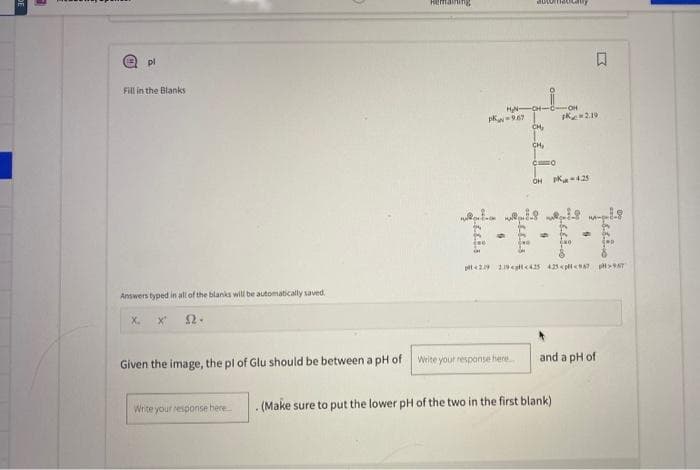
Fill in the Blanks Answers typed in all of the blanks will be automatically saved X. X" 52. -967 Write your response here. Given the image, the pl of Glu should be between a pH of Write your response here... -CH- CH₂ C4₂ CO OH PK-4.25 OH K2.19 18 19 ܘܢܢܐ 29 29 425 425 << > F-T . (Make sure to put the lower pH of the two in the first blank) and a pH of
Fill in the Blanks Answers typed in all of the blanks will be automatically saved X. X" 52. -967 Write your response here. Given the image, the pl of Glu should be between a pH of Write your response here... -CH- CH₂ C4₂ CO OH PK-4.25 OH K2.19 18 19 ܘܢܢܐ 29 29 425 425 << > F-T . (Make sure to put the lower pH of the two in the first blank) and a pH of
Chemistry: Principles and Practice
3rd Edition
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Chapter16: Reactions Between Acids And Bases
Section: Chapter Questions
Problem 16.52QE
Related questions
Question
Transcribed Image Text:pl
Fill in the Blanks
Answers typed in all of the blanks will be automatically saved.
X. X" 22.
Remaining
H₂N-
PK-9.67
autococca
Given the image, the pl of Glu should be between a pH of Write your response here....
CH₂
CH₂
CO
-OH
2.19
OH P-4.25
-1-1
pt 219 219 <p <425 425<pH < 967 967
Write your response here... . (Make sure to put the lower pH of the two in the first blank)
Σ
and a pH of

Transcribed Image Text:I
Show
4/20 answered Time
Meadows. Spencer
pl-Image
a pl
Fill in the Blanks
in
all of the blanks will be automatically sa
x₁ x 22.
o=c
H₂N-CH-C-OH
Write your response here...(Make sure to put the lower pH of the two in the first blank)
pKac = 2.19
Given the image, the pl of Glu should be between a pH of write your response here and a pH of
pKaN = 9.67
CH₂
CH₂
C=0
pKar = 4.25
OH
©
0
H₂N-CH--
AN---
THFI
=1
O=:
CH-C-O
CH₂
CH₂
C=O
www
CH₂
CH₂
C=O
L
OH
****425
H₂NCH-C-OH
CH₂
CH₂
C=O
OH
pH > 9.67
pH<219 2.19<pH<425 4.25<pH<9.67
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning