Q: What ig the heat chamge. of corbon reacts itth Og according tothe following equatim? whem 4.729 ct…
A:
Q: A sample of 10.2 liters of an ideal gas at 23.0 °C and 750.5 torr is compressed and heated so that…
A:
Q: VIII. How would you prepare the following compounds from 2-phenylethanol? be required. (a) Styrene…
A:
Q: Chlorine in wastewater could be digested, isolated and converted to HCl and could be titrated with…
A: Wate water contains many compounds which contaminate it. But these can be removed by many chemical…
Q: Calculate the volume of water (in mL) that must be added to 50.9 mL of a 7.53 mol/L Naclo3(ag)…
A: Here we will use the formula M1 V1 = M2 V2 Where M1 and M2 is the initial and the final…
Q: A monoprotic weak acid, HA (K, = 1.78 × 104), was titrated against NaOH. A 50.00-mL of 0.04870 M HA…
A: Given: Ka of HA = 1.78×10-4 Concentration of HA (M1) = 0.04870 Volume of HA (V1) = 50.00 mL…
Q: 2) What is the heat change when 1.48 g of Chlorine reacts with excess phosphorus according to the…
A:
Q: 1. NaOH is added to a 500mL of 2M acetic acid. If the pKa value of acetic acid is approximately…
A:
Q: Given the following values, what is the concentration of the acid? (With solution) Initial Volume…
A: Given: Initial volume of acid = 10.150 mL Final volume of acid = 11.620 mL Initial volume of NaOH =…
Q: 8. For the mass spec shown below, select the structure that matches. How can you tell? MASS SPECTRUM…
A:
Q: 4. Deduce the structure for Compound Y which has a molecular formula CH,CIO; given its "HNMR and…
A:
Q: A lab technician wants to make 500 mL of a 2.50 mol/L solution from a standard solution of…
A: Preparation of 500 mL of a 2.50 mol/L solution from a standard solution of concentratedsulfuric…
Q: Ka = 1.8 × 10–5 for the ionization of HC2H3O2(aq). Determine the [HC2H3O2]/[C2H3O2–] ratio that will…
A:
Q: What is the maximum number of grams of PHg that can be formed when 47.31 g of phosphorous react with…
A: Given-> Weight of Phosphorus (P4) = 47.31 gm Weight of hydrogen (H2) = 18.96 gm
Q: A coffee-cup calorimeter contains 70 mL of water with an initial temperature of 24.9°C. An unknown…
A: The heat absorbed (q) by the water can be obtained by the multiplication of mass (m) of water with…
Q: (8) Discuss the three main processes of interactions of electrons with matter.
A: Charged particles interact with matter via elastic and inelastic collisions with atomic electrons…
Q: The solubility of KNO3 at 25°C is 80 g KNO3 / 100g H2O. [ Select ] solution a) A solution of 160g…
A: An unsaturated solution is solution, where the solute is present in lesser amount than it can…
Q: In a chromatographic analysis of an unknown mixture, compound A has a retention time of 5.56 minutes…
A:
Q: coffee-cup calorimeter contains 30 mL of water with an initial temperature of 24.8°C. An unknown…
A:
Q: Calculate the molarity and the mass-volume percent solution obtained by dissolving 107.9g H3PO4 into…
A: Given, mass of H3PO4 = 107.9 g Volume of the solution = 649 mL Molarity of solution = ? Mass-volume…
Q: 8.37) An electron in the first excited state of hydrogen is struck by a photon of light of…
A: Given, Wavelength of photon = λ = 656.48 nm a). What is the frequency of the photon ? b). What is…
Q: Complete and Balance The following equation, in the case that the reaction doesnt occur write "NR"…
A: Given unbalanced and incomplete reaction is : MnO4- + OH- + SO32- --------------> ? Complete…
Q: 50 ml of .2M KNO3 from 4.0 M stock solution
A: Given: Volume of KNO3 solution = 50 mL Molarity of KNO3 solution = 0.2 M Molarity of stock solution…
Q: write out the equilibrium expression for the mineral dissolution of Al(OH)3 in water
A:
Q: D H. I. OH -Br DMSO H3C CH3 HOCH,CH,CH, H3C 'CH3 II.
A:
Q: Why is complete clotting necessary during serum preparation?
A: Serum : It is the part of bodily fluid which is clear in appearance, for blood it does not include…
Q: Question 8 Which titrimetry is refrerred to by the following v Uses permangante as its Titrant A.…
A:
Q: What's More Activity 3.2 Identify and underline the amphoteric substances in this chart. Conjugate,…
A: Given chart is : Identify and underline the amphoteric substances in this chart = ?
Q: 6. Show ALL products that you would expect from the reaction of l-methyleyclopentene with NBS? Show…
A:
Q: ow solution was used as a titrant to determine the concentration of Cr' when it reached the…
A: The end point in the titration can be defined as the point that is determined by the change in…
Q: In the Iron REDOX titration, the specific which specific indicator \ produces soluble red-colored…
A: In the iron redox titration the thiocyanate when used as an indicator gives a soluble red coloured…
Q: Chemical Equation: NO2 + H2O → HNO2 + HNO3 1. Compute for the ΔH° rxn of the equation:…
A:
Q: Using the table of the weak base below, you have chosen Methylamine as your weak base in the buffer…
A: Kb is the dissociation constant of a base. Calculations for pKb have been done in the following…
Q: how many milliliters of 2.00 M h2so4 will react with 28.0g of sodium hydroxide?
A:
Q: Two aliquot of a water sample is titrated against a strong acid using phenolphthalein and…
A: Introduction : Phenolphthalein is used as an indicator it turns colourless into acidic medium and…
Q: Why must solutions with high concentrations be diluted prior to analysis via Beer's Law? A. The…
A: Answer= option 1 (the relation between absorbance and concentration not linear at high…
Q: CN A. 4 Ph. A, 5 6
A:
Q: Predict the reactants or products of the following reactions, and balance the equations.
A: Balanced chemical equation, it can be define every reaction in which number of atoms of all the…
Q: contains 20 mL of water with an initial temperatu na mass of 1.388 grams was placed inside maki…
A: Given, Volume of water = 20 mL Density = 1.0 g/mL Mass of water = Volume × density = 20 ×1.0 =…
Q: are intermediate in length as is common in aromatics, the C-B bonds are very long - they are the…
A: Bond length is average distance of two bonded nuclei.
Q: 1. Balance this equation, MnO2(s) + Cl-(aq) —> Mn2+(aq) + Cl2(g) using the half-reaction method.…
A: In acidic medium, oxygen atoms are balanced by adding H2O molecules to the side which is deficient…
Q: Draw the skeletal formula of the following: 1.) Isobutyraldehyde 2.) α-Ethyl-γ methoxycaproaldehyde…
A:
Q: 8. LIOH (aq) CuCro, (aq) →
A: Complete and balanced the given reaction-- LiOH(aq) + CuCrO4(aq) ----> ? + ?
Q: 3. Which do itself? Why? guhic Chemistry an aromatic ion precursor? 9-bromocyclononatetraene or…
A: The reactants given are 9-bromocyclononatetraene and cyclononatetraene.
Q: What is the OH⁻ concentration in this solution 1.05 M C₅H₅N?
A:
Q: 1. If the Ka of an acid is 1.8x10 ^-5, calculate the pKa and pKb values of the acid.
A: Since you have asked multiple type questions, we will solve only first questions for you. If you…
Q: In a 0.24M aqueous solution of acetic acid (CH3CO2H), what is the percentage of acidic acid that is…
A: percentage of acetic acid dissociated is to be calculated
Q: Questions: Calculate the heat gained or lost when 17.0g of water cool from 80.0°C to 25.0°C?…
A:
Q: 1. Propose a fragmentation pattern for the following compound. CH=C(CH,JCH,CH,OH m/z = 71, 68, 41,…
A:
Q: 100 a. 43 80 60 40 20 86 58 71 20 40 60 80 100 120 100 b. 57 80 60 29 40 86 20 20 40 60 80 100 120…
A:
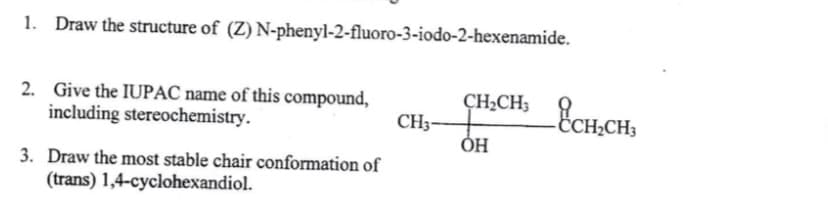
Step by step
Solved in 2 steps with 1 images

- 1- Chloro-1,2-diphenyletane can undergo E2 elimination to give either cis- or trans-1,2-diphenylethylene (stilbene). Draw New man projections of the reactive conformations leading to both possible products, and suggest a reason why trans alkene is the major product?By considering the stereochemical requirements for E2-elimination, and using an appropriate illustration need help indicating whether the more stable conformer (conformer B) of trans-1 can undergo E2 elimination. Thank you :)Write the structure of the compound E,E-2,4-Hexadien-1-ol and label each non-equivalent carbon with a letter, A,B,C..
- Stretch the structure for (S)-3-bromo-2-methylpentane and add the stereochemistry at all stereogenic centers.The compound 3,4-dimethyl-hexan-3-ol of 3R, 4S configuration is treated with a concentrated HBr solution at room temperature. A mixture of two stereoisomers is obtained.If the reaction mixture above is heated, the appearance of several other compounds is observed. 1) Draw the different compounds obtained using the wedge-flywheel representation. 2) What is the majority product? Explain 3) Propose a modification of the experimental conditions in order to obtain the exclusive formation of these compounds obtained after heatingBenzene is one of the compounds used as octane enhancers in unleaded gasoline. It is manufactured by thecatalytic conversion of acetylene to benzene: 3C2 H2(g) ⇌ C6 H6(g). Which value of Kc would make this reactionmost useful commercially? Kc ≈ 0.01, Kc ≈ 1, or Kc ≈ 10. Explain your answer
- On the line drawn below, provide the systematic (IUPAC) name for the following structure. Be sure to include stereochemistry (R/S, cis-trans, or E/Z) when appropriate.A difficult problem in the synthesis of PGF2α is the introduction of the OH group at C15 in the desired configuration. a. Label this stereogenic center as R or S. b. A well known synthesis of PGF2α involves reaction of A with Zn(BH4)2, a metal hydride reagent similar in reactivity to NaBH4, to form two isomeric products, B and C. Draw their structures and indicate their stereochemical relationship. c. Suggest a reagent to convert A to the single stereoisomer X.A difficult problem in the synthesis of PGF2α is the introduction of the OH group at C15 in the desired conguration.a. Label this stereogenic center as R or S.b. A well known synthesis of PGF2α involves reaction of A with Zn(BH4)2, a metal hydride reagent similar in reactivity to NaBH4, to form two isomeric products, B and C. Draw their structures and indicate their stereochemical relationship.c. Suggest a reagent to convert A to the single stereoisomer X.
- What organic product is formed when 1‑methylcyclopentene is treated with NMMO in the presence of H2O and a catalytic amound of OsO4? Clearly show stereochemistry by drawing a wedge and dashed bond for each chiral carbon. Only draw one stereoisomer if more than one can be formed.Stereoisomers of α-bromocinnamic acid: draw the reaction mechiansim with arows for the dehydrobromination of 2,3-dibromo-3-phenylpropanoic acid with ethanol and KOH to produce (Z)-α-bromocinnamic acid and (E)-α-bromocinnamic acid. Also draw the transition state for The (Z)-isomer syn-periplanar transition state and anti-periplanar transition state of the (E)-isomer.Identify A, B, and C, intermediates in the synthesis of the ve-membered ring called an α-methylene-γ-butyrolactone. This heterocyclic ring system is present in some antitumor agents.

