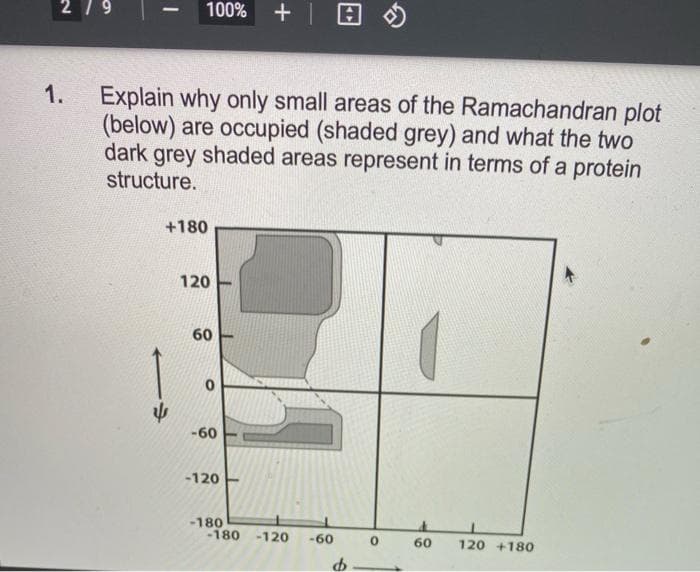
1. Explain why only small areas of the Ramachandran plot (below) are occupied (shaded grey) and what the two dark grey shaded areas represent in terms of a protein structure. +180 120 60 -60 -120 -180 -180 -120 -60 60 120 +180
Q: 20. Which of the following does NOT contribute to the tertiary structure of proteins? a. salt…
A: Proteins are made of polypeptide chain/(s), which are polymers of amino acids. Adjacent amino acids…
Q: (5) You have discovered a brand-new protein and need to determine its tertiary (and possibly…
A: Proteins are composed of polypeptide chain that can shows different three dimensional structures.…
Q: Slightly heating a protein can render it nonfunctional, but a polysaccharide requires a boiling…
A: Proteins and polysaccharides Proteins are molecules composed of adjoining amino acids by a peptide…
Q: Variations in three-dimensional shapes among proteins are due tobonding between thea. amino…
A: Proteins are defined as macromolecules or large biomolecules composed of one or more long chains of…
Q: Beta-mercaptoethanol (BME) disrupts the types of interactions responsible for ____ structure of…
A: Beta - mercaptoethanol (BME) disrupts the disulphide bond found in the protein. Hence the level of…
Q: 1. What is the charge of the primary structure of proteins at pH = 2.0, 4.0,6.0 and 11.0 of the…
A: We are authorized to answer one question at a time since you have not mentioned which question you…
Q: Draw the structure of the following polypeptide at physiological pH. You do not need to indicate the…
A: Protein is composed of various amino acids in various proportions, which determine the structure…
Q: 1) What are the four levels of protein folding. How do you distinguish those different levels? What…
A: The proteins are macromolecules that have important functions in the biological activity. The amino…
Q: All the necessary information to form the three-dimensional structure of the protein is provided by:…
A: Proteins are formed by amino acid residues. These amino acid residues are held together by peptide…
Q: 19.40 Indicate whether each of the following statements describes the primary, secondary, tertiary,…
A: We are authorized to answer one question at a time since you have not mentioned which question you…
Q: 1) Lysine is one of the 20 amino acids that found in protein. Draw the form (s) of lysine that would…
A: We are authorized to answer one question at a time since you have not mentioned which question you…
Q: 29.) A protein with quaternary structure described as C2B2Y2 would be a B) heteropentamer A)…
A: Biomolecules are organic molecules that function inside living organisms. They can be divided into…
Q: SDS-PAGE gels are used to study changes in protein primary structure. Which of the following…
A: Option B is correct.
Q: Which of the following is incorrect? a. A heterodimer is composed by two identical polypeptide…
A: a) The heterodimer is composed of two different subunits, they are not made up of two identical…
Q: Explain what is meant by “The Central Dogma”? In your explanation, describe the structure and…
A: Central Dogma of Molecular Biology explain the flow of information from DNA to proteins.
Q: exce PH pea excess [OH] Make a reasonable suggestion for which А. amino acid X might be, with either…
A: Amino acids are the composed of an alpha carbon bonded to 4 different groups. These groups are a…
Q: 5. Tertiary structure of proteins. Their classification is relative to a given structure. Forces and…
A: Amino acids are biomolecules that are comprised of two functional groups, these are an amino group…
Q: The illustration shows the interactions that form the tertiary structure of a protein segment.…
A: The term protein is derived from a Greek word that means of first importance. They are the most…
Q: Look at the amino acid composition of a protein given in the table and click on the area in the 2D…
A: the isoelectric point of an amino acid is the pH at which the net electric charge of that amino acid…
Q: In X-ray crystąllography, the wavelength of an X-ray is - 1.5 Å. This is useful in determining a…
A: X-ray crystallography is a technique used in the determination of three-dimensional atomic…
Q: In an ESI spectrum, you see a peak at m/z = 1293.51. You know that peak corresponds to a charge of…
A: The m/z is the mass to charge ratio. m/z= 1293.51 the charge is +10.
Q: Look at the amino acid composition of a protein given in the table and click on the area in the 2D…
A: the isoelectric point of an amino acid is the pH at which the net electric charge of that amino acid…
Q: An intrinsically disordered protein (IDP) is a protein that lacks a fixed three- dimensional…
A: Given; Freely joined chain length of a single amino acid = 3Å Total number of constituent amino…
Q: 6.All of the following types of interactions cooperate in stabilizing the tertiary structures of…
A: Tertiary structure of protein is three dimensional structure of protein which is formed by single…
Q: Fill in the blank: Identify the type of non-covalent/covalent interactions present in the following…
A: The Ramachandran plot also termed as Rama plot is the way to visualize the position of backbone…
Q: Proteins frequently form complexes in which 2, 3, 4, or even more individual proteins (“monomers”)…
A: The osmotic pressure is the pressure required to stop the flow across a semi-permeable membrane at a…
Q: 1. Draw the structural formula of the amino acid valine that predominates in solution at each of the…
A: Hi! Thank you for the questions. As you have posted multiple questions, I will be answering the…
Q: 1. Are small biomolecules that are solubilized by the Cl atoms present: a. CH3(CH2)10CH(OH)2 b.…
A: ''Since you have asked multiple questions, we will solve the "Second question" for you as per our…
Q: You have a soluble protein that is highly flexible and is only 23 kDa in size. What is the most…
A: Proteins are the polymers of amino acids which are arranged in the three-dimensional structure of…
Q: 5 (a) Describe in detail how you will determine the primary structure of protein. You have been…
A: The primary structure of a protein is generally referred to the sequence of amino acids in the…
Q: 7. Draw and give the full names of the amino acids in the following dipeptides.
A: An amino acid is made up of two functional groups namely carboxyl and amino group. A dipeptide is…
Q: Describe the 4 levels of protein structure: primary, secondary, tertiary and quaternary. And, note…
A: Biomolecules are organic molecules made up of mainly carbon and hydrogen, but other elements are…
Q: 3. Which of the following processes involves the loss of a protein’s native structure?…
A: Proteins are polypeptides made up of amino acids. The biochemically and structurally stable form of…
Q: 2. Why would a protein have an apparent size of 40 kDa (kiloDaltons) at pH 6.5 but when the protein…
A: Proteins are the linear polymers of amino acids attached together via peptide bonds. There are…
Q: e configuration of a protein molecule? Provide an exam
A: Protein characteristics are determined not only by the sequence of amino acid residues in…
Q: 2. a. In the following diagram of a portion of a protein, label the types of interactions that are…
A: The complete structure of protein can be described at four different levels of complexity -…
Q: 5. You need the following standard protein concentrations: 4mg/mL, 1mg/mL, 0.25mg/mL, and…
A: Proteins are nitrogenous organic macromolecules that are essential for the proper functioning of…
Q: Sickle cell anemia results from a substitution of a valine for a glutamic acid. What do you expect…
A: Sickle cell anemia: Normal red cells are round like doughnuts shape, but in sickle cell anemia, it…
Q: The 3 structure of a protein incorporates any 2 structure the protein has, but results primarily…
A: A protein undergoes many types of interaction in order to stabilize itself. These interactions are…
Q: 3. A. As the bulk solvent in biological systems, water exerts a huge effect on protein structure and…
A: Proteins are the building blocks of life. Proteins are made up of amino acids. Amino acids when they…
Q: Describe in detail how to detect the primary structure of protein.
A: Hello! Since you have posted multiple questions, we are answering only first question. If you need…
Q: 2. A. Draw the tri-peptide Phe-Gln-Ser at neutral pH, given the amino acids below. B. Classify each…
A: There are many biomolecules are present in the body. Carbohydrates, lipids, proteins, nucleic acids…
Q: Using the Chou-Fasman rules (without calculating numerical probability) predict the secondary…
A: Chou fasman rules is a method to predict the secondary structural in polypeptide sequence. It is…
Q: Describe in detail how to determine the primary structure of protein.
A: According to guidelines we have to answer the first question. So, please kindly post the remaining…
Q: 7. Convert the following Haworth projections to a Fischer projection. Name the monosaccharides you…
A: Carbohydrates are the polyhydroxy ketones or polyhydroxy aldehydes or their derivatives. In…
Q: Chicken egg contains a lot of proteins which supports the growth and development of the chicken…
A: Proteins are composed of twenty standard amino acids attached together via peptide bonds. The linear…
Q: 1. The hydropathic index represents the sum of hydrophobic and hydrophilic residues of a…
A: Proteins are one among the 4 macromolecules that makes up all forms of cellular life. Proteins are…
Q: 3. One of the 20 standard amino acids is unusual in that its side chain contains a ring that…
A: Amino acids are the building blocks of the protein. There are 20 standard amino acids which is used…
Step by step
Solved in 2 steps with 1 images

- Which of these nH measurements is indicative of a partially positively cooperative protein? -1 0 1 2A gel filtration column with a fractionation range of 1.5-20 kDa is used to separate out the proteins shown below. If these proteins are collected into separate fractions in between the void volume and total volume, in which order will they elute? Indicate if any of the proteins are found in the void volume or total volume fractions. Protein Z - 3330Da Protein Y - 13kDa Protein X - 1.3kDa I. Total volume fraction II. Third protein fraction III. Second protein fraction IV. First protein fraction V. Void volume fractionTo which of the proteins does peak 4 correspond to? PLEASE EXPLAIN WHY A. A B. B C. C D. D E. B and C
- Two proteins of different molecular weight (mw) are mixed and need to be separated. When eluted through a Sephadex-G50 chromatography column, what is the order of elution (first followed by second)? A. protein with mw of 40 kDa; protein with mw of 20 kDa B. both proteins will elute at the same time C. both proteins will not elute as the combined mw is larger than 50 kDa D. protein with mw of 20 kDa; protein with mw of 40 kDThe limit for G-200 beads is 5000-600,000. When you pass two proteins- Protein A (75, 000), Protein B (1,000,000), through these beads, the protein that will elute out last is – They both elute out at the same time. Protein B Protein AHOW MANY of the following five items represent modes of protein denaturation? 1.) heating a protein sample 2.) cooling a protein sample (slowly) 3.) putting a protein sample in a saltier environment 4.) eating a protein sample (such as with a whisk) 5.) adding acid to a protein sample
- will UPVOTE Kindly answer the question. 1. Do you think free amino acids will give a positive result with the biuret test? Explain why.In order to prepare a standard curve, you will do serial dilutions of the standard protein. Assume the protein concentration in tube A is 3 mg/ml and you take 0.5ml and add it to the next tube (tube B) containing 0.5ml of buffer. What is the concentration in tube B?Why would your protein measurement not match to package label? Let us disregard the idea that the package is wrong. Provide one plausible explanation. Use several sentences to outline your reasoning.
- Give only typing answer with explanation and conclusion You want to make 94 µL of the diluted Cell-Free extract for estimation of total protein. To do this, how much water will you add to the correct volume of the undiluted cell-free extract? Note: Cell-free extracts will need to be diluted 1:25 in water.Find a method that uses some form of HPLC for the analysis of proteins. What was the stationary phase used? How does this kind of stationary phase separate the proteins? What kind of mobile phase was used? Was the method isocratic or was a gradient used? How were the proteins detected?The following protein quantification utilizes Cu ions except fora. Lowryb. Biuretc.Bradford

