Q: 26. Select the correct statement a) Ionization energy and metallic character have the same trend. b)...
A:
Q: Answer the following questions about activating and deactivating groups. Give specific examples. 1....
A: As per the company guidelines, we are authorised to solve only one question at a time. Please post r...
Q: many electrons in an atom can have the quantum numbers n = 3, 1 = 2? a) b) 5 9. How 10 d) 18
A: For a particular 'n' value, total number of azimuthal quantum number , l = n-1 . For every 'l' valu...
Q: Without doing any calculations, match the following thermodynamic properties with their appropriate ...
A: for the given endothermic reaction, N2(g) + 2O2(g) →2NO2(g)
Q: Lewis structure name of highlighted group H H н— о С — С — н H H H : 0— С — н | H :0-H H :0: H -C. N...
A:
Q: c) Calculate the theoretical density of the Zno. Hint: calculate the volume of unit cell from consid...
A:
Q: Calculate the formula mass of the molecule from its structure. IC IN 10 formula mass: amu A Rotate X...
A: The formula mass of a substance is the sum of the average atomic masses of each atom represented in ...
Q: A 25 mL pipet delivers 24.7761 g H2O which is weighed against brass weights at 25°C. Calculate the v...
A: The calibration of an apparatus is to configure an apparatus by comparing a reading on one apparatus...
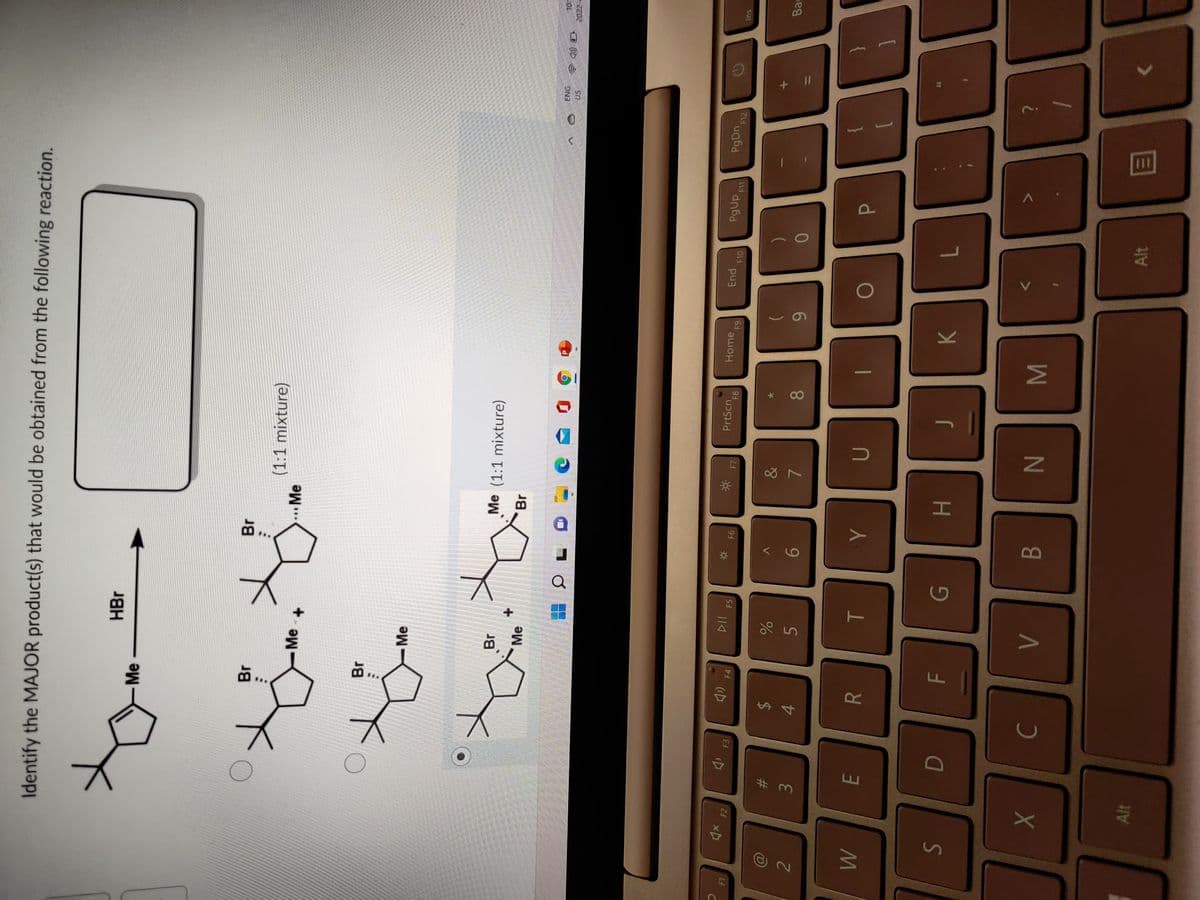
Q: 1. Identify (draw the structure) of the missing reactant, reagent, or major product required to comp...
A: Answer of the question given below,
Q: "Synthesis gas is a mixture of carbon monoxide and water vapor. At high temperature synthesis gas wi...
A:
Q: Titration of 25.0 mL of 0.200-M HA with 0.2 M LIOH, Calculate the initial pH, the pH after addition ...
A: Answer : initial pH: [H+] = sqrt (Ka * concentration ) = sqrt (1.78*10^(-5)*0.2) =1.89 *10^(-3) pH ...
Q: 5. Which of the following statements is (are) true? Energy is directly proportional to wavelength. T...
A: Option b is correct that is III, IV, V III. The energy of electromagnetic radiation increases as its...
Q: Question 3 Determine the ΔH for the following reaction, N2(g) + 2O2(g) → 2NO2(g) ...
A: Correct answer = 180.6 KJ Solution: For solving this type of problem, we simply used Hess's law for ...
Q: Lewis structure name of highlighted group H -C= Н — С — Н н —N— Н H Н —с — н 0=C-N - H H H H H H -N ...
A:
Q: emulsion in the initial (jar) test?
A: In this question we have to explain that which of the emulsifiers produced the most stable emulsion ...
Q: Estimate the value of the absolute zero temperature.
A: Absolute zero temperature is the lowest temperature in the thermodynamic system and at this temperat...
Q: Classify each of these chemical compounds: type of compound (check all that apply) compound molecula...
A:
Q: An electrical engineer is analyzing an electroplating run and wants to calculate the charge that has...
A:
Q: 4. (i) Identify the different enthalpy changes and construct the Born-Haber cycle for the formation ...
A:
Q: Fill in the squares in the following chemical equations: NaOH/H,O ELOH
A: Concept: 1) aldol reaction 2) Robinson annulation ; Michael reaction followed by aldol reaction is ...
Q: Which of the following is an incorrect designation for an atomic orbital? a) 1s b) 3d c) 2p d) 3f 8.
A:
Q: A[CH,0] For the reaction below, what is with respect to At 2 C2H4(g) + 2 O3(g) – 4CH20(g) + Ö2(g) 4 ...
A: CH2O is a product and O3 is a reactant. The rate of consumption of a reactant is related to the rate...
Q: Because Hydrogen is on the metal side of the periodic table, it does NOT react with itself to be dia...
A:
Q: Q28 Q30
A: We have to calculate the molar solubility of CaF2 in 0.25M solution of NaF. Ksp of CaF2 = 4×10-11 We...
Q: he function of glycerol in embalming fluids is A. to act as a humectant B. to dehydrate ...
A: Embalming fluids are substances used to slow down the decomposition rate. Embalming prevents the de...
Q: The detonation of nitroglycerin proceeds as follows: 4 C3H5N3O9(l) → 12 CO2(g) + 6 N2(g) + O2(g) + 1...
A: Given :- Given mass of Nitroglycerin =4.5 gram Volume of nitroglycerin = 2 ml Density of nitroglycer...
Q: 14. How many unpaired electrons are present in the orbital diagram of Se? a) b) c) 0. d) 3 12 O
A:
Q: A scientist measures the standard enthalpy change for the following reaction to be -52.7 k) : C,H4(9...
A:
Q: Provide the proper IUPAC name for the alka CI HO.
A: The numbering will be start from -OH group and then double bond and in last chlorine atom. For -OH ...
Q: Draw the following using line structure only: 1,2,3-trimethylcyclopropane b.1,3-dichlorocyclopen...
A: In the question it is given to draw the line structure of the following compounds according to IUPAC...
Q: Draw the simplest set of curved arrows that shows how the structure on the left could be turned into...
A: Lewis structure represent movement of electrons in an atom or molecule. Lewis structure are also cal...
Q: Find the energy per molecule of CO2 released in eV and compare this to the energy per CO2 molecule r...
A:
Q: Arrange the following from lowest to highest entropy: SiO2(s), SiBr4(4), SİCL (g), SiC(s). Assume 1 ...
A:
Q: Given the standard enthalpy changes for the following two reactions: (1) 2Ni(s) + 02(g)→2N¡O(s) AH° ...
A: Given, 1) 2Ni(s) + O2(g) ➝ 2NiO(s) ∆H° = -479.4 kJ 2) 2Hg(l) + O2(g) ➝ 2HgO(s) ∆H° = -181.6 kJ 3...
Q: What is the molarity of a solution that contains 75.0 g of KNO3dissolved in 0.350 L of solut...
A: Given : mass of KNO3 = 75.0 g volume of solution = 0.350 L To calculate :- molarity of...
Q: 2022 HE 231SP 2022 1. Give an açceptable (common or systematic name to the following compounds: (a) ...
A:
Q: 4 rate=k[A]a[B]b Assuming volumes add, can I use volume % for concentration in this equation instead...
A: Generally we are using molarity terms in rate law equation. Unit of molarity is mol/L which is also...
Q: A scientist measures the standard enthalpy change for the following reaction to be -1057.0 kJ : 2H2S...
A:
Q: Q3 Write structures showing the specified configurations for each of the following compounds. Make y...
A: 3. A chemical structure determination includes a chemist specifying one molecular geometry, when f...
Q: (References) Without doing any calculations, match the following thermodynamic properties with their...
A: Given:- The reaction is:- 2N2(g) + O2(g) ----->2N2O(g) {endothermic} We have to find: •∆Hrxn •∆S...
Q: A certain number of moles of gas is contained in a 29.2 liter flask at 15.6°C. When the temperature ...
A: According to Charles law, when number of moles (composition) and pressure is kept constant then volu...
Q: Indicate the correct set of reactions from the choice below that would accomp following synthesis. H...
A:
Q: Gaseous ethane (CH,CH,) reacts with gaseous oxygen gas (0,) to produce gaseous carbon dioxide (CO,) ...
A: Ethane reacts with oxygen to form carbon dioxide and water. The equation for the balanced chemical r...
Q: Aqueous hydrochloric acid (HCI) reacts with solid sodium hydroxide (NaOH) to produce aqueous sodium ...
A:
Q: 20. Select the correct electron configuration of Co3+ [Ar]4s² 3d [Ar]4s? 3d* [Ar] 3d [Kr] 4s2 3d a) ...
A:
Q: Section A the solid is warming but also not melting. Section B is Melting. Section C the Liquid is...
A: Answer : From temperature, -5oC to 5 oC, solid states gets heated ( part a) from temperature 5 oC ( ...
Q: Consider the reaction: 2NH3(g) + 3N20(g)4N2(g) + 3H20(g) Using standard thermodynamic data at 298K, ...
A: 2NH3(g) + 3N2O(g) ----> 4N2(g) +3H2O(g) Number of moles of NH3= 1.86 moles
Q: Draw the product when the hydrogen atom bonded on the deuterated carbon undergoes a thermal sigmatro...
A: The given molecule may undergo sigmatropic hydrogen shift in thermal as well as photochemical condit...
Q: a student mixes the following solutiond. what is the ph of the solution? .2 L of .1 M Ca(OH)2 .3 L...
A: pH of a solution is given by the following formula. pH=-log[H+] H+ and OH- ions neutralise each othe...
Q: 24. Which of the following arrangements is in order of increasing size? a) Ca2+ K* > Cl-> S2- b) S2...
A: Given, Ca2+ , Cl- , K+ , S2- the size of ions increasing order is :
Step by step
Solved in 2 steps with 2 images

- cmplete the reacting by adding necessrt reagents. write in thapce provided. make sure to write regents in chemical formula . n separate regents with comma & space (ex. h2so6, h202)Pls ignore the highlighted part __________ is reduced in thefollowing reaction: Cr2O72- + 6S2O32-+ 14H+→ 2Cr3+ + 3S4O62- + 7H2OI’m currently trying to write a lab report for the synthesis of dimolybdenum tetraacetate [Mo2(O2CCH3)4] from the reaction of molybdenum hexacarbonyl, Mo(CO)6 in glacial acetic acid and acetic anhydride under a nitrogen atmosphere, involving the difficult formation of a quadruple bond and requires high heat and long reaction time (approximately 20 hours). But these are the question I’m stumped on: 1. Why need the reaction be done under nitrogen? We also added dichlorobenzene and hexanes during the reaction. 2. Explain the purpose of dichlorobenzene and hexanes. 3. Why does the reaction take 20 hrs?
- Please complete reactions in clear handwritten of all subpartsThe replacement of CH3OH to dimethyl sulfoxide, DMSO (CH3)2S=O) as a solvent in the substitution reaction below results in what?Given: Mass spec for 4-acetylbiphenyl. Knowing that 196 m/z is the major product, how would you assign 181 m/z and 152 m/z? (i've also attached a picture of the reaction scheme)
- I am working on a practice assignment for my organic II course and am having difficulty with a question that asks to identify the reaction sequence used to synthesize isopropylcyclopentane. I would really appreciate the help!Solution:- 3. Determine the amount, in of a (2.20 M s olution of dichloromethane needed to completely react with 15.72g cyclohexene to give 1,2-dibromocuclohexanw. Assume 12% excess is needed in order to react completely. a . How much 1,2 -dibromocyclohexane would theoretically be produced ? c. How many ML of the 2.20 M Br2 solution are required?Really hoping for solutions since I’m having a hard time with this. Pls. skip if unsure or not willing to answer the subitems (these are all connected for one item). Thanks in advanced. A kinetics experiment was performed for the formation of a novel compound, UNO, from its reactants, Chemicals G, R, A, D, and E. The first six runs (Runs 1-6) were all performed at 301 K, while the next two runs (Runs 7-8) at varying temperatures. These are tabulated in the table shown below. Table 1. Reaction rate for the reaction of the different chemicals at a given concentration at 301 K. Run [G], M [R], M [A], M [D], M [E], M Rate, M/s 1 0.80 0.01 0.20 0.50 2.00 0.180 2 0.80 0.02 0.20 0.50 0.02 3.54 x 10-3 3 0.80 0.01 0.20 0.50 0.02 1.80 x 10-3 4 0.80 0.01 0.60 0.50 0.02 1.80 x 10-3 5 1.20 0.01 0.20 0.50 0.02 4.02 x 10-3 6 0.80 0.01 0.20 0.15 0.02 1.95 x 10-2 a. Calculate the rate order of each chemical and overall rate order of the…
- can i get help drawing out actual structures including the nucleophilic addition of Cy2NH to parafomaldehyde and its hemiaminal intermidiate and the condensation step when it is displaced by terminal alkyne forming allene, also what is dioxane getting rid of as the solvent, thanksCan you pls assist me with the mechanism of Benzyl alcohol being oxidised to Benzyl aldehyde and to Benzoic acid using KMnO4 as an oxidant. Showing arrows and everything.Describe every aspect of the procedure clearly and explicitly. Include temperatures, appearance of the reaction (include pictures!). How is the reaction monitored? Is the order of addition important? 4,4'-DIBROMOBIPHENYL [Biphenyl, 4,4'-dibromo-] Submitted by Robert E. Buckles and Norris G. Wheeler1. Checked by R. S. Schreiber, Wm. Bradley Reid, Jr., and Robert W. Jackson. 1. Procedure In a 15-cm. evaporating dish is placed 15.4 g. (0.10 mole) of finely powdered biphenyl (Note 1). The dish is set on a porcelain rack in a 30-cm. desiccator with a 10-cm. evaporating dish under the rack containing 39 g. (12 ml., 0.24 mole) of bromine. The desiccator is closed, but a very small opening is provided for the escape of hydrogen bromide (Note 2). The biphenyl is left in contact with the bromine vapor for 8 hours (or overnight). The orange solid is then removed from the desiccator and allowed to stand in the air under a hood for at least 4 hours (Note 3). At this point, the product weighs…





