(R)-2-butanol reacts with phosphorus tribromide to give A (C4H9Br). Treatment of A with sodium cyanide in DMF gives B (C5H9)N. B is optically active. Draw the structure of B. • Use the wedge/hash bond tools to indicate stereochemistry where it exists. • Show stereochemistry in a meso compound. 90-85 To th OO. [F
(R)-2-butanol reacts with phosphorus tribromide to give A (C4H9Br). Treatment of A with sodium cyanide in DMF gives B (C5H9)N. B is optically active. Draw the structure of B. • Use the wedge/hash bond tools to indicate stereochemistry where it exists. • Show stereochemistry in a meso compound. 90-85 To th OO. [F
Chapter5: Stereochemistry At Tetrahedral Centers
Section5.SE: Something Extra
Problem 70AP
Related questions
Question
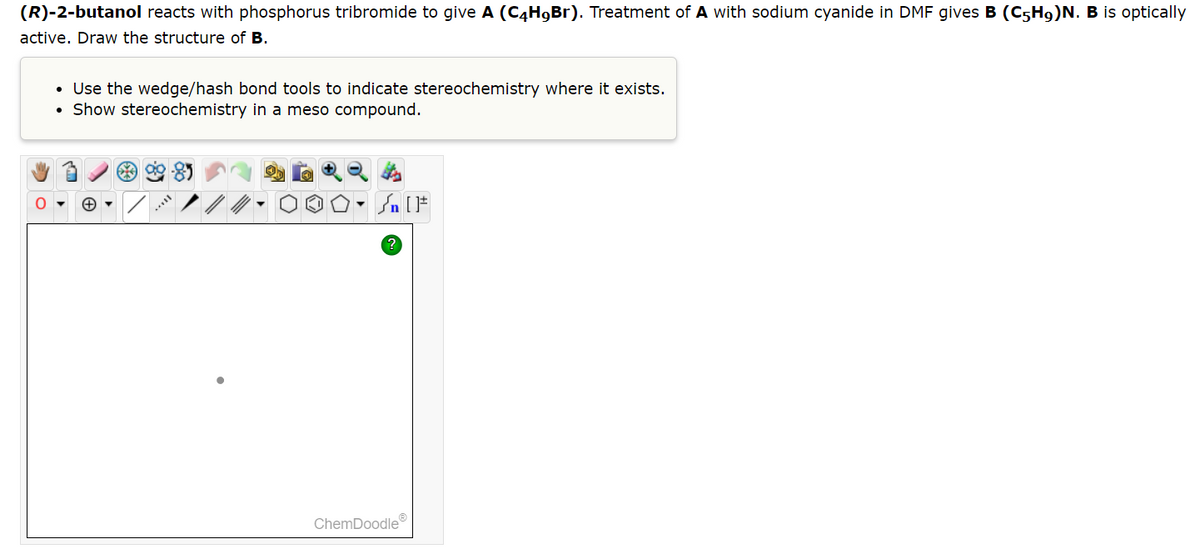
Transcribed Image Text:(R)-2-butanol reacts with phosphorus tribromide to give A (C4H9Br). Treatment of A with sodium cyanide in DMF gives B (C5H9)N. B is optically
active. Draw the structure of B.
• Use the wedge/hash bond tools to indicate stereochemistry where it exists.
Show stereochemistry in a meso compound.
Sn [F
?
ChemDoodleⓇ
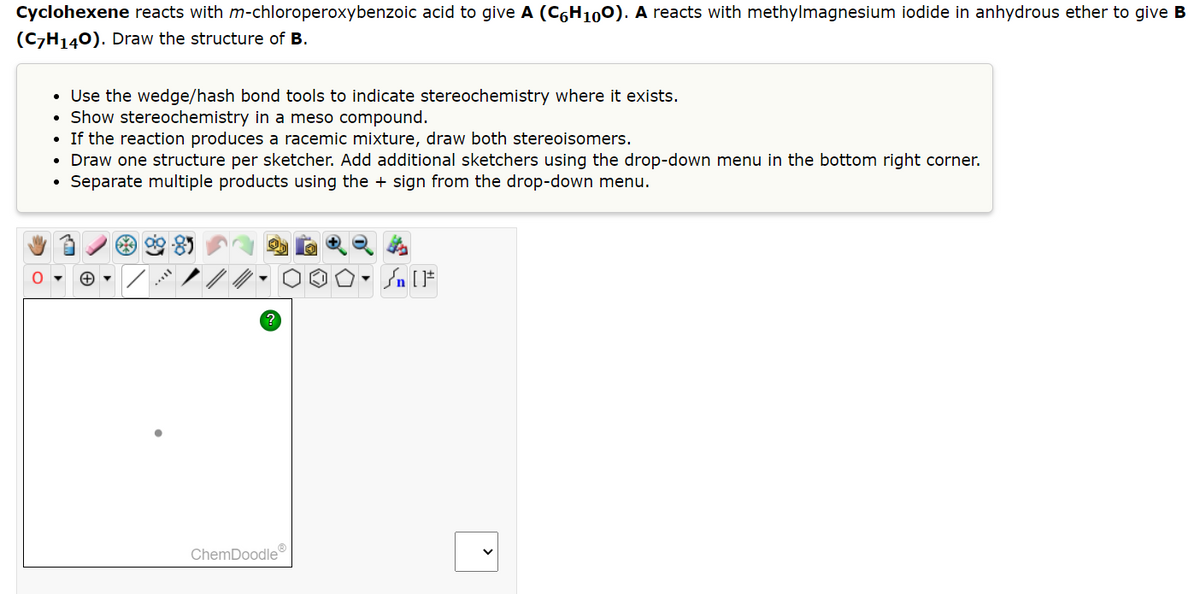
Transcribed Image Text:Cyclohexene reacts with m-chloroperoxybenzoic acid to give A (C6H100). A reacts with methylmagnesium iodide in anhydrous ether to give B
(C7H₁40). Draw the structure of B.
O
• Use the wedge/hash bond tools to indicate stereochemistry where it exists.
Show stereochemistry in a meso compound.
If the reaction produces a racemic mixture, draw both stereoisomers.
●
• Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner.
Separate multiple products using the + sign from the drop-down menu.
?
ChemDoodleⓇ
Sn [F
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

