Q: if the [OH-] is 1.45x10 to the power -10 what is the pH?
A:
Q: Understand:- 2. Explain how the following factors affect acidity. - Charge - The Role of the Atom…
A: Ans.2 i) Charge and acidity: When acidic compounds donate hydrogen ions or accepts electrons, they…
Q: a) 50.00 mL of 0.1500 M NaOH is used to titrate 15.25 mL of H3PO4 to the endpoint (neutralization).…
A:
Q: The two half-reactions for the lithium battery are shown below. Describe the direction of…
A: In any cell, oxidation always takes place at anode and reduction takes place at cathode. Oxidation…
Q: Calculate the pH for a buffer made with 230.0 mL of 0.189 M furoic acid (HC5H3O3) and 20.0 mL of…
A:
Q: + HBr ROOR H₂O H₂SO4 H₂0 H₂SO4 H₂SO4 1) BH3 2) H₂O₂/NaOH 2 Br₂ 2 HCI 1) Hg(0Ac)2, H₂O 2) NaBH4 1)…
A: Fill in the blanks with appropriate structures to identify the major product of the reaction.
Q: 5. Draw several resonance contributors for the cyanate ion, [OCN]. Circle the two lowest energy…
A:
Q: Calculate the amount of fertilizer needed to meet the recommended rate for eggplants which is…
A: Recommended rate of fertilizer for eggplant = 120-60-15 Kg/ha Nitrogen = 120 Kg Phosphorus = 60 Kg…
Q: 1. what grouo does the ff organic compound belong? a. ketone b. ether c. cyloalkane d. esther 2.…
A:
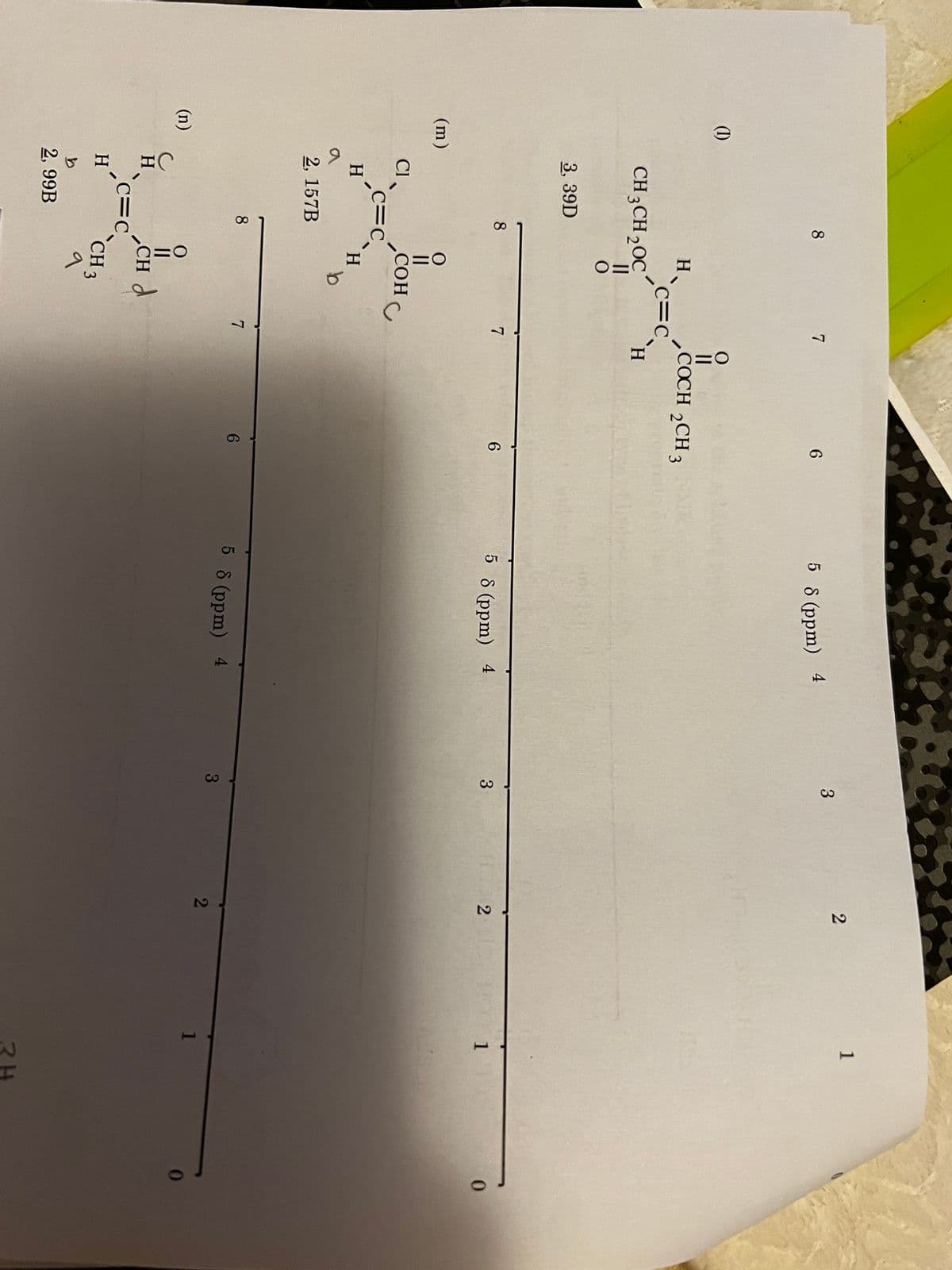
Q: The arrow points to a particular carbon atom that carries an H atom or group of H atoms. This/these…
A: In this question, we will see that appearing of hydrogens in proton NMR spectra as a singlet,…
Q: General Chemistry 4th Edition McQuarrie Rock • Gallogly Sodium sulfate is slowly added to a solution…
A: Given : 0.0500 M = Ca2+( aq ) 0.0300 M = Ag+( aq ) Ksp of CaSO4 = 4.93 × 10^-5 Ksp of AgSO4 =…
Q: A galvanic cell at a temperature of 25.0 °C is powered by the following redox reaction: 2Cr3+ (aq) +…
A: We can use Nerst Equation to solved the above problem.
Q: 0.05 N AgNO3 titrated with (10 ml) NaCl. Calculate the N of NaCl? [when volume of AgNO3 titrated…
A: Here we have to determine the Normality of NaCl from the following given titration with AgNO3…
Q: What is the volume in liters of 13.7 mol of krypton gas at STP?
A: We have to calculate the volume in liters of 13.7 mol of krypton gas at STP
Q: A balloon is filled with 2 liters of air at sea level. If the balloon is taken up a mountain so that…
A: Given: The volume of the balloon at the sea level is 2 liters. We have to calculate the volume of…
Q: Finish the diagram (from top to bottom) Mixture, silica, air bubble, silica gel, sand, glass wool…
A: We have to answer the given questions about the column chromatography and the thin layer…
Q: how many mols of solute are in 125mL of a 2.0M hydrochloric acid solution?
A: We have to predict the number of moles.
Q: Present 5 chemical reactions involved in biological and industrial processes affecting life and the…
A: Answer - Present 5 chemical reactions involved in biological and industrial processes affecting life…
Q: Calculate the Eºcell given the following half-cells. anode: Ca2+ (aq) + 2e- → Ca(s) cathode: Fe³+…
A: Redox potential is a measure of the tendency of a chemical species to acquire electrons from or lose…
Q: Student A did not see any spots on her TLC plate after visualization under iodine vapor. Which of…
A: Thin layer chromatography
Q: if a volume of a gas is at 19 L and 45C with the pressure of 25 ATM what will the pressure be in…
A: we need to find the pressure when volume of gas changes
Q: In which equation(s) do/does water act as a Bronsted-Lowry base? You may choose one or more…
A: Given -: Acid base reactions In which equation water act as Bronsted-Lowry base
Q: What is the mfp of 0.0253 eV neutrons in Zr? For Zr: A = 55.8, p = 6.5 g/cm³, os = 8 b, oa = 0.18 b.
A: Given: For Zr, A = 55.8 g/mol Density = 6.5 g/cm3 σs = 8b σa = 0.18b Na = 6.023 x 1023 (Avagadro's…
Q: Choose the two atoms from the periodic table that, when bonded together, would theoretically form a…
A: Polar bond is the bond that has net dipole moment and non polar is the bond that has zero dipole…
Q: Arrange the following complexes in terms of ligand strength: [Cr(OH)6]3–, [Cr(NH3)6]3+, and…
A: The ligand strength is determined by the value of ∆° ie splitting energy. Various ligands are…
Q: i. NaH, DMF ii. CH3CH₂Br iii. NaOH, H₂O iv. H₂O*, heat ?
A:
Q: l2O5+5CO 5CO2+l2 80.0 g of iodine (V) oxide reacts with 28.0 g of carbon monoxide what is the…
A:
Q: (Q89) What is the value (in kJ) of the standard Gibbs free energy change (AGOrxn) for the following…
A: Given-> E°Ni2+/Ni = -0.23 V E°Au3+/Au = 1.50 V
Q: Which among the following statements best explains the purpose that sulfuric acid serve in the…
A:
Q: Write down the condensed molecular formula, line and angle structure and full 3-D structures for…
A: Condensed formula: In this, a molecule is the formula where the symbols of atoms are listed in order…
Q: Classify each reaction type: 1) MN + QP → QN + MP 2) CxHyOz + O2 → H2O + CO2 3) JR → R + J 4) B + D…
A: Here we are required to classify the type of reaction
Q: A chemist measures the energy change AH during the following reaction: 2 NH3(g) → N₂(g) + 3H₂(g) ΔΗ…
A: Here for that reaction ∆H = 160 kJ As , ∆H is postive value so , we can say it is endothermic…
Q: Q3/ Answer True or False for each of the following: ( 1) Salicylic acid can be prepared from…
A:
Q: Which of the following combination of alcohol and haloalkane will proceed to form an ether? a…
A: Answer is written below
Q: The standard cell potentials for following reaction at standard room temperature are E Fe/Fe =…
A: Given-> E°Fe2+/Fe = -0.447 V E°Ag+/Ag = 0.7996 V
Q: Above what Fe2+ concentration will Fe(OH), precipitate from a buffer solution that has a pH of 8.13?…
A:
Q: The standard cell potentials for following reaction at standard room temperature are = -0.447 V E…
A:
Q: Which of the following is expected to form an ideal solution?
A:
Q: (Q100) The diameter of a titanium-46 nuclide is 7.214 fm (1 fm = 10-15). Given that the mass of…
A: Atom is made up of protons, neutron and electrons. In atom, there is nucleus in center and electrons…
Q: Use the free energies of formation to calculate the equilibrium constant (K) for the following…
A:
Q: Provide the following: A) Given B) Formula C) Calculations and D) Answer.| Calculate the normality…
A: The given problem is related to the standardization of iodine solution using arsenic trioxide. The…
Q: A hydrocarbon mixture known to contain methane, ethene and butane only has a volume of 6.00L, a…
A:
Q: Indicate whether the statement below is true or false. Both charge and mass are conserved during a…
A: Given statement: Both charge and mass are conserved during redox reactions.
Q: What is formed when a halogen molecule replaces hydrogen molecule(s) in an aromatic hydrocarbon? 2.…
A: The answer have been solved below
Q: what is the percent of silver in silver nitrate?
A: Since, MM of Ag = 107.8682 g mol-1 MM of AgNO3= 169.87 g mol-1 % Ag = ?
Q: What is the name of Reaction 1 and 2? Choices: A. FC Acylation, B. SN Alkylation, C. FC Alkylation,…
A:
Q: determine the number of grams of iodine gas in a 3.0 L container at 300K and a pressure of 1.5 ATM
A:
Q: The following reaction produces: || CH3CH₂-C-OCH(CH3)2 + NaOH CH3COONa+ CH3CH(OH)CH3 CH3CH2COONa+…
A:
Q: What is the underlying, fundamental force for the formation of all chemical bonds? Select one: O A.…
A: Chemical bond is the electrostatic force between two or more atoms to hold them together. There are…
Q: Based on the general procedure for this reaction, how much of each reagent is needed to produce the…
A:
I'm having trouble to solve L.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 6 images

- Hexanoic acid was added to an immiscible biphasic solvent sysem, water and CCl4 at 20.0OC and the equilibrium concentrations of hexanoic acid were determined to be 3.66 g/L in H2O and 67.0 g/L in CCl4. Caluclate the distrubution coeffiecent (D1) of hexanoic acid in CCl4 with respect to water.What is ΔSsurr for a reaction at 28.6 °C with ΔHsys = 38.9 kJ mol-1 ? Express your answer in J mol-1 K-1 to at least two significant figures.Answer Q45, 46,
- What is the Ka and Kb expression for a and b? a) C6H5COOH b) CN-Please create a caption for this table. Solution NaCl Conc. (%) Osmolality (mOsm) % transmittance Absorbance % hemolysis % crenation C distilled 0 0 0.001029 4.987584625 100 0.03354 1 0.177179111 54.61 0.001551 4.809388202 96.42720001 0.05837 2 0.297126222 91.58 0.01012 3.994819487 80.09527231 0.08444 3 0.442542222 136.4 3.849 1.414652089 28.3634704 0.134 4 0.590164444 181.9 64.8 0.188424994 3.777880643 0.2125 5 0.74752 230.4 95.64 0.019360433 0.388172513 0.3368 6 0.89644 276.3 99.56 0.001915112 0.038397585 0.5336 7 1.095648889 337.7 99.98 8.68676E-05 0.001741676 0.9834 8 1.336711111 412 100 0 0 2.1 9 1.755568889 541.1 100 0 0 7.9 10 2.674395556 824.3 100 0 0 57.83 11 4.490211111 1384 100 0 0 99.72Q5: If the Ksp of Ca3N2 is 6.06 x 10-3, how many moles of calcium nitride (s) could you dissolve in 1.589 L of solvent (assume that calcium nitride (s) dissolves in water to form Ca2+(aq) and N3- (aq))? Enter your answer with at least 3 sig figs. Please provide only typed answer solution no handwritten solution needed allowed

