0.5 100 200 300 400 500 600 700 -0.5 Wavelength (nM) The gray one: 2.4 M On a single graph, plot the absorbance spectrum of myoglobin with 0 M and 2.4 M GUHCT. Compare the two spectra and comment on how the addition of the denaturant GUĤCI affects the absorbance spectrum of myoglobin. Absor
0.5 100 200 300 400 500 600 700 -0.5 Wavelength (nM) The gray one: 2.4 M On a single graph, plot the absorbance spectrum of myoglobin with 0 M and 2.4 M GUHCT. Compare the two spectra and comment on how the addition of the denaturant GUĤCI affects the absorbance spectrum of myoglobin. Absor
Biochemistry
6th Edition
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Reginald H. Garrett, Charles M. Grisham
Chapter22: Gluconeogenesis, Glycogen Metabolism, And The Pentose Phosphate Pathway
Section: Chapter Questions
Problem 5P
Related questions
Question
Give answer all questions with explanation
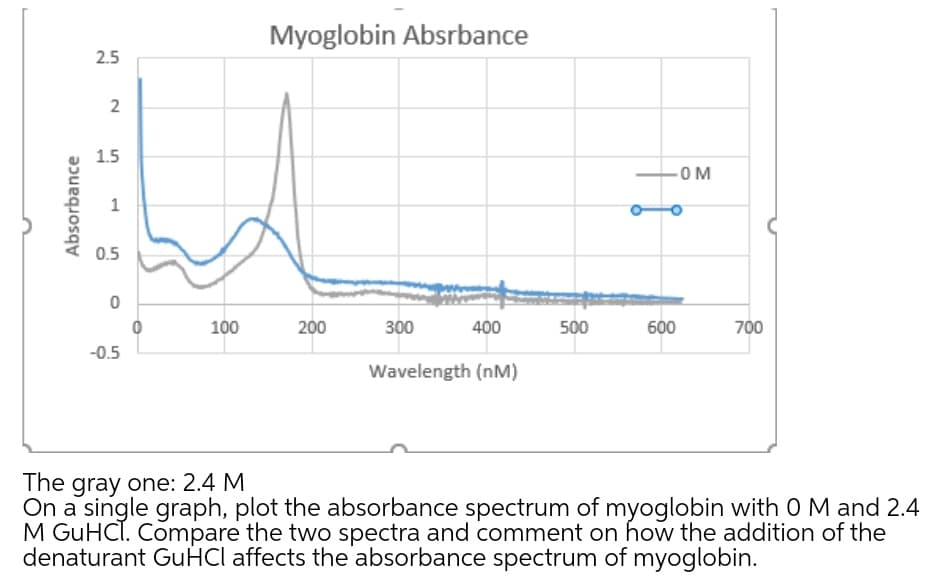
Transcribed Image Text:Myoglobin Absrbance
2.5
1.5
OM
0.5
100
200
300
400
500
600
700
-0.5
Wavelength (nM)
The gray one: 2.4 M
On a single graph, plot the absorbance spectrum of myoglobin with O M and 2.4
M GUHCI. Compare the two spectra and comment on how the addition of the
denaturant GUHCI affects the absorbance spectrum of myoglobin.
2.
1.
Absorbance
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Anatomy & Physiology
Biology
ISBN:
9781938168130
Author:
Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:
OpenStax College

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Anatomy & Physiology
Biology
ISBN:
9781938168130
Author:
Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:
OpenStax College