Q: A mixture of 4.5 moles of aluminum and 3.0 moles of oxygen is allowed to react: 4 Al(s) + 3 O₂(g) →…
A:
Q: Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product…
A: The Bronsted-Lowry acid-base theory states that the acid is a substance that gives H+ ions and the…
Q: Consider the reaction below. 2.7 mol of A and 6.7 mol of B are added to a 4 L container. At…
A: moles of A = 2.7 molmoles of B = 6.7 molvolume of container = 4 L[A} at equilibrium = 0.10 M[B] at…
Q: How many grams of NaOH would be in 25 mL of 0.100 M NaOH?
A:
Q: How many micrograms of Hg2C12 (Ksp = 1.3 x 10^-15) is required for the preparation of a 2.5 L…
A: Consider solubility equilibrium equation for Hg2Cl2 :Hg2Cl2(s) <=> 2Hg+(aq) + 2Cl- (aq)Let s…
Q: The vapor pressure of the liquid C4H10 is measured at different temperatures. The following vapor…
A: According to the clausius clapeyron equation,where P2 = vapour pressure at temperature 2 = 621.3 mm…
Q: Draw the products resulting from addition of a Grignard reagent to an aldehyde. Use a dash or wedge…
A: Grignard reagent (RMg-X, Organo magnesium halide) is a source of carbanion which acts as a base as…
Q: Other Questions: Please save these for after the lab!!! Come back to these after Titration #2. 1. On…
A: A redox reaction is defined as a reaction in which oxidation and reduction take place simultaneously
Q: Which correctly identifies the major product and its stereochemistry for this reaction? IIBr, 11₂0₂…
A: Given that, the reaction is:
Q: or Br Br ? Br Br
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: 3. The solubility of CSF2(s) in water is 2.1 x 104 M. Calculate its Ksp.
A: The question is based on the concept of solubility product principle.It states that when a weak…
Q: Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product…
A:
Q: Hi just wondering were the 2.303 came from
A:
Q: 1) 9-BBN, THE 2) NaOH, H20
A: Given is organic reaction. This is hydroboration oxidation reactionThe given reagent is 9-BBN…
Q: VSEPR predicts the electronic shape around an atom based on the number of electronic domains…
A: Prediction of electronic shape is done by VSPER looking into the electronic domain.The most stable…
Q: 2. Soaps (surfactants) are amphiphilic, meaning they have both polar and nonpolar domains. For…
A: Answer:Hydrophilic means water loving and hydrophobic means water repelling. It means if a specie is…
Q: What IR stretch would most readily distinguish carnauba wax from paraffin wax? 3rbt 910 Answer:
A: Infrared (IR) spectroscopy is a valuable tool for distinguishing between different compounds based…
Q: A В но с но OH PCC POC PBr
A: Since you have asked a question with multiple subparts, we will answer only first 3 subparts for…
Q: Nitric acid is produced commercially by the Ostwald process. In the first step ammonia is oxidized…
A: e
Q: Which has the lowest boiling point?
A: The temperature at which the vapor pressure of a liquid becomes equal to the atmospheric pressure is…
Q: spontan only at low temperatures; only at high temperatures; at all temperatures? Use the data table…
A: Answer:A chemical reaction can only be spontaneous when is less than zero.Also, we know that
Q: As an alternative to "metal oxides," some sunscreens utilize "organic" (carbon-based) ingredients…
A: Answer:During the bond formation two types of overlapping takes place:Head to head overlappingSide…
Q: Draw the two products of the reaction shown below. Assume only 1 equivalent of HBr is consumed in…
A:
Q: A student is collecting their Electrophilic Aromatic Substitution product, which has just been…
A: Recrystallization is a purification technique employed for crystalline solids. In this method, the…
Q: In the following net ionic equation, identify each species as either a Bronsted-Lowry acid or a…
A: According to Bronsted-Lowry Acid base theory Acid (HA) is species which donate proton and after…
Q: Br NaSCH 3 THF Draw SN2 product
A: Since you have asked multiple questions, only one question can be answered at a time. Please repost…
Q: C-H 413 c-c 348 C-N 293 C-0 358 C-F 485 -CI 328 276 Average Bond Enthalpies (kJ/mol) Single Bonds…
A: Enthalpy change of reaction may be given as ; ∆H = ΣBER - ΣBEPWe calculate enthalpy change for the…
Q: 1) NaNH2 2) Br
A: In this reaction terminal triple bonded carbon is SP^3 hybridized,so the attached hydrogen is acidic…
Q: See Figure 13-9. Which structure best matches the NMR shown? A B с D
A:
Q: 1 minus X(s)|X4+ (8.17 M) || Mn 04 (0.24 M)|Mn 02 (s) |O H¹ minus The reduction potential for X4+ is…
A: Answer:Here:n is equal to number of electrons transferred from anode to cathode in the balanced cell…
Q: QUESTION 46 Calculate the solubility of lead(II) iodide, Pbl₂, in 0.95 M Pb(NO3)2. (Ksp lead(II)…
A: The objective of this question is to calculate the solubility of lead(II) iodide, PbI2, in a 0.95 M…
Q: Adding table salt to water elevates its boiling temperature and speeds up the cooking process…
A: moles of salt = 0.291 molmass of solvent i.e. water = 2 kg as the density of water is 1 g/LKb =…
Q: ОН 1eq. tBuOOH, Ti(OiPr)4 (+)-DET
A: Given that, the reaction is:
Q: Which halide reacts most rapidly via an S,,1 mechanism? d Br B C D
A: Answer:In Sn1, S means substitution, n means nucleophilic and 1 means unimolecular, so, Sn1…
Q: Predict the major product of the following reaction, and then draw a curved arrow mechanism for its…
A: Alcohols gives dehydration reactions when they react with a strong acid like H2SO4, tosic acid…
Q: q15: If 1.0 mol of Mg is mixed with 2.0 mol of Br2, and 0.52 mol of MgBr2 is obtained, what is the…
A: If 1.0 mol of Mg is mixed with 2.0 mol of Br2, and 0.52 mol of MgBr2 is obtained, we have to…
Q: Determine the mechanism and product for the given reaction by adding atoms, bonds, nonbonding…
A:
Q: A sample of vinegar (20.0000 g) contains 1.0763 g of acetic acid. What it the mass% of acetic acid…
A: There are several terms to express the concentration of the solution. Mass percentage is one of the…
Q: Name the functional groups and circle any chiral centers in these molecules?
A: The carbon atom in organic molecules is attached to four groups which are completely different atoms…
Q: Calculate the pH and pOH of 306 mL of a phosphate buffer that is composed of 0.314 M HPO2 and 0.512…
A: = 0.314 M = 0.512 MKa = 4.50*10-13Volume of buffer = 306 ml
Q: Refer to the following phase diagram (not to scale!) for hydrogen : P atm 12.8 1.00 0.071- 13.9 14.0…
A: The hydrogen phase diagram shows the phase behavior with changes in temperature and pressure.
Q: 10. Provide the reagents necessary for the following transformation. Circle 1 answer. H₂C-CH₂-C CH…
A: The reactant is alkyne since it has one C-C triple bond. On the other hand, the product is an…
Q: H3C. CH3 H ОН
A:
Q: Starting Material OH Terpineol KMNO4, KOH H₂O A R-carvone ... O S-carvone ki OH Limonene
A: The objective is to determine the starting reactant that would lead to the given product in the…
Q: OH Н PB13
A: The reaction of alcohol (R-OH)with PBr3 is used for conversion of an alcohol to alkyl bromide. PBr3…
Q: The substance xenon has the following properties: Normal melting point: 161.3 K Normal boiling…
A: A question based on phase diagram. Based on the given data about phase diagram of Xe, the follow-up…
Q: Atom H N 6 2 2 Reactant side Product side 2 N₂ H4 + H₂O₂ → N₂ + H ₂ O On the reactant side there are…
A: According to the law of conservation of mass, we must balance each chemical equation.To balance it,…
Q: The reaction A-B2C is known to be zero order with a rate constant of 5.0 x 10 mol/L/s at 25°C. An…
A: The rate of a chemical reaction is a measure of how quickly reactants are converted into products.…
Q: 2 Describe all possible ¹H NMR peaks in the following compound: myrtenal CHO
A: Number of signals in 1H NMR: Tells us how many sets of protons with different electronic environment…
Q: Predict the major organic product of the reaction of 2-methyl-1-propene or 2-methylprop-1-ene and…
A: Alkene reacts with water (H2O) in presence of acid to produce alcohol. This reaction follows…
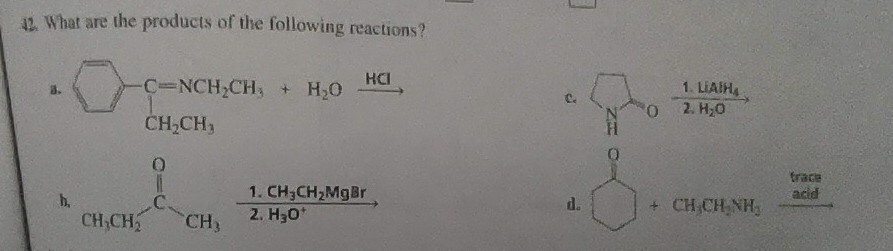
Step by step
Solved in 3 steps with 2 images

- 2) What is the main product for each of the following reactions? (d) Styrene + CH3CH2COCl AlCl3, then Br2, Fe -----> ? (e) Benzene + 1-chloro-2,2-dimethylpropane, AlCl3 -----> ? (h) Benzene + HCOCl, AlCl3 then 1-chlorobutane, AlCl3 -----> ?What is the product of the reaction P4O6 + H2O? A) H3PO4 B) H2PO4 C) H3PO3 D) H2PO3 E) H2P4O73. What is the main product for each of the following reactions? (a) p-Methylanisole + CH3CH2COCl, AlCl3 --------> ? (b) Toluene + Br2, light, then CH3CH2COCl, AlCl3 --------> ? (c) Acetophenone + CH3CH2Cl, AlCl3, then Br2, Fe -----> ? (d) Styrene + CH3CH2COCl AlCl3, then Br2, Fe -----> ? (e) Benzene + 1-chloro-2,2-dimethylpropane, AlCl3 -----> ? (f) Benzene + HCOCl, AlCl3 then 1-chlorobutane, AlCl3 -----> ? 4. Using arrows to show electron movement, write the stepwise mechanism for the major products in the reactions (3a and 3f) listed above.
- 2. How many substitution product/s is/are formed when metabromo anisole is treated with ammonia?A. 0-no reactionB. 1C. 2D. 36 H + + 6 I- + ClO3- → 3 I2 + 3 H2O + Cl- Which oxidizing reagent is in this reaction?A hydrocarbon of unknown structure has the formula C8H10. On catalytic hydrogenation over the Lindlar catalyst, 1 equivalent of Η2 is absorbed. On hydrogenation overa palladium catalyst, 3 equivalents of Η2 are absorbed. (a) How many degrees of unsaturation are present in the unknown structure? (b) How many triple bonds are Present? (c) How many double bonds are present? (d) How many rings are present? (e) Draw a structure that fits the data.
- 6 H + + 6 I- + ClO3- → 3 I2 + 3 H2O + Cl- Which oxidizing reagent is in this reaction? A. I2B.I-C. H+D. ClO3-6 H + + 6 I- + ClO3- → 3 I2 + 3 H2O + Cl- Which oxidizing reagent is in this reaction? A. I2B. I-C. H+D. ClO3-Chemistry 60. Give the principal product(s) expected, if any, when trans-1,3-pentadiene reacts under the following conditions. Assume one equivalent of each reagent reacts unless noted otherwise. (a) H2O, H3O+ (b) Na+EtO- in EtOH (c) Maleic anhydride heat
- Which of the following reactions result in a positive ∆ Ssys? A. Pb(NO3)2 (aq) +KCl (aq) ---> PbCl2 (s)+ KNO3 (aq) B. HCl (g) + H2O (l) ---> HCl (aq) C. H2 (g) + I2 (g) ---> 2HI (g) D. 2H2O (g) ---> 2H2 (g) + O2 (g) E. H2O (g) ---> H2O (l)Give the major product for the reaction. (CH3)2CHCHBrCH3 + -OC(CH3)31. What type of reaction is occuring in step 3? (halogenation, hydrohalogenation, reduction, keto–enol tautomerism, dehydrohalogenation, acid-catalyzed hydration, base-catalyzed hydration) 2. Which reagent is necessary for step 3? (Br2, HBr, H2/Pt, NaNH2, H20/H2SO4/HgSO4)



