1. Help Dr. Howell... Draw the product of the reaction in the space given below H H.SO 100°C KMnO GH,0, coldidikute A 1. Nell F2CH₂CH₂CH.B H₂80, CHOOH E OH PB H,80, H,80, Na, Cr₂O, THC Pyridine B D NaOCH CH CH,OH,OCH,CH, 4 C NOCNK I & J LIAIH Waßr M
1. Help Dr. Howell... Draw the product of the reaction in the space given below H H.SO 100°C KMnO GH,0, coldidikute A 1. Nell F2CH₂CH₂CH.B H₂80, CHOOH E OH PB H,80, H,80, Na, Cr₂O, THC Pyridine B D NaOCH CH CH,OH,OCH,CH, 4 C NOCNK I & J LIAIH Waßr M
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter18: Electrochemistry
Section: Chapter Questions
Problem 29Q: When jump-starting a car with a dead battery, the ground jumper should be attached to a remote part...
Related questions
Question
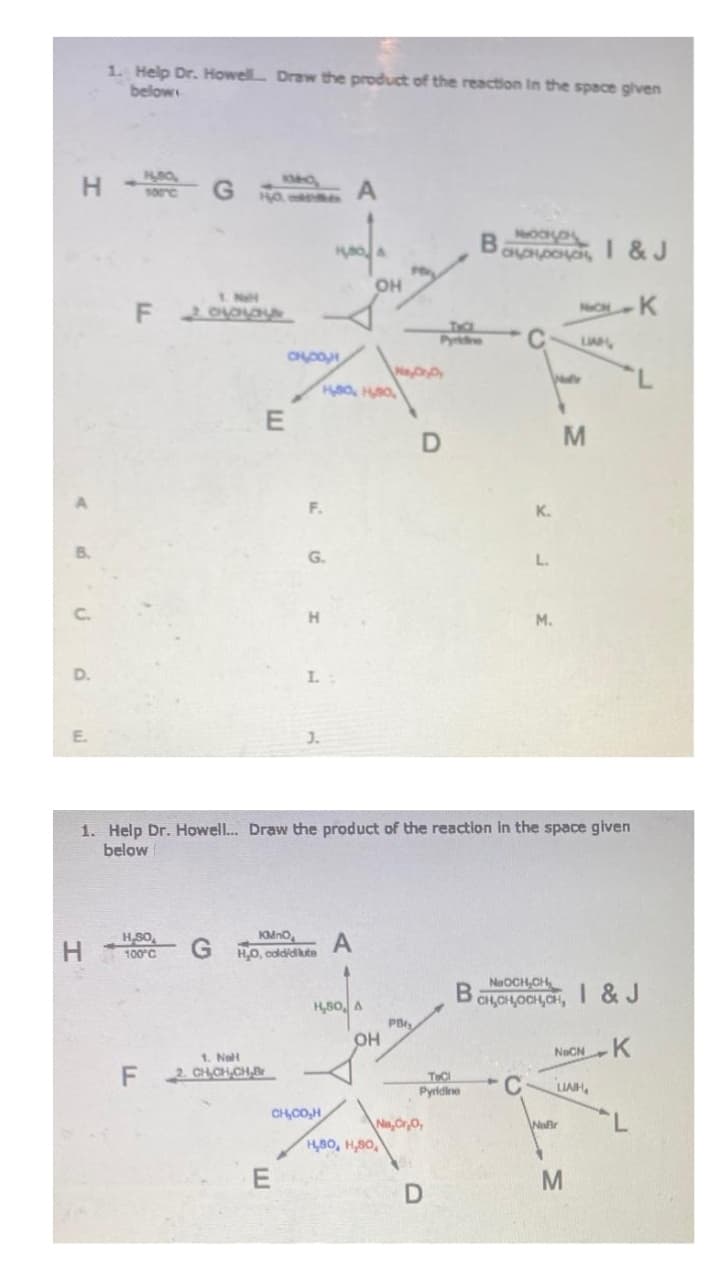
Transcribed Image Text:HH GHO
A
C
D.
E.
1. Help Dr. Howell... Draw the product of the reaction in the space given
below
H
F
H.SO
100°C
E
F
CH,₂00,4
1. Nell
2. CH CH CH.Br
G.
H
E
I.
KMnO₂
GHO, cold/dute
1,80, H,90,
CH,COSH
OH
A
4
H₂80, A
Ne, OryDy
OH
H,SO, H,80
D
1. Help Dr. Howell... Draw the product of the reaction in the space given
below
PB
THCL
Na, Cr₂O,
TOCI
Pyridine
D
B
B
MODEL
CHYCHLOCHÝCH,
K.
L.
C
Wate
M.
NACH - K
I & J
LIAP
M
NaBr
NaOCH₂CH₂
CH,CHLOCH,CH, & J
I
M
NBCN K
LIAH
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.