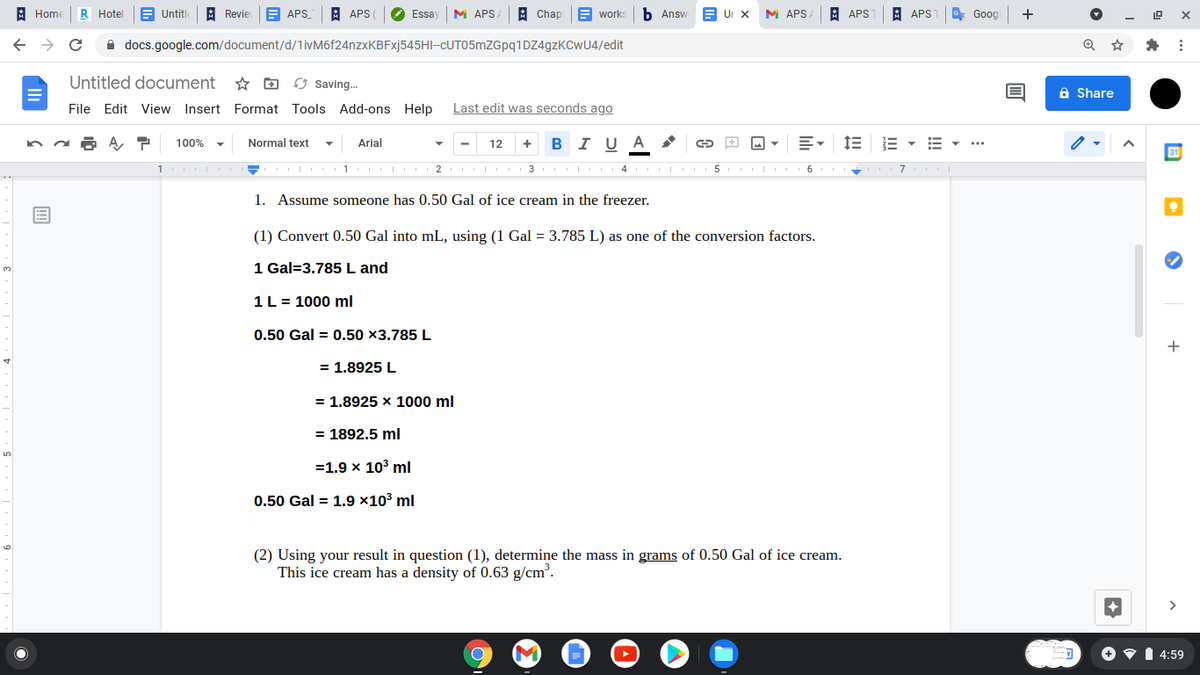
1. Assume someone has 0.50 Gal of ice cream in the freezer. (1) Convert 0.50 Gal into mL, using (1 Gal = 3.785 L) as one of the conversion factors. 1 Gal=3.785 L and 1L = 1000 ml 0.50 Gal = 0.50 ×3.785 L = 1.8925 L %3D = 1.8925 x 1000 ml %3D = 1892.5 ml =1.9 x 10° ml 0.50 Gal = 1.9 x10³ ml (2) Using your result in question (1), determine the mass in grams of 0.50 Gal of ice cream. This ice cream has a density of 0.63 g/cm.
1. Assume someone has 0.50 Gal of ice cream in the freezer. (1) Convert 0.50 Gal into mL, using (1 Gal = 3.785 L) as one of the conversion factors. 1 Gal=3.785 L and 1L = 1000 ml 0.50 Gal = 0.50 ×3.785 L = 1.8925 L %3D = 1.8925 x 1000 ml %3D = 1892.5 ml =1.9 x 10° ml 0.50 Gal = 1.9 x10³ ml (2) Using your result in question (1), determine the mass in grams of 0.50 Gal of ice cream. This ice cream has a density of 0.63 g/cm.
Introductory Chemistry: An Active Learning Approach
6th Edition
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Mark S. Cracolice, Ed Peters
Chapter3: Measurement And Chemical Calculations
Section: Chapter Questions
Problem 128E: In Active Example 3-29 you calculated that you would have to work six weeks to earn enough money to...
Related questions
Question
- Express each result in correct number of significant figures and with correct unit.
Transcribed Image Text:: Home
R Hotel
E Untitle A Revie E APS
A APS
Essay M APSA
A Chapi E works b Answ B Ur x MAPS A
A APS1 A APST
* Goog
+
A docs.google.com/document/d/1ivM6f24nzxKBFxj545HI-CUT05mZGpq1DZ4gzKCwU4/edit
Untitled document * D 3 Saving.
a Share
File Edit View Insert Format Tools Add-ons Help
Last edit was seconds ago
+ BI UA
E- 1E E -
100%
Normal text
Arial
12
31
1
I 1 I 2 3 I 4
5
|
1. Assume someone has 0.50 Gal of ice cream in the freezer.
(1) Convert 0.50 Gal into mL, using (1 Gal = 3.785 L) as one of the conversion factors.
1 Gal=3.785 L and
1L = 1000 ml
0.50 Gal = 0.50 ×3.785 L
+
= 1,8925 L
= 1.8925 x 1000 ml
= 1892.5 ml
5.
=1.9 x 103 ml
0.50 Gal = 1.9 ×10³ ml
6.
(2) Using your result in question (1), determine the mass in grams of 0.50 Gal of ice cream.
This ice cream has a density of 0.63 g/cm.
+ • 1 4:59
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning



Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning



Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning
