predict the products formed when cyclohexanone reacts with the following reagents. (k) sodium…
A:
Q: Which one of the following compounds would react with CH3CH2MgBr, followed by acidic hydrolysis, to…
A: This is multiple questions so I will solved first question for you. You can see the details…
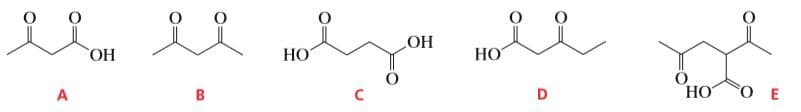
Q: Which of the following compounds will decarboxylate when heated?
A: Decarboxylation is a chemical reaction in which a carbonyl group is removed and carbon dioxide is…
Q: Which of the following cannot be oxidized by Tollen's reagent? O Formaldehyde O cyclohexanone O…
A: Aldehydes and ketones are the organic compounds which contains the -CHO and -CO- group. They contain…
Q: Which of the following transformations represents malonic ester synthesis ? I CH2(CO,Et)2 R. COOH II…
A: Given:-
Q: Predict the products formed when cyclohexanone reacts with the following reagents. ) Tollens reagent
A: Tollen’s test, also called Silver mirror test is used to distinguish between aldehydes and ketones.…
Q: What ester would be formed in this reaction? OH butanoic acid Z HO pentan-1-ol H+
A: In given reaction acid acts as catalyst. Catalyst increases speed of reaction it does not get…
Q: Predict the products formed when cyclohexanone reacts with the following reagents.phenylhydrazine…
A: The products formed when cyclohexanone reacts with the given reagent phenylhydrazine and weak acid…
Q: Ph cat. H,SO4 H2O heat Ill.
A: Hydrolysis of acetals is a reverse reaction of acetal formation. In the presence of aqueous acid…
Q: H,SO4 (СH), СНCH, OC-ОCH,СH, H,O
A:
Q: Draw the product formed when the following a.ß-unsaturated ketone is treated with [1] (CH,=CH),CuLi;…
A:
Q: Draw the chemicals that form when the following chemicals are oxidized: a) 3-Chloropropanal b)…
A: Oxidation of alcohols occur to form aldehydes, ketones or carboxylic acids depending upon the nature…
Q: Which of these is the most viable route for the preparation or synthesis of a carboxylic acid? O…
A: Which of these is the most viable route for the preparation or synthesis of a carboxylic acid? O…
Q: Complete the following reaction pathways by filling in any missing reagents, intermediate compounds,…
A:
Q: Which of the following molecules gives a condensation reaction with benzaldehyde? A.…
A: Which of the following molecules gives a condensation reaction with benzaldehyde?
Q: 6. Which of the following molecules can be oxidized to a carboxylic acid? НО HO- ОН ОН b. ОН d. a.…
A: Since for oxidising the molecules to carboxylic acid we need 2 H at least on the C with OH so that…
Q: Which of the following carbonyl compounds is the least reactive? O A. Acid halide O B. anhydride O…
A:
Q: Which of the following functional groups will be formed when the hemi-acetal is reacted with…
A:
Q: What product is formed when Ethyl phenyl Ketone is treated with NABH4?
A: NaBH4 is a reducing agent for the reduction of aldehydes and ketones. Aldehydes are reduced to…
Q: 5.41 Draw the structures of the aldehydes that might be oxi- dized to yield the following carboxylic…
A:
Q: How you can distinguish between each of the following pairs, ethanal and propanal? H3C H. H,CH,C H.…
A: Ethanal and propanal can be distinguish by Iodoform test. Iodoform test is used to identify the…
Q: Which of the following esters DOES have an enolizable hydrogen? OEt OEt OEt OEt (1) (2) (3) (4)
A: “Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: H, SO, CH,OC=0CH,CH(CH, )CH,CH, H,O
A: Acidic Hydrolysis of ester- when an ester is heated under reflux with ( H2SO4+ H2O) such as dilute…
Q: /hich of the following reactions will result in formation of a ketone? 1) 0=C=0 CA) MgBr 2) H3O* 1)…
A: Given are organic reactions
Q: What hydrolysis products are formed when the following compound is treated with aqueous acid?
A: The hydrolysis products are formed when the given compound is treated with aqueous acid has to be…
Q: 12. Which of the following compounds gives a positive Tollens' test? (a) OH (b) OH (c) OH (4) (e)…
A: We have to discuss which compound gives Tollen's test.
Q: LIAIH4 reacts with all of the carbonyl compounds to form alcohol products except: Amides Aldehydes…
A: Question is four compounds given Amides Aldhehydes Ester Acid chloride Which of compounds is not…
Q: Identify each of the following as a hemiacetal, acetal, or neither OH OH t OH OCH H. OH OH
A:
Which of the following compounds will decarboxylate when heated?
Step by step
Solved in 2 steps with 1 images


