1. Calculate the percent composition of sucrose ( C12H22011) %O = 176.00 g/mol x 100 = 51.41% 342.34 g/mol O- 11 x 16.00 = 176.00 g/mol 22.22 g/mol %H = 22.22 g/mol x 100 = 6.49% 342.34 g/mol H- 22 x 1.01 %3D C- 12 x 12.01 g 144.12 g/mol = 342.34 g/mol % C = 144.12 g/mol x 100 = 42.10% 342.34 g/mol %3D Total 100 % Hint: Use the atomic mass of elements in the table below (taken from the Periodic Table of Elements) in solving for Molar Mass and Percentage Composition. Element Atomic Mass Element Atomic Mass Al 26.98 amu CI 35.45 amu C. 12.01 amu Fe 55.84 amu 18:88 8MB 14.01 amu Mg 24.31 amu
1. Calculate the percent composition of sucrose ( C12H22011) %O = 176.00 g/mol x 100 = 51.41% 342.34 g/mol O- 11 x 16.00 = 176.00 g/mol 22.22 g/mol %H = 22.22 g/mol x 100 = 6.49% 342.34 g/mol H- 22 x 1.01 %3D C- 12 x 12.01 g 144.12 g/mol = 342.34 g/mol % C = 144.12 g/mol x 100 = 42.10% 342.34 g/mol %3D Total 100 % Hint: Use the atomic mass of elements in the table below (taken from the Periodic Table of Elements) in solving for Molar Mass and Percentage Composition. Element Atomic Mass Element Atomic Mass Al 26.98 amu CI 35.45 amu C. 12.01 amu Fe 55.84 amu 18:88 8MB 14.01 amu Mg 24.31 amu
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter3: Stoichiometry
Section: Chapter Questions
Problem 76E: In 1987 the first substance to act as a superconductor at a temperature above that of liquid...
Related questions
Question
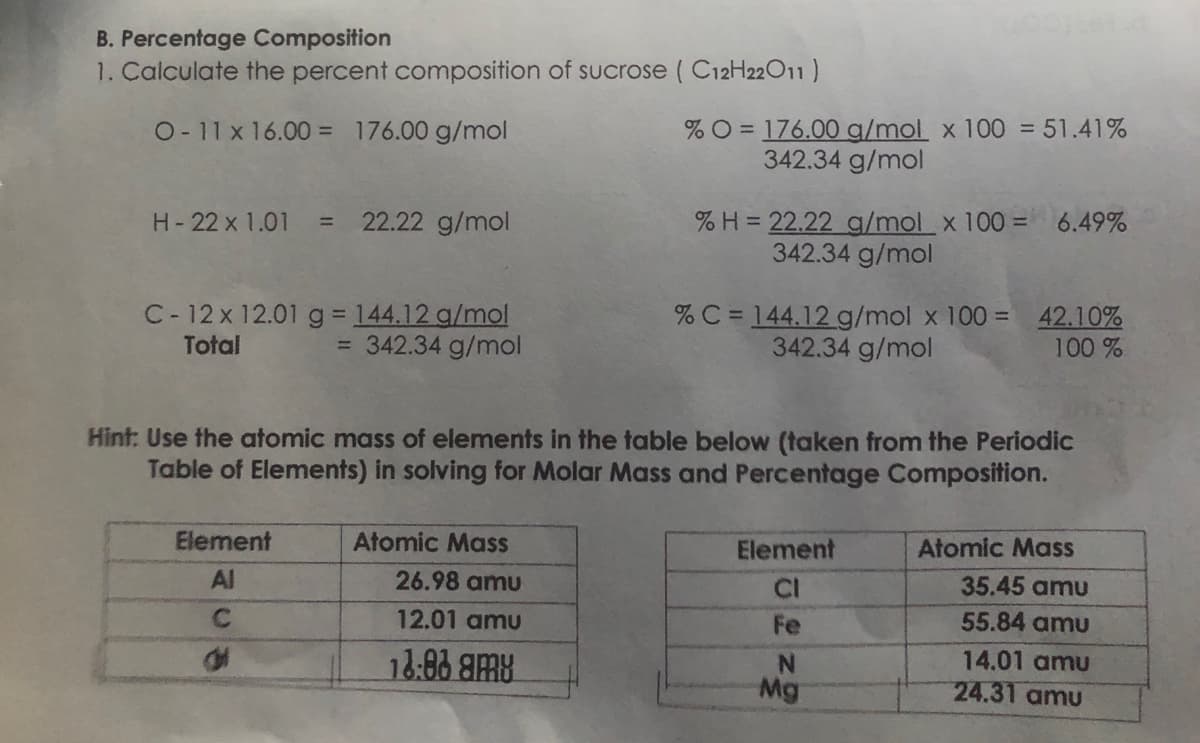
Transcribed Image Text:B. Percentage Composition
1. Calculate the percent composition of sucrose ( C12H22011)
%O = 176.00 g/mol x 100 = 51.41%
342.34 g/mol
O- 11x 16.00 = 176.00 g/mol
% H = 22.22 g/mol x 100 = 6.49%
342.34 g/mol
H-22 x 1.01
22.22 g/mol
%3D
C- 12 x 12.01 g 144.12 g/mol
= 342.34 g/mol
%C = 144.12 g/mol x 100 = 42.10%
342.34 g/mol
Total
100 %
Hint: Use the atomic mass of elements in the table below (taken from the Periodic
Table of Elements) in solving for Molar Mass and Percentage Composition.
Element
Atomic Mass
Element
Atomic Mass
Al
26.98 amu
CI
35.45 amu
12.01 amu
Fe
55.84 amu
14.01 amu
N.
Mg
24.31 amu
RWB 982L
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax