Q: 2-aminophenol hydrochloride is pictured at right. As is, the acid is fully protonated, the H from…
A: 2-aminophenol hydrochloride, HO-C6H4-NH3+Cl- is a diprotic acid with pka values of 4.70 and 9.97.…
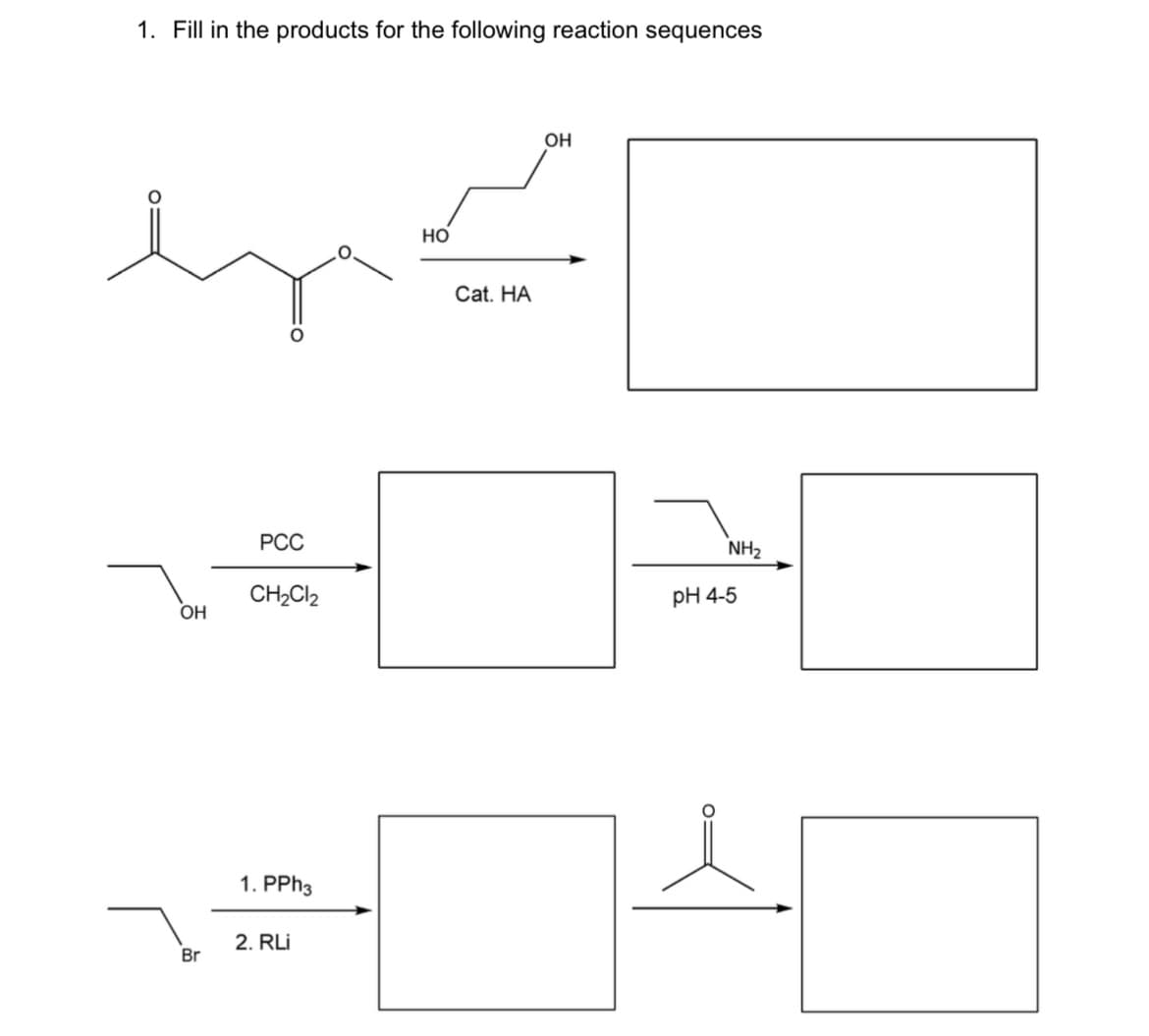
Q: Starting from ethane, provide a synthesis of the compound below showing all the reagents necessary…
A:
Q: 1.00 M NaOCN solution. Ka for HOCN is 3.3 x 10-4.
A:
Q: Consider the following hydroboration/oxidation of an alkene. OH 1. BH3, THF -CH3 2. HаОг, NaOH, H-О…
A: The intermediate formed in the given reaction = ?
Q: 2. Find the oxidation number of each kind of atom in the following: a. P2Os b. NH4 c. NazCr2O,
A:
Q: Learning Task 5: What is the mole fraction of sodium chloride and water in a solution, prepared by…
A:
Q: The combustion of propane (C3H8) produces CO2 and H2O: C3H8(g) + 5O2 (g) --->…
A: Given that Moles of C3H8 = 4.6 mol Moles of O2 = 2.5 mol
Q: Chemistry I. Draw the structure of the main organic product of the following reactions. Show the…
A: First question may be something wrong so i am working on remaining questions.
Q: explain and find how to get the derivative of benzil. Solve it asap
A:
Q: A reaction requires the addition of 350.0 mL of a solution with a pH of 3.50. What mass of HNO3 (in…
A: pH :- The negative logarithm to the base 10 of hydrogen ion concentration of a solution expressed…
Q: CONTENT OF IRON IN DRINKING WATER IS NORMALIZED FROM 1. kind of source of water supply 2. toxic…
A:
Q: 2NO(g) + Cl2(g) = NOCl2(g) NOCl2 + NO(g) = 2NOCl(g) If this mechanism is correct, what can be…
A: Chemical kinetics is branch of chemistry in which we deal with speed at which rate of reaction…
Q: determine the ph of a 0.5 M solution of HCL
A: The answer is as follows:
Q: 3- Write the fragmentation pattern of the following compounds: a- CH,CH,COOCH,CH,CH, ÇH2Br b-
A:
Q: Calculate the AS (J/K) for the phase transition of a 0.50 mol water described below: H2O(s), O°C –…
A: It will undergo through 3 different steps,we need to calculate entropy change of each steps.
Q: c) making the polymer involves coolln d) they are only made in labs (CH,CH,),CuLi CI b) CH3 NH…
A:
Q: oues Amines cwith moretham six carbon ame Solublein. O Water D aqueous Hes O aqueaus NaHog…
A: Here we have to predict the solubility of amine having more than six carbon atoms in different…
Q: ) Calcium hydride (CaH2) reacts with water to form hydrogen gas: CaH2 (s) + 2H2O (I) ---> Ca(OH)2…
A:
Q: 6) A certain reaction is first order, and 5 s after initiation of the reaction, 10 % of the reactant…
A: Order of the reaction determine experimentally. The time required to consume half of the initial…
Q: A 2000 mL solution containing 4.0 g of MgSO4 hydrate and 5.0 g of NaCl must be made isotonic using…
A: All the calculations are done using Freezing point depression method and the answer is 42.6 grams of…
Q: Name and draw the major product(s) of electrophilic chlorination of the following compounds: a)…
A:
Q: What is the mass number of an isotope with an atomic number of 57 and that has 72 neutrons?
A:
Q: 2-aminophenol hydrochloride is pictured at right. As is, the acid is fully protonated, the H from…
A: 2-aminophenol hydrochloride, HO-C6H4-NH3+Cl- is a diprotic acid with pka values of pKa1 = 4.70 and…
Q: Which of the following is a zwitterion?
A: Zwitterion structure: Zwitterion it is a dipolar ion Amino group which contains a positive group…
Q: 1.) A gas originally at 27oC and 1.0 atm pressure in a 3.3 L flask is cooled at constant pressure…
A: For a definite amount of gas, at constant pressure, the volume of the gas increases with an increase…
Q: For a chemical reaction that is spontaneous at standard conditions, which of the following holds?…
A:
Q: At the same temperature, compared to 0.050 M NaCl, a 0.10 M glucose (C6H1206) solution is O…
A: NaCl undergo dissociation . And osmotic pressure is a colligative property which depends upon the…
Q: answer the question 2NO N2O2 fast N2O2 + H2 N2O + H2O slow N2O + H2 N2 + H2O slow The…
A: Moleculity is the number of reacting molecules which collides to each other to form product.
Q: lodine molecules react reversibly with iodine ions to produce triiodide ions. I2(aq) + l'(aq) I5…
A:
Q: 5. A solution is prepared by adding 1.00 x 1013 mole of strong base NaOH to 1.00 L of aqueous…
A: In this question we have to tell the pH of the given solution.
Q: K eq for the following Write the formula for the reaction: 2NH, () +3H28) N26) 3 (g) edio
A: Keq is the equillibrium constant. It gives the relation between concentration of product and…
Q: What is the molarity of the chlorate anion in solution after 42.00 mL of 3.000 M NAOH is added to…
A:
Q: Your body is: O a. an open system Ob. a closed system an isolated system
A: Open system allow transfer of mass and energy both from system to its surrounding. Closed system…
Q: If you have 2.0 grams of a radioactive substance left after 5 half-lives, how much did you start…
A:
Q: What is K for a reaction if ∆G° =-64.2 kJ/mol at 25.0 °C? (R = 8.314 J/mol ・ K)
A:
Q: A 3.82 g sample of magnesium nitride is reacted with 7.73 g of water:…
A:
Q: 3 P Question 41 Consider these reactions: Reaction 1: H2 (g) + Cl2 (9) 2HCI(g) AH = -184.6 kJ…
A:
Q: Exposure to arsenic can result to subacute to severe toxicity and can sometimes be lethal and cause…
A: The question is based on the concept of gravimetric analysis. We have to calculate percentage of…
Q: 1. Name the following molecules: a) CH b) CCCH(CH,)CH, H,CHO CH CH CH- -CH 2. Draw the following…
A:
Q: Calculate ∆U, w, q (in kJ) and ∆S (in J/K) when one mole of an ideal monatomic gas at 25°C is…
A: Given : Temperature = 25 °C Initial volume = 10 L Final volume = 40 L Number of moles = 1 mole
Q: Neutral carbon has six electrons. In its ground state, which of the following shells will be fully…
A: Given, Neutral carbon has six electrons. In its ground state, which of the following shells will be…
Q: Question 16 For the compound Nome the compond Identify ench chiral center Are geo metric is omors…
A:
Q: Can someone explain which carbon the stereochemistry is inverted and how to arrive at the specific…
A: A question based on introduction to organic chemistry that is to be accomplished.
Q: Chemistry Complete Compound 1 IR Data and name the component (name and structure). Molar mass is…
A: A question based on IR spectroscopy that is to be accomplished.
Q: S) Show the steps necessary to make 4-methylhexanoic acid using a malonic ester synthesis.
A: To synthesis a Product 4-methylhexanoic acid usic malonic ester as starting material ....
Q: Can solid benzene exist on the surface of the planet Vulcan (surface
A: Can solid benzene exist on the surface of the planet Vulcan (surface temperature 270 K and…
Q: Give the plan for the next retrosynthesis. Other compounds may be used. All reagents must be…
A: To get the product from the starting compound, need the following reactions 1) Dehydration : Removal…
Q: When 3.22 g of an unknown compound composed of the elements C, H and O is dissolved in 30.0 g of…
A:
Q: er 2 800 705 kJ 600 497 kJ PE (kJ) 400 200 175 kJ 100 Reaction Progress Ceated by E Lee for Virtual…
A:
Q: s) Name the following compounds properly. a) H3C. NH 0-CH3 b) H3C CH3 elah H3C-
A:
Step by step
Solved in 2 steps with 2 images

- Draw the structure of fulvic acid chelated with Pb²+ at pH 8.0.please explain why this happends, im having trouble in this section. Part A Chymotrypsin, an enzyme that hydrolyzes peptide bonds in proteins, functions in the small intestine at an optimum pH of 7.7 to 8.0. How is the rate of the chymotrypsin-catalyzed reaction affected by each of the following conditions?The BOD rate constant was found to be 0.151 day^-1 (base e) at 28.5 Celsius. What is the rate constant day^-1 (base e) at 6.0 Celsius?
- When some sugars dissolve in water they spontaneously undergo changes in optical rotation called mutarrotation. The Mutarrotation of D-glucopyranose is catalyzed by acid and bases. 2-Hydroxypyridine is a more effective catalyst than phenol and pyridine for this reaction because: a.Both oxygen and N in 2-hydroxypyridine act as bases increasing the rapid interconversion of sugar b. The OH of 2-hydroxypyridine serves as the base while the current N as the acid. c. 2-hydroxypyridine acts both as a base to remove the proton from the hydroxyl group in the hemiacetal and as an acid to provide a proton to the oxygen in the hemiacetal. d.Phenol and pyridine are very expensive.What is the product of the reaction sequence shown in Image 34?The rate constant for the uncatalyzed reaction of two molecules of glycine ethyl ester to form glycylglycine ethyl ester is 0.6 M- 1s - 1. In the presence of Co2 +, the rate constant is 1.5 * 106 M- 1s - 1. What rate enhancement does the catalyst provide?
- 10.2 Complete the following reactions: Show the step-by-step process. Do not use shortcut methods. Make it as detailed as it can be. Encode (not hand-written)!C3H6 + H2O + H2SO4 --> C3H8O + H2SO4 what is the catalyst in the reaction above?What is the product of the reaction P4O6 + H2O? A) H3PO4 B) H2PO4 C) H3PO3 D) H2PO3 E) H2P4O7
- Explain the mechanism of Maillard reaction in uht milk.(include graphics).This is the type of organic reaction wherein a single reactant splits into two products and is accompanied by the formation of a small molecule. addition substitution elimination condensation What is the name of the angle that a nucleophile approaches the carbon on the C=O bond? Borge-Dante Bürgi–Dunitz Baran-Daly Bunsen-DebyeOzonolysis, osmylation, and epoxide formation with mCPBA are all what type of reactions? a.) reduction b.)oxidation c) bothd) neither

