1. From the constituents of the assay, identify "enzyme X" and write out the reaction it catalyses, and the coupling reaction. State why the coupling reaction is needed.
1. From the constituents of the assay, identify "enzyme X" and write out the reaction it catalyses, and the coupling reaction. State why the coupling reaction is needed.
Chapter21: Heparin Infusion Calculations
Section: Chapter Questions
Problem 1.3P
Related questions
Question
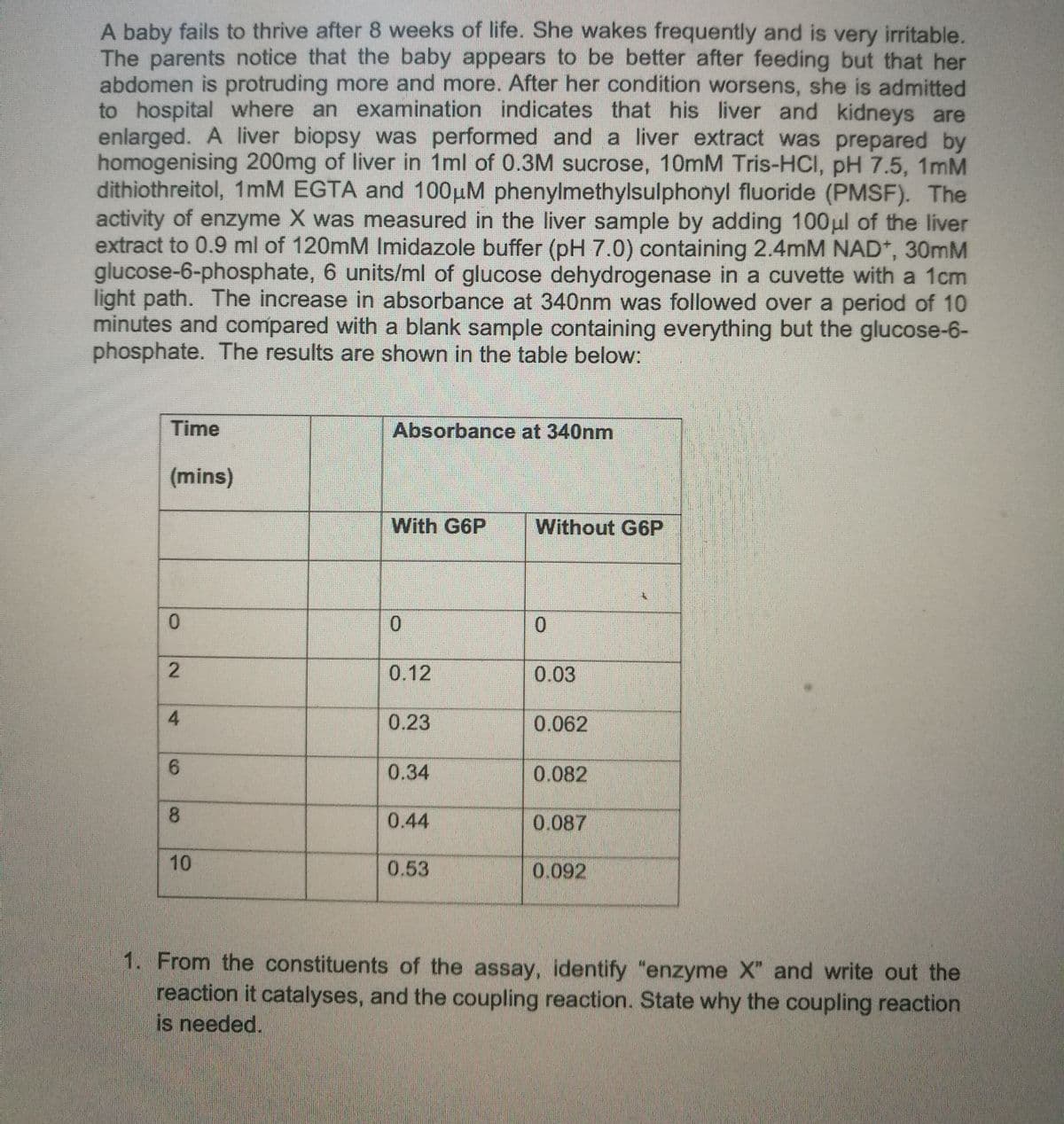
Transcribed Image Text:A baby fails to thrive after 8 weeks of life. She wakes frequently and is very irritable.
The parents notice that the baby appears to be better after feeding but that her
abdomen is protruding more and more. After her condition worsens, she is admitted
to hospital where an examination indicates that his liver and kidneys are
enlarged. A liver biopsy was performed and a liver extract was prepared by
homogenising 200mg of liver in 1ml of 0.3M sucrose, 10mM Tris-HCl, pH 7.5, 1mM
dithiothreitol, 1mM EGTA and 100μM phenylmethylsulphonyl fluoride (PMSF). The
activity of enzyme X was measured in the liver sample by adding 100μl of the liver
extract to 0.9 ml of 120mM Imidazole buffer (pH 7.0) containing 2.4mM NAD+*, 30mM
glucose-6-phosphate, 6 units/ml of glucose dehydrogenase in a cuvette with a 1cm
light path. The increase in absorbance at 340nm was followed over a period of 10
minutes and compared with a blank sample containing everything but the glucose-6-
phosphate. The results are shown in the table below:
Time
(mins)
0
2
4
6
8
10
Absorbance at 340nm
With G6P
0
0.12
0.23
0.34
0.44
0.53
Without G6P
0
0.03
0.062
0.082
0.087
0.092
1. From the constituents of the assay, identify "enzyme X" and write out the
reaction it catalyses, and the coupling reaction. State why the coupling reaction
is needed.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

