Q: What is procedure in extraction of beta-carotene from carrot.
A: Extraction is a technique which is used to a substance from a mixture. It is the first step to…
Q: OH қ OH -Ph Ph H₂SO4 Ph Ph
A:
Q: A chemist is trying to identify an unknown metal, and finds that 25.0 cm³ of the substance has a…
A: Answer: Relation between Mass, volume and density of a substance is: d=MV
Q: From the given reaction: 2 I2 + KIO3 + 6HCl = 5ICl + KCl + 3H2O How many grams of iodine are…
A: Given - Mass of ICl = 28.6 grams
Q: What is the pH of a 0.045 M solution of TRIS base? Answer:
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Curved arrows are used to illustrate the flow of electrons. Using the provided resonance structures,…
A:
Q: Consider a hypothetical chemical reaction: A+B C+D (In this equation A, B, C and D stand for some…
A:
Q: number of student in the classroom, exact or inexact number
A: We know that are the numbers which give the accurate value of the quantity , whereas the inexact…
Q: PG³0 О OPG' OPG2 Н Ф OH Он PG³0
A: Organic transformation.
Q: 4. Consider a sample of gas at 393 K and 22 atm. (a) Calculate the molar volume V (L/mol) of the…
A:
Q: A solution make by mixing 20.0g of a non-volatile compound with 125ml of water at 25 degree c has a…
A: Given: The mass of the non-volatile compound dissolved = 20.0 g The volume of water = 125 mL The…
Q: what happens when you add NaOH vs HCl to sodium phosphate dibasic?
A:
Q: Draw the skeletal structure of pentanal from the Lewis structure (shown below). H Η :Ο:…
A:
Q: Curved arrows are used to illustrate the flow of electrons. Follow the curved arrows and draw the…
A:
Q: Area Of Field Health And Medicine Food And Agriculture Consumer Product Some Chemical Components…
A: Consumer goods Consumer goods, often known as final goods, are items that are purchased by people or…
Q: Question 2NO₂ (g) -->2NO(g) + O₂(g) k = 0.54 (1/M-s) The following is part b. How long (in seconds)…
A:
Q: 1. Complete the reaction and indicate how IR spectroscopy could be used to monitor the reaction…
A:
Q: Consider the following mechanism for the decomposition of ozone: 03(g) → O₂(g) + O(g) 03(g) + O(g) →…
A: The chemical species present in mechanism step but missing in oberall chemical equation are…
Q: Fill in the Blanks Answers typed in all of the blanks will be automatically saved X. X" 52. -967…
A:
Q: 2. In an FCC structure, the atoms are found on the corners of the cubic unit cell in the centers of…
A: In FCC : Number of total atoms in a unit cell =4 Number of atoms at corners = 8 ( contribute each…
Q: A molecule has an empirical formula of CH, and its molar mass is known to be 26 g/mol. What is its…
A:
Q: CCicioalkanes CI Complete the issued, calculated and geometric structural formulas for the following…
A: The name of the cycloalkanes would be written as per the number of Carbon atoms. In expanded…
Q: EtO₂C .CO₂Et Br CHO NaOEt EtO₂C EtO₂C CHO
A: Here we have to synthesize the following given target compound from the given starting material…
Q: In Example 2-1, we established that the mass ratio of magnesium to magnesium oxide is 0.455 g…
A:
Q: The molar mass (to two decimal places) for Fe2(CO3)3 is ______________ g/mol.
A: Molar mass is mass of 1 Mole a substance.
Q: onsider the following elements in their stable forms: 1. Krypton 2. Phosphorus 3. Magnesium 4.…
A: Allotropy is the property of chemical elements that exist in two or more different forms. Diatomic…
Q: 25. Which of the following choices is an example on anthropogenic complexing agents: A) Humic and…
A: Anthropogenic Complexing agents improve cleaning efficiency by inactivating water hardness.
Q: 2. Draw the complete molecular orbital diagram (energy diagram like you have seen in class) for…
A:
Q: The rate of the reaction A + 3B → C + 2D was reported as 2.7 M/sec. Calculate the rates of…
A:
Q: Draw the skeletal structure of pentanal from the Lewis structure (shown below). Η Η Η Η Η :Ο: ||…
A: In the skeletal or bond line angle formula, each covalent bond between carbon-carbon or…
Q: Complete the following table for an isotopically labeled atom or ion: Complete the following table…
A: Given incomplete table is : Complete the given table ? Number of protons (p) = Atomic number (Z)…
Q: How many grams of oxygen (O) are present in a 6.41 g sample of potassium nitrate (KNO3)? Enter your…
A:
Q: You have a semipermeable membrane that rests at -90mV. You also the two ions Na and Cl. Sodium has a…
A: Eion = RT/ZF log [Co]/[Ci] where Eion is the reversal potential, R is the gas constant, T is the…
Q: OH H CCNH H----OC NH, H CHO -H C= I U 9
A:
Q: A sample of 65 mg of a perfect gas at 0.800 bar has its volume doubled and its absolute temperature…
A:
Q: Me2NH, CH,O HCI Me Me
A: The given reaction is an example of Munnich reaction in which, the α-proton of ketone undergoes…
Q: A student is asked to make up 100.0 g of a 13 % (w/w) sucrose solution. The students weighted out 13…
A:
Q: What is the chemical reaction for cellular respiration?
A: chemical reaction for cellular respiration :
Q: Anhydrous, primary standard sodium thiosulfate can be prepared from the pentahydrate, Na2S2O3.5H2O.…
A: Answer: Molar concentration of solute in solution is: Molarity=Mass of solute×1000Molar mass of…
Q: 20. Which of the following statements is TRUE? A) Entropy is an extensive property. B) Entropy is…
A: 20. Entropy is a measurable property of disorderness or randomness of a system. For a ordered…
Q: A sheet of aluminum (density=2.60 g/cm³) has dimensions of 4.75 mm x 1.80 m x 2.48 ft. Calculate its…
A:
Q: An analyst was measuring the performance of a HPLC column. The test solute was Toluene (5 µL). The…
A: To find out the mobile phase velocity, number of theoretical plates and height equivalent to…
Q: Selenium dichloride, SeCl2, has a molar mass of 149.86 g/mol. What is the mass in grams of 0.325…
A: Given, moles of SeCl2 = 0.325 moles molar mass of SeCl2 = 149.86 g/mol mass of SeCl2 = ?
Q: The rate of a certain reaction is given by the following rate law: rate=k[N₂] [H₂] Use this…
A:
Q: What is the area in cm² of a sheet of paper that is 8.50 in x 11.0 in?
A:
Q: For the compound below, locate the pattern we just learned (lone pair next to a n bond) and draw the…
A: Resonance structure: If the Lewis structure of a molecule or ion cannot explain by a single…
Q: An electrical charged atom or group of atoms. What is it
A: All the substances in thE universe are made up of tiny particles known as atoms. Atoms can be…
Q: Correctly classify the following reaction: CH3CH=CH2 + H₂CH3CH₂CH3 a) Elimination reaction b)…
A: Since you have asked multiple type questions, we will solve only first questions for you. If you…
Q: 4.Make a prediction using your model. If an electron were to emit a photon and move from the n=3…
A: Introduction It is mathematically possible to determine the wavelength of light using the Rydberg…
Q: The vapor pressure of a sugar solution is 11.59 mm Hg at 20 °C. What is the mole fraction of sugar…
A: When a nonvolatile solute is dissolved into a volatile solvent the vapor pressure of the solvent…
can u do br and nh2 one please .
no.3 please
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

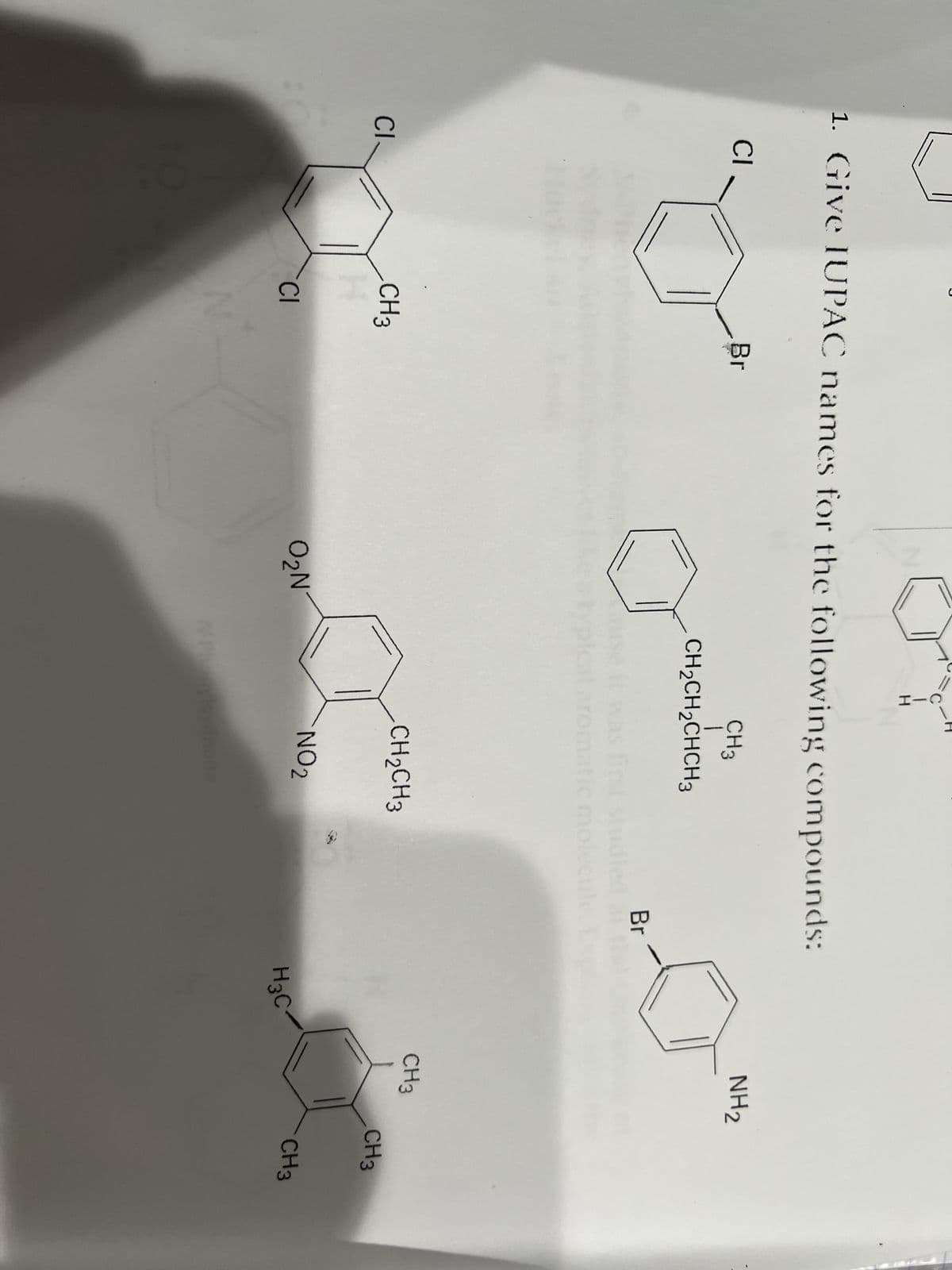
- Give IUPAC names for the following substances (red = O, blue = N):1. The of alka zeltzer in iced water will give a result and observation of _________. alka zetltzer dissolved readily alka zeltzer partially dissolved No reaction 2. In the Le Chatelier's Principle, Co(H2O)6 with oxidation of +2 is _________ in color. blue violet pinkANSWER ALL QUESTIONS 1. write out all the isomers of the compound with molecular formula C4H10O. 2. select the normal/primary isomer and treat it with conc.H2SO4 and heat to produce A. Identify the type of reaction 3. Treatment of A with HCl/H2O gives B and with cold KMnO4/ OH- gives C . Give the name and structures of A, B and C 4. Treatment of A with Hot KMnO4/OH- gives D followed by acidification of the mixture to give E. What is D and E ?
- What is each compound’s systematic name? 1. (CH3)3CCH2CH2CH2CH(CH3)2 2. BrCH2CH2CH2CH2CH2NHCH2CH3 3.(CH3CH2)4C 4.CH3OCH2CH2CH2OCH3select the expected major organic product. I II III IV VFrom the given two compounds in the image, which reagents would differentiate the pair? a. Br2 in CH2Cl2 b. CrO3, H2SO4, acetone c. concentrated HCl with ZnCl2 d. aqueous FeCl3 e. aqueous NAHCO3 f. ammoniacal AgNo3
- what is the compound that most easily reacts with bromine in addition? a) CH4 b) C2H2 c) C3H8 d) C4H101. Fill in the blanks: (Refer to picture 1) In the space provided, give the starting materials or reagents (A to D) needed to produce the compounds shown in each step. (Refer to picture 2)Give the structures of compounds E to I.Identify IUPAC names of A,B,C. Kindly answer quickly
- What is the common name of the product of belowNumber 1 (a to f). Kindly give it's new iupac name, and common name. Number 2 (a to d). Kind draw it. Number 3 (a and b). Kindly write the formula and gibe the common name for the sulfur containing organic products of the following reactions.Cr2(CH3COO)4(H2O)2 was prepared using the following reaction: 2Cr3+ + Zn(s) --> 2Cr2+ + Zn2+ 2Cr2+ + 4CH3COO- + 2H2O --> Cr2(CH3COO)4(H2O)2 (s) Discuss this reaction and how the reagents react with one another to form the product.

