Q: What is the product of the following transformation? H₂O, H* heat 1 2 3 4 OH is the Im NH₂ OH 3 1…
A:
Q: Explain using an example the relationship of chemical potential to phase equilibrium.
A: Chemical potential is a thermodynamic property that describes the potential energy of a substance to…
Q: chemical reaction has rate constants of 50L/mol s at O C and 100 L/mol s at 10 C. Most nearly, what…
A: R is gas constant and it is equal to 8.314 j/mol k
Q: Bicyclo[2.2.0] hexane -> cyclohexane-1,4-dione ?
A: The bromination( 1 equivalent of Br2) under photochemical conditions, the bicyclo[2.2.0] hexane…
Q: X = Cl or l Select one alternative: Olodocyclohexane is more stable than chlorocyclohexane The pKaH…
A: Answer: E1 elimination reaction takes place through carbocation formation and since carbocation is…
Q: Jeff followed to procedure for Experiment 4, and collected the following data, which he recorded on…
A: Answer: In this experiment an unknown substance has been made to react with HCl and we have to find…
Q: You are using a Vernier Colorimeter to measure the concentration of Yellow dye #6 in Powerade Which…
A: Yellow dye #6 has a maximum absorption at around 430 nm, which means that it will absorb light most…
Q: Write a balanced chemical equation based on the following description: aqueous silver nitrate reacts…
A:
Q: If 65 mL of hydrochloric acid (HCI) is diluted with water to a total volume of 225 mL, what percent…
A:
Q: Write the thermochemical equation for the reaction represented in the graph below (include the…
A: We have to write a thermochemical equation for the reaction shown
Q: Preparation of the buffer You have a solution of 50X TAE buffer. You want to prepare 320 mL of 1X…
A:
Q: B) How many moles of KHP were used? C) What volume of NaOH was needed? D) What it the molarity of…
A: For the neutralization reaction between KHP and NaOH, for both components 1 mol amount is required,…
Q: 2. s) Xenon-125 undergoes radioactive decay. The half-life of the decay is 16.9 hours. How much of a…
A:
Q: In reaction with aqueous ammonia, Zn(OH)2 forms a complex ion. Explain the term complex ion, and…
A: A complex ion is a charged species that is formed by a central metal ion or atom that is bound to…
Q: 7. Which compound would have the lowest boiling point at standard pressure? a. CH4 b. CH3CH₂CH3 C.…
A: To compare the organic compounds on the basis of their boiling points.
Q: Write the chemical equation for the reaction of propylamine with water. Express your answer as a…
A: In this question, we are asked to write and explain the reaction of propylamine with water including…
Q: You have been asked to determine which heavy metals, if any are in a sample of sewage sludge. Why is…
A: In environmental science, it is essential to determine the presence and concentration of heavy…
Q: For the reaction: 2A-B The concentration of A was measured for one hour and the following data were…
A: For a general reaction, aA + bB→cC + dD The rate of appearance and disappearance can be given as…
Q: During hot days, fish stay at the deeper cooler part of a body of water. Can you explain this…
A: Answer: In a solution substance that is in lower quantity is called as solute and that is in excess…
Q: Curved arrows are used to illustrate the flow of electrons. Using the provided arting and product…
A:
Q: 3. Calculate the rate constant of a reaction at 450 °C if the reaction rate is 6.75 x 10-6 M/s at 25…
A: Given, Activation energy (Ea) = 35.5 kJ/mol = 35.5 × 1000 J/mol = 35500 J/mol Rate constant (k): k1…
Q: 3. Compared to a linear alkane, a cyclic alkane with the same number of carbons would contain: a. 2…
A: Given linear alkane and cyclic alkane
Q: Study this chemical reaction: Zn + Cl₂ → ZnCl₂ Then, write balanced half-reactions describing the…
A:
Q: The enthalpy for combustion for ethanol at 298 K is -1370.7 kJ/mole and occurs via the following…
A: Internal energy is a fundamental concept in thermodynamics that refers to the total energy of a…
Q: Draw the product of the reaction shown below. Ignore inorganic byproducts. cat. H2SO4 HNO3 (1 equiv)
A: Thus is a Nitration reactionn of aromatic compound: Aromatic compound reacts with the nitrating…
Q: A solution has a [H 3O +] of 1.930x 10 -10 M. What is the pOH?
A:
Q: A solution is prepared that is initially 0.34 M in propanoic acid (HC₂H₂CO₂) and 0.15M in sodium…
A: Given: The initial concentration of propanoic acid (HC2H5CO2) = 0.34 M The initial concentration of…
Q: 40 30 20 10 пишиться 4000 3500 Н Laff 3000 озн 2500 2000 Wavenumbers или 1500 1000 а мно
A: The question is based on IR spectroscopy. we need to analyse and interpret the spectra.
Q: The following results were obtained in the replicate determination of the lead content of a blood…
A:
Q: What is the pH of a 0.785M solution of formic acid,HCHO2?The Ka of HCHO2 is 1.77x10-4?
A:
Q: Liquid octane CH3CH26CH3 reacts with gaseous oxygen gas O2 to produce gaseous carbon dioxide CO2 and…
A: Given, mass of octane (CH3(CH2)6CH3) reacts = 1.1 g mass of Oxygen gas (O2) reacts = 2.0 g Actual…
Q: 7) The equilibrium constant for a chemical reaction was found to depend on tempera- ture over the…
A: This problem is based on Thermodynamics and here we will be using second law of thermodynamics to…
Q: any I carbon molecules Inf
A: Given that, a synthesis scheme is shown below We have to carry out the above synthesis.
Q: Drawn are four isomeric dimethylcyclopropanes. A B C D a. How are the compounds in each pair related…
A: Since you have posted a question with multiple sub-parts, we will solve the first three subparts for…
Q: Write the correct IUPAC name for each of the following structures
A: For the systematic name or IUPAC name of organic compounds there are some rules as follows; Choose…
Q: A solution is prepared that is initially 0.18M in hydrocyanic acid (HCN) and 0.44M in sodium cyanide…
A: Given: The concentration of HCN = 0.18 M The concentration of NaCN = 0.44 M We have to complete the…
Q: 12. The reaction between a carboxylic acid and an amine is an example of: a. addition C.…
A:
Q: Label the stereogenic center(s) in each drug as R or S. L-Dopa is used to treat Parkinson's disease…
A: Stereogenic centre: A carbon with 4 different atoms or groups attached to it
Q: Decide whether the Lewis structure proposed for each molecule is reasonable or not. molecule…
A: We have been given Lewis structures of three molecules and we have been asked to check whether these…
Q: Complete the balanced molecular chemical equation for the reaction below. If no reaction occurs,…
A:
Q: Complete the table below. Round each of your entries to 2 significant digits. You may assume the…
A:
Q: Read each description in the first column of the table below. If any chemical element with atomic…
A: The periodic table is a tabular arrangement of chemical elements in order of increasing atomic…
Q: draw the 3 TLC plates of three parts from the experiment of reduction of benzil by NaBH4 to…
A: The question is based on thin layer chromatography, TLC. It is a method of separating components…
Q: How many lattice paths are there from the point (−3, 10) to the point (5, −2) if the pathway must…
A: To find the number of lattice paths from the point (-3, 10) to the point (5, -2) by only moving…
Q: polysaccharides 1) what is the predominant functions group in polysaccharides 2) what types of…
A: Ask other parts in another questions. The first 2 parts have been answered below. Natural…
Q: The molar mass of copper(II) chloride is 134.5 g/mole. O True False
A:
Q: 3. Cl(aq) + Ag+¹(aq) →
A: Given reactant are- Cl-(aq) + Ag+(aq) -----> ??? The reaction between Cl- (aq) and Ag+(aq) is…
Q: The preparations of two aqueous solutions are described in the table below. For each solution, write…
A: A chemical reaction occurs between an acid and a base to give salt and water. This reaction is known…
Q: DI. Sulfuric acid reacts with CuO(s) to form soluble CuSO4(aq). El. Zinc displaces H*(aq) from…
A: D1. The chemical reaction for CuO (s) with sulfuric acid, H2SO4 (aq) is, CuO (s) + H2SO4 (aq) =>…
Q: choose which set is the correct coefficient for the following equations: SbCl3+Na2S----Sb2S3+NaCl
A: In balanced chemical reaction number of each atom on reactant side is equal to number of each atom…
D.24.
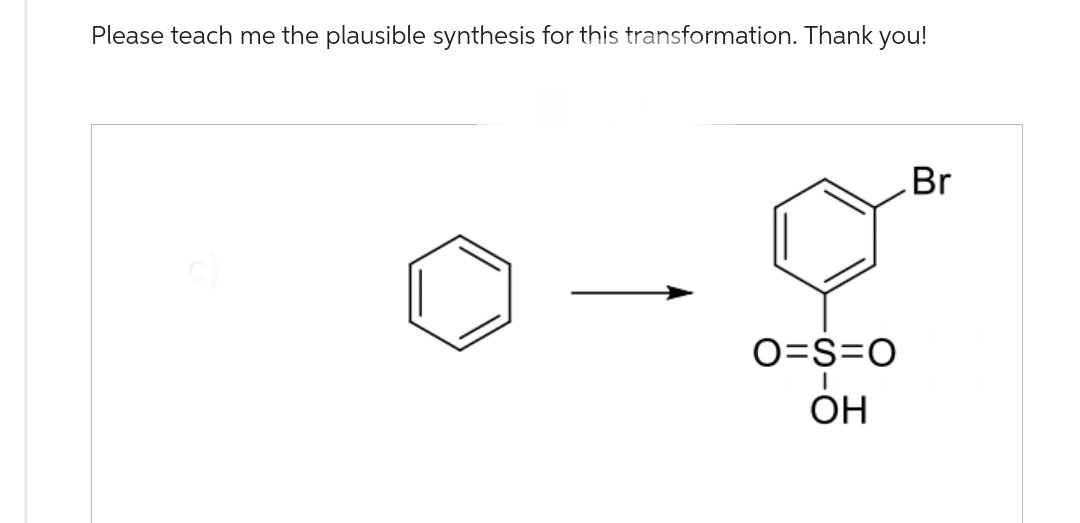
Step by step
Solved in 2 steps with 1 images

- Kindly give out a detailed mechanism for the transformation belowWhich compounds from 5a-d will react with hydroxide in the fashion shown. Which reaction will be the fastest and slowest? Explain using knowledge of carbonyl chemistry, pka and leaving groups.What is the main product from the reaction sequence below?
- Why does this reaction not undergo SN2 ?A4 Express the retro synthesis scheme using the suggested starting material (SM) for the following target compound (TM). After performing the retro synthesis analysis, synthesize the target compound according to the analyzed scheme.Please state which alkene gives a meso compound upon reaction with bromine (Br2), thank you

