Q: Based on the following reaction, which way would the reaction shift to restore equilibrium due to…
A: Given reaction:- C(s) + 2H2(g) <==> CH4(g) ∆H = -74.6 KJ => As heat negative means it is…
Q: A student needs 15.0g f ethanol for an experiment. If the density of the alcohol is 0.789 g/mL, how…
A:
Q: What is the initial volume of a system that expanded to 85 L against pressure of 5.23 atm doing 25.0…
A: Given : final volume = 85 L Pressure = 5.23 atm Work done = 25kj Tip : workdone =…
Q: 1) 1/2 N2 (g) 6.96 x 10-16 2) 2 NO2 (g) 4.5 x 10-13 + 1/2 02 (g) NO 2 NO ← (g) Keg (g) + O2 (g) 3) 2…
A: The given equilibrium reaction is as follows: (1) 12N2(g) + 12O2(g) ⇌ NO(g)…
Q: A scientist mixes 0.02 g of a strong monoprotic base in 83 ml of water and obtains a pH of 12. He…
A: mass of strong monoprotic base = 0.02 g Volume of strong monoprotic base = 83 ml pH = 12
Q: Why is it essential to practice on how to prepare solutions (stock solution/dilutions)? How does…
A: The preparation of solutions is a fundamental concept that is essential in many scientific and…
Q: A 150.0 mL sample of 0.250 M NaOH is mixed with a 100.0 mL sample of 0.289 M HNO3 in a coffee cup…
A:
Q: 11. The equilibrium constant Kp for the following reaction is 0.403 at 1000 °C. FeO(s) + CO(g) =…
A: Answer :
Q: Figure 요 | -| 요 H N-H C :NH₂
A: Hybridization refers to the mixing of atomic orbitals to form new hybrid orbitals with different…
Q: The pressure on a 115 mL volume of gas is changed from 720 mmHg to 800 mmHg at constant temperature.…
A: At constant temperature: P1V1 = P2V2
Q: Which set of quantum numbers cannot be correct? A. n = 1, l = 1, ml = 1 B. n = 2, l…
A: for principal quantum number n, azimuthal quantum number (l) values can be integers from 0 to n-1.…
Q: . Each of the following molecules have a resonance structure which should be considered when…
A: We know that atomic orbitals mix together to form stable and lower energy orbitals called hybrized…
Q: a solution of sodium nitrate and potassium sulfate are mixed together, no reaction will occur. True…
A: We need to writIf a solution of sodium nitrate and potassium sulfate are mixed together, no reaction…
Q: If 4.67mL of water is added to 7.92mL of an 0.111M aqueous Ni2+ solution, by what percentage would…
A: Absorbance is defined as the negative log of transmittance. Absorbance can also be calculated by…
Q: 2. Nitric acid, which is among the top 15 chemicals produced in the United States, was first…
A: 2. Recall the given reaction, NaNO3 s + H2SO4 l → NaHSO4 s + HNO3…
Q: Which of the following amino acids contain nonpolar side chains? Select all that apply. A. Serine B.…
A: Amino acids are the building blocks of proteins and have unique properties that contribute to the…
Q: 2. A chemical reaction is temperature sensitive, as shown below. AB+C Temp °C 2884 10 20 30 40 Keq…
A: ll
Q: What product(s) are formed in the acetylation of DL-phenylalanine? ОН А. 128 хо В. О NH 1 с. D. 00…
A: •Here Given acetylation of DL- phenylalanine. •We have to predict product of reaction.
Q: Part A Dinitrogen tetroxide decomposes to form nitrogen dioxide according to the following reaction:…
A: Given : equilibrium reaction Tip : if Qp > Kp then recation shift toward reactant if Qp < Kp…
Q: Given the following information: Br2 (g) + Cl₂ (g) →>>> 2 BrCl (g) Keq = 23.8 at 250 °C What would…
A: Given : equilibrium reaction
Q: A. Reaction Rate Data: Mixture Vol.4.0M Vol Vol acetone 1.0M (mL) HCI (mL) 1 2 3 4 1 2 3 4 m = n= P=…
A: The order of a reaction refers to the power to which the concentration of a reactant is raised in…
Q: According to the nutritional information on a package of jasmine rice, each serving (1/4 cup or 45.0…
A: According to the nutritional information on a package of jasmine rice, each serving (1/4 cup or 45.0…
Q: What is the isoelectric point value of His-Arg at Ph 7? Then at Ph 12 and pH 5?
A: We need to find What is the isoelectric point value of His-Arg at Ph 7 and Then at Ph 12 and pH 5
Q: The reaction of an aqueous solution containing 0.425 M oxalic acid (H₂C2O4), 4.15 M fluoride ions,…
A: Given : equilibrium reaction Tip : if Q is less than Keq then reaction will shift to product side…
Q: 1. Calculate the AH°rxn for the following reaction. (AH°f [SiO2(s)] = -910.9 kJ/mol; AH°f [SiC14(g)]…
A: Given: SiO2(s)+4HCl(g)→SiCl4(g)+2H2O(g) ∆Hfo[SiO2(s)]=-910.9…
Q: Why do ionic and polar covalent substances conduct electricity and nonpolar molecules are not…
A: Ionic and polar covalent substances conduct electricity because they contain ions or polar molecules…
Q: Show how you would synthesize each tertiary alcohol by adding an appropriate Grignard reagent to a…
A: Show how you would synthesize each tertiary alcohol by adding an appropriate Grignard reagent to a…
Q: A pharmacist mixes 100 mL of 38% w/w hydrochloric acid with enough purified water to make 360 mL. If…
A: A pharmacist mixes 100 mL of 38% w/w hydrochloric acid with enough purified water to make 360 mL. If…
Q: A 275-g sample of nickel at 100.0°C is placed in 100.0 mL of water at 22.0°C. What is the final…
A: According to the question, Given,Mass of Nickel (m) = 275 g Temperature of Nickel (TNi) = 100…
Q: The solubility of CaCO3 is pll dependent (Kal (H₂CO3) -4.3 x 10-7, K₁2 (H₂CO₂) -5.6 x 10-¹¹.) Use…
A: The solubility product constant, also known as Ksp, is a measure of the saturation of a solid…
Q: Relative Intensity 120 100 80 40 20 0 15 178 20 Lipu 30042 40 55 sense 00 71 7805 60 2-Propenoic…
A: Mass spectrometry is one of the best techniques for analyzing the structure of a molecule and…
Q: Grams of KHP 0.313 0.3179 0.3159 Moles of KHP (Equivalent to Mols NaOH) Millimeter NaOH used to…
A: ♧ The chemical reaction between KHP and NaOH takes place in the following way -…
Q: Place the following three choices in order of decreasing vapor pressure. CCl 4 C 5H 10 CBr 4
A: Vapour pressure is the measure of the tendency of a material to change into gaseous or vapour state.…
Q: Place the following three choices in order of increasing melting point. Xe Kr Ar
A:
Q: A sample of Cl2 gas has a volume of 13L, how much does it weigh? Round to 2 decimal points
A:
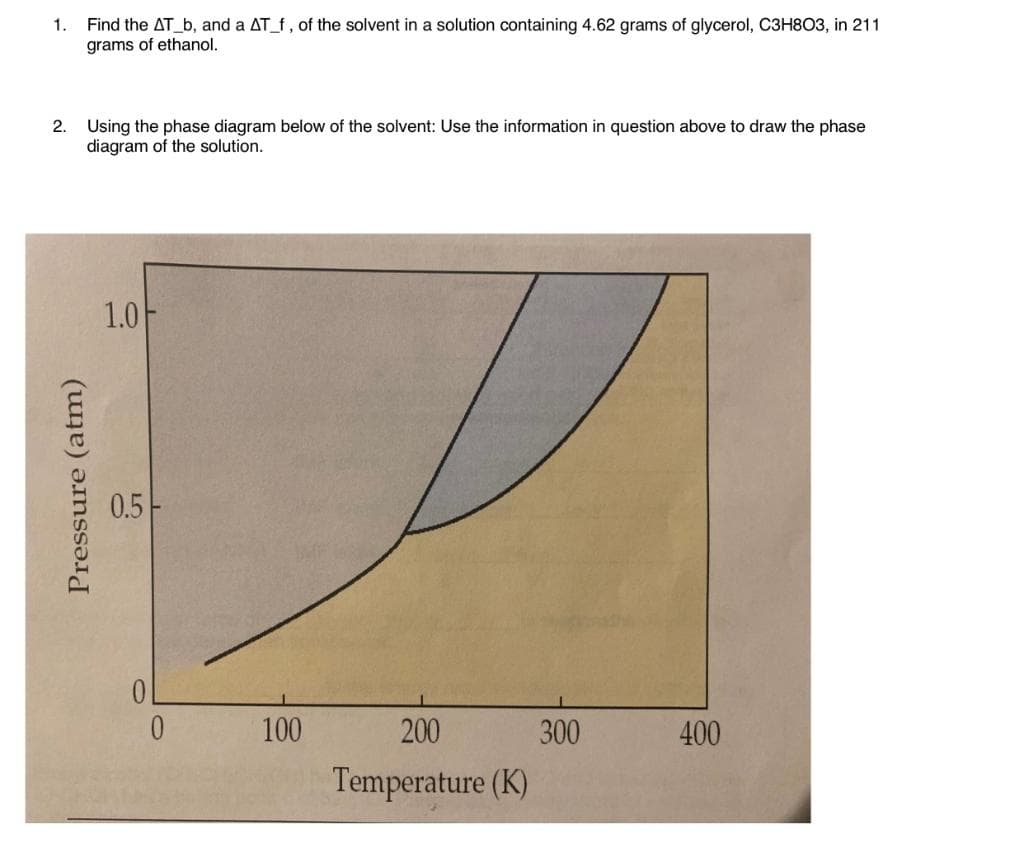
Q: According to the phase diagram above, what is the state of matter of the substance being analyzed at…
A: We need to find According to the phase diagram, what is the state of matter of the substance being…
Q: 4. Identify the following as ionic (I), polar covalent (PC), or nonpolar covalent (NPC) compounds (…
A:
Q: Which one of the following electron configurations would allow a hydrogen atom to absorb a photon of…
A: Answer:1s 1s would allow a hydrogen atom to absorb a photon of light, but not emit one.
Q: Typically, when we use a buret, we measure an initial volume and a final volume, then calculate the…
A: To read the volume in a burette, the lower meniscus is recorded as the final reading. The capacity…
Q: Estimate fs0. Assume the energy transfer efficiency (ε) = 0.6 Electron donor: Ethanol Electron…
A:
Q: Hvor mange kirale sentere har følgende molekyl? N N
A:
Q: Why is it that the ionic activity is equal to the molality (mol/kg) in DHLL (Debye-Hückel Limiting…
A: Why is it that the ionic activity is equal to the molality (mol/kg) in DHLL (Debye-Hückel Limiting…
Q: Use the following chemical equation to answer the question. 4K(s) + O2(g) → 2 K2O(s) If a staff…
A:
Q: At constant volume, the heat of combustion of a particular compound, compound A, is −3046.0…
A: According to the question, The mass of compound A = 1.753 gThe molar mass of the compound A = 112.07…
Q: 1.1(a) What fractions of the molecules inH2andUF6have kinetic energies greater than100…
A:
Q: why is the blue particle the solvent and the red particle is the solute?
A: In a solution, the solvent particles are typically greater in number than the solute particles…
Q: See Figure 4-9. The number of delocalized electrons in Compound H is [Select] Compound H has two…
A: It is based on the concept of resonance. Resonance is one of the factors which help in determing…
Q: Thallium-208 has a half-life of 3.053 min. How long will it take for 120.0 grams to decay to 10.0…
A:
Q: 1. Perform the following synthesis. A. You must use a Grubbs olefin metathesis. B. You cannot use…
A: Initially secondary alcohol esterified with acrylic acid in presence DCC condition. Then silyl…
Q: A student synthesizes tin oxide to create a compound with generic formula SnxOy. She does this by…
A: The mass of tin is = 30.80 g The mass of tin oxide is = 39.10 g The mass of oxygen is = 39.10 g -…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

- a) What would you expect the magnitude and sign of ΔHSOLUTE, ΔHSOLVENT, and ΔHMIXING to be for CH2Cl2 as solute and acetone as solvent? Circle your choices below. Then, fill in the most important IM force that exists between solute molecules, between solvent molecules and between solute and solvent molecules. b) Based on this, what is the overall enthalpy change (HSOLUTION) for dissolving CH2Cl2 in acetone expected to be? (negative/positive/ near zero). Would you expect CH2Cl2 to dissolve in acetone? (yes/no). Explain your answers to both of the questions above. Consider the energy and entropyExtra information: freezing point depression constant for the solvent (20.0 degrees Celcius kg/mol for cyclohexane)Boiling Point Elevation/Freezing Point Depression T = m K where, for freezing point depression: T = T(pure solvent) - T(solution) and for boiling point elevation: T = T(solution) - T(pure solvent) m = (# moles solute / Kg solvent) Kb = boiling point elevation constant. Kf = freezing point depression constant. Kb and Kf depend only on the SOLVENT. Below are some common values. Use these values for the calculations that follow. Solvent Formula Kb(°C / m) Kf(°C / m) Water H2O 0.512 1.86 Ethanol CH3CH2OH 1.22 1.99 Chloroform CHCl3 3.67 Benzene C6H6 2.53 5.12 Diethyl ether CH3CH2OCH2CH3 2.02
- Assuming that pentane and hexane form an ideal solution, A. Calculate the amount of pentane to be added to 65.0 g of hexane to obtain a minimum dG of mixing from mixing those two components. [MM pentane; 72.15, MM hexane; -86.18] B. Calculate the dS of mixing the components of the mixture above.Please indicate true (T) or false (F) as appropriate: Justify I) The vapor pressure of a solvent is reduced by the presence of a non-volatile solute II) In an ideal solution, the vapor pressure is increased by the presence of a solute Volatile. III) Sea water freezes at a lower temperature than sugar water. IV) In the laboratory, chemists use this effect to judge the purity of a component solid: if impurities are present, the freezing point of the compound is lower than the accepted value. A) TFTF B) TTFT C) FTFF D) TTTTThe solvent for an organic reaction is prepared by mixing 70.0 mL of acetone (C3H6OC3H6O) with 56.0 mL of ethyl acetate (C4H8O2C4H8O2). This mixture is stored at 25.0 ∘C∘C. The vapor pressure and the densities for the two pure components at 25.0 ∘C∘C are given in the following table. What is the vapor pressure of the stored mixture? Compound Vapor pressure(mmHg) Density(g/mLg/mL) acetone 230.0 0.791 ethyl acetate 95.38 0.900
- 1. Suppose each pair below is mixed well. What comes out as the simplest example of liquid mixing?A. 12 M sulfuric acid solution and 12 M sodium hydroxide solutionB. water with 80% ethanolC. ethane with methaneD. acetic acid and waterDraw and label completely the liquid-vapor phase diagram of the acetone chloroform system given the following data. a) Only one liquid phase existsb) There is an azeotrope at 64.0 mole% chloroform with taze = 64.4 oC.c) Pure acetone and chloroform boil at 56.1 and 61.2 oC respectively.When pure furfural (solvent) is added to a mixture containing 0.2 mass fraction diphenylhexane and 0.8 mass fraction docosane, two separate layers can be obtained. If the composition of the two layers are as shown in the table, how much solvent was added to 500 kg of solution of docosane and diphenylhexane?
- 1. What is the mole percent of methanol in your starting solution (composed of 1.5 mL and 6 mL isopropanol)? 2. Using the vapor-liquid equilibrium data graph (NOTE: You have to make this graph using the data provided) and your answer from question 1, answer the following: If your first distillate was 49 mole percent methanol, how many theoretical plates were in your distillation setup? Data Mole % of Methanol Temperature (°C) Vapor Liquid 66.22 95.35 90.10 67.94 89.10 79.00 70.22 80.00 66.05 72.67 68.50 52.20 74.78 57.00 40.80 77.06 42.85 29.30 78.94 29.00 19.50 81.00 13.20 8.10It is desired to prepare 2.5 m^3 of a 60 mol percent methanol - water solution. Determine the volumes of methanol and water required to be mixed at ambient temperature given that the partial molar volumes of methanol and water are 58.3x10^-6m^3/mol and 17.2 x 10 m^3/mol respectively. The density of the methanol is 782.51 kg/m^3. Do not use chatgpt. Thank you!If the mixing ratio of CO2 is 350ppmv calculate the CO2 concentration in terms of moles/L and molecules/cm3 . What is the partial pressure of CO2?

