Q: Draw a molecular level picture of bromine at 350K and a molecular level picture of bromine at 320K.…
A: To find: 1. Draw a molecular-level picture of bromine at 350K2. Draw a molecular-level picture…
Q: D-o +) S.
A: We know that, According to Huckel's rule, an aromatic compound must have (4n + 2)π electrons that…
Q: Help me solve the problem which answer is right?
A: Arrhenius base: These are class of base that increase hydroxide concentration in the solutions. More…
Q: Draw a labeled molecular diagram to explain how the Law of Mass Conservation applies to the chemical…
A: Law of conservation of mass states that, the mass of reactants before the reaction is equal to the…
Q: Draw cyclobutene, fill in all H’s .
A: Given that, We have to draw the structure of cyclobutene including all H.
Q: Br2
A: Reaction of alkynes with Br2 is an example of electrophilic addition reaction. Br2 gets added to…
Q: Step 1 Draw step 1. Include lone pairs and formal charges in your answer.
A: Given reaction is dehydration of alcohol which happens in the presence of H2SO4 in which H+ and OH…
Q: The attractive and repulsive forces in an atom are rather complex. An electron is attracted to the…
A: Solution : Coulombic attraction is defined as the attraction of oppositely charged…
Q: Give the name of the alkyne below?
A: IUPAC name of the organic compound is composed of the following three components: Root name =…
Q: Please help slove which answer is right
A: Answer
Q: Calculate the Rf, for the spot from Bernie marker(B) Whose market is more polar and least polar?
A:
Q: Part D Draw the structure of the tertiary alcohol that has the formula Ç,H100. Draw the molecule on…
A: To solve this problem we have to draw the structure of tertiary alcohol .
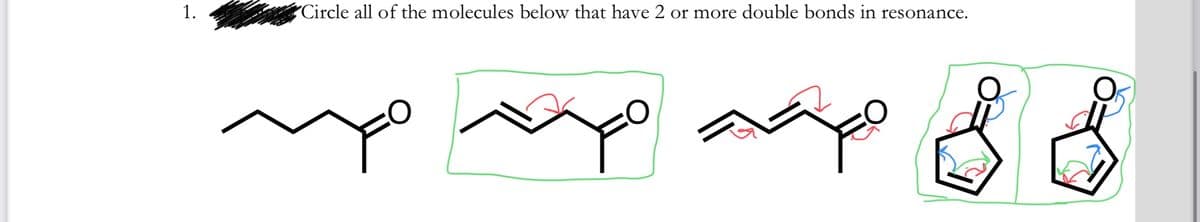
Q: Circle the entire molecule if it is aromatic or circle the portion of a molecule (if any) that is…
A:
Q: H. H H. エ エ
A: More s character in the hybridization of carbon leads towards more acidic hydrogen attached to it.…
Q: Why is less energy required to break a C-H bond to form a more highly substituted radical? Can…
A: Concept - Free radical are chemical species that have odd number of electrons. They are electron…
Q: Resonance, hybridization, Lewis structures: Draw the most important resonance contributor of the…
A:
Q: Peak position, peak characteristics, bond type, functional group?
A: To label peak positions, characteristics, bond type and functional groups.
Q: :0: H H. H. H H A B +Z-
A:
Q: H3C H3C- st Edi CH3 CH3 CH3
A:
Q: Draw both resonance structures of the anion formed by the reaction of the most acidic C-H bond of…
A:
Q: draw the arrow formalism on the 1st structure (top) that accounts for the 2nd resonance structure…
A: The arrow formalism on the 1st structure (top) that accounts for the 2nd resonance structure…
Q: Follow the flow of electrons indicated by the curved arrows in the following polar reaction. Draw…
A: The arrows in an organic reaction indicate the direction of the flow of electrons from one species…
Q: 1. Is the molecule below aromatic? If so, circle the atoms that are part of the aromaticity. Also…
A: Aromatic criteria: (4n+2)π electrons So in the ring there are 6π electrons that follows (4n+2)π…
Q: many h-bond donor and H-bond acceptor are there?
A:
Q: Name the following unbranched molecules from the numbered cards. When using a locator number use a…
A: Since you have posted question with multiple sub-parts, we are entitled to answer the first 3 only.…
Q: Draw an exact 3-D representation of the molecule from the model.
A: Hi, since you have posted questions with multiple subparts. We will answer the first three subparts…
Q: Which curved arrows movement corresponds to the formation of the product. Br Br Br
A:
Q: Atenolol
A: Question 4: Due to it's structural peculiarity i.e asymmetric carbon atom. This chiral carbon centre…
Q: the structures of fluorene and fluorenon
A: In this question we have to know about fluorene and fluorenone , their structure and also their…
Q: For each pair of molecules, determine whether umpolung (polarity reversal) should be considered to…
A:
Q: d. е. f.
A:
Q: Circle the most stable cation. Place a box around the least stable cation. N. me
A:
Q: (Chapter 9) Write all of the resonance contributors for the following molecule. If the various…
A: The resonance structure of a compound is formed when the lone pair of electrons present on an atom…
Q: Step 2: When a curved arrow starts from a lone pair and points to an atom, a new sigma bond will…
A:
Q: What is the significance of electrons to chemical bonds?
A: Answer Electron play a major significant role in a chemical bond…
Q: Curved arrows are used to illustrate the flow of electrons. Using the provided resonance structures,…
A:
Q: HO. но HO
A:
Q: Match each compound on the left to its configuration on the right. If an arrow is given, answer only…
A: If the more priority group are above the plane and least priority group below the plain then if…
Q: Why is the circled compound more reactive for Sn1?
A: Why compound (a) is more reactive for SN1 is to be explained.
Q: A student argues that the two nitrogens in the compound below are sp² hybridized, but atom a is…
A: The basicity of the atom can be predicted by the donation of the electron pair. The electron…
Q: Use curved arrows to show the movement of electrons in each equation.
A: Please find below the reaction mechanism for reaction 1. Since in the above reaction we have free…
Q: I need to know the names of these two structurers for my notes but I am not sure how to name them.
A: The name of the given structure can be written as,
Q: Which of the following two molecules is more stable?
A:
Q: e) Circle the most basic nitrogen atom in the compound below. NH2
A: Since you have asked multiple question, we will solve the first question for you. If youwant any…
Q: Why is my second drawing wrong?
A: In this question, Firt we will draw sp3 neutral intermidiate then ester product drawn after removing…
Q: my answer to number 4 is D but I need help with my explanation.
A: Since reaction goes through carbocation as intermediate therefore that compound which forms the…
Q: Which arrow (x, y, or z) points toward the most acidic proton in the molecule to the right? (Arrows…
A: For acidity we see stability of anion formed when H^+ is removed .hence first of all we see…
Q: p-bromophenol Draw the molecule on the canvas by choosing buttons from the Tools (for bonds), Atoms,…
A: Draw structure of p-bromophenol
Q: 1. How many π-BMO and π-ABMO does this compound have ? 2. How many π electrons does the molecule…
A: In cyclopentadienyl cation, the five carbon atoms of the ring contain five unhybridized p-orbital.…
Q: The following compound is a:
A:
Can someone help me with this question and let me know if I am doing this correctly, please.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- a model of each molecule shown above: Is the molecule in the left box the same moleculeas the molecule in the right box? Use your models to answer the question, and recall that...4)On the following molecules, draw the curved arrows to convert the left-hand resonance structure form to the right-hand resonance structure formCircle the molecules that are most stable.
- Break compound A into several small (less or equals to 6 carbon atom) molecules. You need to explain the reasons why you choose to break those bonds.circle most polar carbon and give partial charges using d- and d+ notationAdd the unshared (lone) electron pairs to the structure in the drawing window below.

