Q: Name and classify each compound a. CH3CH₂CH₂NH2 CH₂CH₂NCH b. CH₂ C. CH3CH₂NHCH2CH3 d. CH3CH₂CH2NHCH3
A: IUPAC nomenclature of amine: Name and Identify the longest chain of carbon bonded to the nitrogen…
Q: Choose the appropriate starting material for the products seen in the reaction below: A + B + Br
A:
Q: 3. Complete the information in the following table: Number of Valence Element Rb Energy level in…
A: Electronic configuration is arrangement of electrons in shells, subshells and orbitals.
Q: EXPERIMENT Prepare 2 clear glasses. Label the glasses with letter A and B Put cold water in letter…
A: Chemical kinetics can be defined as the branch of chemistry that deals with rates of chemical…
Q: 16.) Oxidative deamination occurs primarily on which of the following? a. mitochondria b. liver c.…
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: Lithium Oxygen Fluorine Bromine Silicon 3 15/2s! Orbital Diagram 14 1 to
A: Dear student , since you have posted multiple parts questions we will allow to solve only first…
Q: Give a short note of Baclofenac Sodium
A: Please find your solution : (Note : in question there should be either Diclofenac sodium or…
Q: An argon gas occupies 56.2 L at 760 torr. If the volume of gas is decreased to 1/4 of the original…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Fill in the blanks: A buffer solution is consists of 0.100 M HCN and 0.200 M NaCN. HCN + H₂O H30* +…
A:
Q: Below you will see a wedge-dash diagram, and an incomplete Newman projection with the designated…
A:
Q: Which of the following can be classified as an Arrhenius base? - CO32- - LiOH - H2O - all of…
A: According Arrhenius base is define when substance dissolved aqueous solution it's release OH- ions…
Q: Label each pair of compounds below as: a. conformational isomers b. stereoisomers C. constitutional…
A: We have to determine the relation between the given structures
Q: In this experiment, the problem is that the volume of the volumetric pipet is not accurately known…
A: As we know density and volume are related to each other Density = mass/ volume As volume is…
Q: H₂ с- CH3 CH3 C ОН H₂ I H₂ С. CH3 H₂ ОН
A: Hybridization is the overlapping of atomic orbitals to form same energy hybrid orbitals. In…
Q: At STP, three 500-ml flasks are filled with N2, 02, and He gas. Which of the following statements is…
A: There are three states of matter solid, liquid and gas. Solid has higher density than liquid and…
Q: Balance the SR and find it's ionic and net ionic. Al (s) + H3PO4 (aq)
A: Given that, Als + H3PO4 aq → We have to balance the given equation. Also, we have to write the ionic…
Q: olution is formed when the solute uniformly disperses oughout (or dissolves in) the solvent. The…
A: The process which absorbs energy is known as endothermic process and the process which release…
Q: Percentage purity of a sample of 0.1350 g of As2O3 assayed iodometrically using 23.5 mL of 0.1055N…
A: Here we have to determine percentage of purity of As2O3 in 0.1350 g of sample when it use 23.5 ml of…
Q: 2. Given the following table of standard reduction potentials for a hypothetical element X under…
A: #C: The frost-Ebsworth diagram is plotted by taking n*Eo on the Y-axis and oxidation states of…
Q: All of the following statements regarding galvanic cells are TRUE EXCEPT: (A) Electrons flow from…
A: Galvanic cell is used to convert chemical energy stored to electrical energy as a result of chemical…
Q: Feed gas containing of 78.5mol % H₂, 21% of N₂ & 0.5% of Ar is mixed with recycle gas and enters a…
A: NH3 is produced by Heber's cycle. Material balance method used here.
Q: Gaseous methane (CH4) will react with gaseous oxygen (O₂) to produce gaseous carbon dioxide (CO₂)…
A:
Q: Question # 3: Draw or name the following organic compounds. You may draw complete structural or line…
A:
Q: Provide the correct IUPAC name for the compound shown here. CI
A: We can write the IUPAC name of the compound according to the given rule... 1. first we identify the…
Q: Fill-up in the blanks: Common Name 1. N-tert. pentyl- valeramide 2. (5) IUPAC Name (1)…
A: 4) amide 8) anhydride
Q: 11. Order: Ticar (ticarcillin disodium) 1 g IVPB q6h, infuse in 50 mL D5W over 45 minutes. The…
A: The solution of the given question is
Q: Calculate energy of a photon having the frequency of 1.68 x1015 1/s
A:
Q: 185 mL. of a solution contains 1.55 g glucose. Calculate the molarity of solution. Glucose =…
A: Given, mass of glucose = 1.55 g Volume of the solution = 185 mL = 0.185 L molar mass of glucose =…
Q: Starting from the structure below (sighting down the indicated bond). rotate the back carbon to…
A: In this question, we will draw the Newman projection conformation in the eclipsed form. Eclipsed…
Q: for letter d,e please check if correct D.CH3CH2CH2CH(CH(CH3)2)CH2C(CH2)2(CH3)2CH2CH2OH…
A: The complete structures are already given in the solution. The condensed formula for D and E are…
Q: What are the processes occurring in the electrolysis of an aqueous solution of potassium sulphate:…
A: Here we have to to predict the reaction occur at the respective electrode during elctrolysis of…
Q: Calculate the max using Woodward-Fieser Rules te
A: The woodworld fisher rule is used to calculate the Lambda maximum value of the given compound.
Q: Assign R, S configurations to each indicated chirality center in the molecules below. OH B. HO COOH…
A:
Q: Consider the reaction A + B arrow C. If the initial concentration of A is 5.0 mol L–1 and the…
A:
Q: Answer the following questions regarding the reaction coordinate diagram below.
A: Given; diagram
Q: 4. Use short notation to write the electron configuration of: A) Se B) Te-2
A: Electrons are filled in a different atomic orbital in an atom. According to the Aufbau rule, the…
Q: Place the letter corresponding to the correct answer in the blank to the left of the reaction. Br Br…
A:
Q: 1. Calculate the mass of solid NaOH required to make 1.0L of a 0.50M NaOH solution.
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: the type of measurement to the unit given below. the appropriate labels to their respective targets.…
A: The appropriate labels are: 1) millilitre- volume
Q: QUESTION 10 Which element has atoms with average mass about 4 times those of the average aluminum…
A: 10. Atomic mass or average mass of aluminium is 27g. The 4 times mass of aluminium is 4 × 27 = 108 g…
Q: III. The initial rates for the reaction of hydrogen and nitrogen: 3H₂(g) + N₂(g) → 2NH₂ (g) were…
A: Given that, a reaction is 3H2 g + N2 g → 2NH3 g. The given data is Experiment Initial [H2] (M)…
Q: Determine the valence electronic configuration of an atom in period 6 and 4A group.
A: Atom present in period 6 and group IV A is Lead (Pb). It's valance electronic configuration is 6s2…
Q: Provide IUPAC names for the following structures. H₂C Br CH3 a. Br Br H₂C b. Br Br
A: The IUPAC name of the given structures are:
Q: List all the electronic transitions possible for a. CH4 b. CH3CI c. H₂C=O
A: List all the electronic transitions possible for CH4
Q: Suppose 0.159 g of lead(II) nitrate is dissolved in 150. mL of a 40.0 m M aqueous solution of…
A: Here we are required to find the Molarity of lead cation
Q: 2. Sketch (as accurately as you can) steady state i vs E curves for solutions prepared from: a) 1 mM…
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: Question is based on the following definitions. Plants can propagate, or reproduce themselves, by…
A: This reproductive form resembles bulb most closely as in that too by dividing plant starts growing…
Q: Ch. rest (equilibrium only) Be sure to look up the Ka values before you leave class or use the KA…
A:
Q: 43 44 45 Tc Ru Rh Pd Ag Cd In Technetium Ruthenium Rhodium Palladium Silver Cadmium Indium [97]…
A:
Q: Carboxylic Acid Draw the condensed formula of the following compounds: 4.…
A:
draw structure of enol form
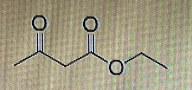
Step by step
Solved in 2 steps with 2 images


