. Consider the chromatogram shown below (used with permission from the Honors Thesis of Mark Bickley, TU Chemistry alumnus). The chromatography column used to separate the three analytes (A, B, and C) is 17.0 cm in length. The elution time for an unretained solute is 2.05 min. B. Time (min) a) Calculate the adjusted retention time for compound B. b) Calculate the retention factor for compound B.
. Consider the chromatogram shown below (used with permission from the Honors Thesis of Mark Bickley, TU Chemistry alumnus). The chromatography column used to separate the three analytes (A, B, and C) is 17.0 cm in length. The elution time for an unretained solute is 2.05 min. B. Time (min) a) Calculate the adjusted retention time for compound B. b) Calculate the retention factor for compound B.
Chapter31: Introduction To Analytical Separations
Section: Chapter Questions
Problem 31.32QAP
Related questions
Question
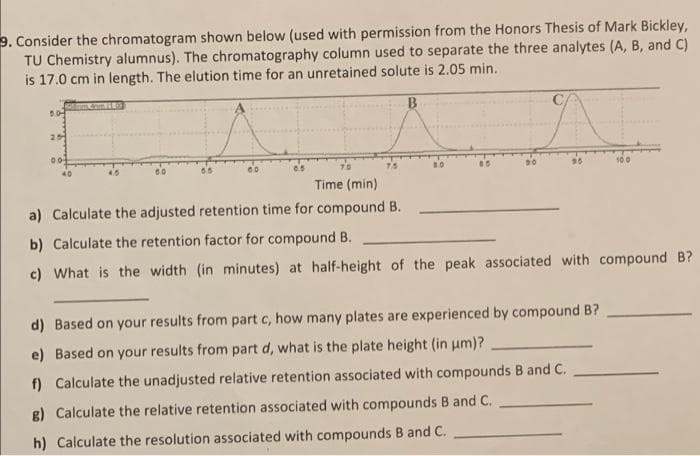
Transcribed Image Text:9. Consider the chromatogram shown below (used with permission from the Honors Thesis of Mark Bickley,
TU Chemistry alumnus). The chromatography column used to separate the three analytes (A, B, and C)
is 17.0 cm in length. The elution time for an unretained solute is 2.05 min.
25
T0
7.5
100
Time (min)
a) Calculate the adjusted retention time for compound B.
b) Calculate the retention factor for compound B.
c) What is the width (in minutes) at half-height of the peak associated with compound B?
d) Based on your results from part c, how many plates are experienced by compound B?
e) Based on your results from part d, what is the plate height (in um)?
f) Calculate the unadjusted relative retention associated with compounds B and C.
g) Calculate the relative retention associated with compounds B and C.
h) Calculate the resolution associated with compounds B and C.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT