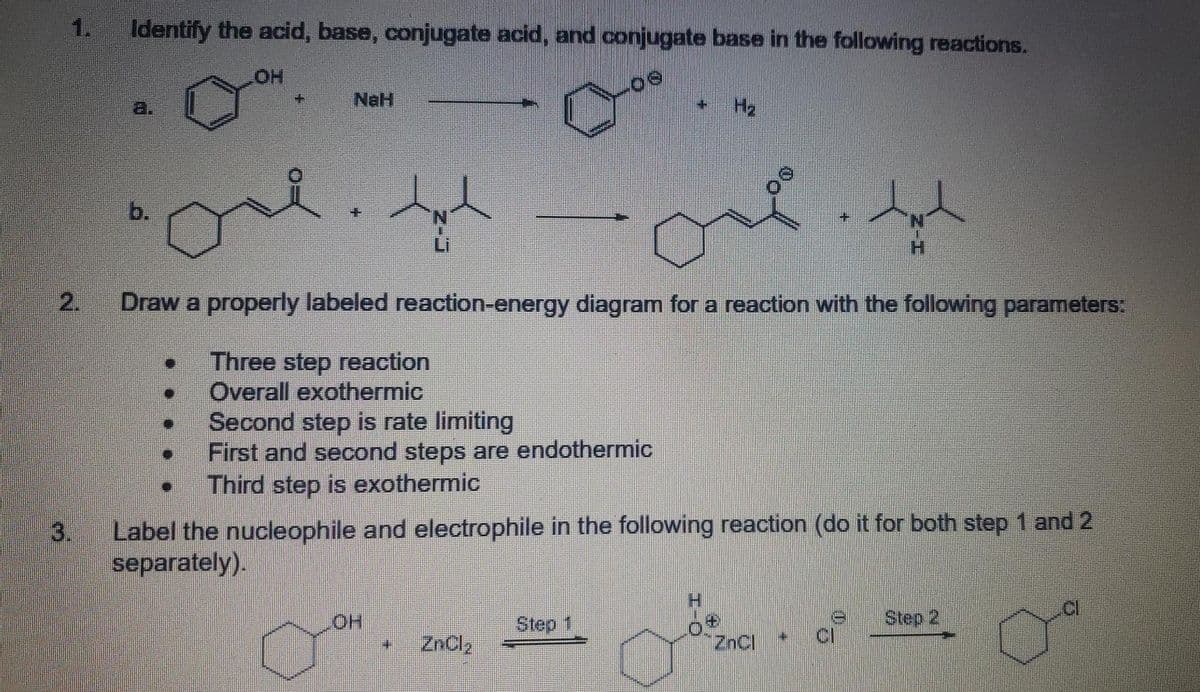
Q: 3. 3. Label the acid, base, conjugate acid and conjugate base in each of the following reactions: +…
A:
Q: Calculate the pH of a 1.2x10-3 mol/L solution of Sr(OH)2. Express your answer to two decimal places.…
A: Sr(OH)2 is act as a strong base and hence it dissociate completely in solution. The dissociation of…
Q: Apply the Bronsted theory of acids and bases to the following reaction: H3AsO4(aq) + H2O( l ) ⇆…
A: Answer:- This question is answered by using the simple concept of conjugate acid and base. Conjugate…
Q: What is the pH of a solution if [H+] = 1.50 × 10-4 M
A: Given :- [H+] = 1.50 × 10-4 M To be calculated :- pH of solution formula :- pH = -log[H+]
Q: In which one of the following reactions does an H2O molecule behave as a Lewis base? O A. Na(s) +…
A: Answer:- This question answered by using the simple concept of Lewis acid and Lewis base. The…
Q: What is the pH of a solution where [H3O+] = 2.3*10-9 M?
A: [H3O+ ] = 2.3 × 10-9 M By the definition of pH, we know that ; pH = - log [H3O+] pH = - log (2.3 ×…
Q: Indicate acids and bases 1) HCl(aq)= H+(Aq) + Cl- (aq) -I know HCl would be an acid, so would that…
A: This is acid base reaction.
Q: 3. Identify the Acid, Base, Conjugate Acid, and Conjugate Base in the following equations: a) HCN…
A: In Bronsted-Lowry's concept of acids and bases, every acid has a conjugate base and every base has a…
Q: а. Acidic, Basic pH РОН [H*] [OH] or Neutral? Solution 9.65 a M M b. Acidic, Basic pH РОН H [OH] or…
A: Hydrogen ion concentration is an important parameter of an aqueous solution. The concentration of…
Q: [H;O'] [OH'] pH pOH acidic or basic ? 1.7 X 10:1ºM 8.79
A: pH = -log[H+] pOH = -log[OH-] pH+pOH = 14 If pH>7, basic If pH<7, acidic
Q: Fill in the missing chemical formulae in the tables below. add conjugate base base conjugate acld…
A: Conjugate base is formed by the removal of H+ ion from the acid. Hence, Conjugate Base of HI is I-…
Q: 1. Identify acid or base, give its conjugate form. A) HCO, в) он C) H;SO. D) H:PO.
A:
Q: In which container is hydrofluoric acid (HF) least ionized? 0.10 M HF 0.10 M HF 0.10 M HF 0.10 M HF…
A: Common ion effect: According to this effect the solubility of an ionic precipitate will decreases if…
Q: Determine the conjugate acid of each base: (a) F −; (b) NO 3−.
A: Conjugate acid is based on Bronsted-Lowry acid-base concept, according to which acid and base react…
Q: Label the acid, base, conjugate acid, and conjugate base in the following reaction: H2O + CN–…
A: Acid is the species which can donate a proton, H+. Base is the species which can accept a proton,…
Q: 51. Fill in the missing information in the following table. Acidic, Basic, or pH pOH [H+] [OH]…
A: The pH of a solution can be calculated as follows: pH=-logH+ We know that; pH+pOH=14 The pOH of the…
Q: Calculate the pH of a solution
A: Given :- mass of NaOH = 2.580 g volume of water = 150.0 mL To calculate :- pH of the solution
Q: pH pOH [H*] [OH]] Acidic, Basic or Neutral? a. Solution 9.60 a M M pH РОН [H*] [_HO] or Neutral?…
A:
Q: Consider the following reaction: HF(aq) + NH3(aq) 2 NH4*(aq) + F'(aq) In this reaction NH4*(aq) is:…
A:
Q: A certain weak base has a K, of 7.50 x 10-. What concentration of this base will produce a pH of…
A:
Q: When the following reaction goes in the reverse direction (from products to reactants), what is the…
A: H2O is base and CN- is conjugate Base so option- 1st is the correct choice among the mentioned…
Q: Write the formula for the conjugate base of each acid. (a) H2SO4 (b) H3BO3 (c) Hl (d) H3O+ (e) NH4+…
A: When an acidic molecule or ion donates H+ ions and accepts OH- ions then we get the conjugate base…
Q: OH NAOH NaOCI, ACOH Он CI но socl, DMP mild base он
A:
Q: Fill in the missing information in the following table. [H*] M [OH]] M pH pOH Acidic, Basic or…
A: The relationships needed to solve the problem are 1) pH + pOH = 14 2) pH = -log[H+] 3) pOH =…
Q: Which two species act as the acids in the following reaction? HF CH3CO0 2F+CH3COOH OA CH3COO, F¯ O…
A:
Q: nsider the following reaction: O4(aq) + HCO3°(aq) 2 SO4²(aq) + H2CO3(aq) nich statement is true of…
A: According to Bronsted-Lowry concept an acid is defined as proton donor and base is defined as proton…
Q: In the reaction H2PO4- + H2O -> H3PO4 + OH-, Which pair represents an acid and its conjugate base?…
A: Within the Bronsted–Lowry acid–base theory, a conjugate acid is a chemical molecule generated when…
Q: A solution of HCl has a pH of 1.94. How many grams of HCl are there in 298 mL of this solution? g…
A: Given, the pH of HCl solution =1.94volume of soltuion =298 mL =0.298 Lwe are asked to calculate the…
Q: Given: HCO_3-(aq) + H_3O+(aq) = H_2CO_3(aq) +H_2O a. What is the conjugate of H_2CO_3(aq)? b.…
A: A conjugate acid has one more hydrogen and positive charge than the base that created it. A…
Q: 7.30×10−7. What concentration of this base will produce a pH of 10.14?
A:
Q: For the reaction: H2CO3 (aq) + CN- (aq) ⇌ HCN )aq) + HCO3- (aq) label each species as an acid or a…
A: Acid is defined as a species which donates proton(H+) whereas Base is defined as an species which…
Q: Label the acid, base, conjugate acid, and conjugate base. H,PO, + CI → HPO,² + HCI
A: H2PO4- is Acid, and Conjugate base is HPO4 2-
Q: A solution is made by dissolving 13.9 g of HONH:CIO3 in 500.0 mL of water. Does HONH3* have any…
A: Bronsted-Lowry acid-base theory: The Bronsted-Lowry acid-base theory states that the acid is a…
Q: Calculate [OH-] fore each solution, express each answer to two significant figures: pH = 1.92 pH =…
A: Calculate [OH-] fore each solution, express each answer to two significant figures:
Q: For the reaction H2CO3 + H2O ↔ H3O+ + HCO3-, H3O+ is the conjugate base. true or false?
A: A conjugate base is formed when an acid loses a proton/ hydrogen ion.
Q: When the following reaction goes in the reverse direction (from products to reactants), what is the…
A: Answer given as follows
Q: equation of the AJ write the dissociation Pretend acid HX in water. BJ Label the acid, base,…
A:
Q: 1. Classify each reactant as an acid or base. CF + NH4* a. HCI + NH3 b. CH3COOH+ H2O CH3COO" + H;O*…
A:
Q: In the reaction H2CO3+2H2O——> 2H3O^+ +CO3^2 What is H2O acting as? A) acid B) base C) conjugate acid…
A:
Q: NaOH(aq) + HCOOH(aq) ⇌ NaHCOO(aq) + H2O(l) Identify the acid, base, conjugate acid, and conjugate
A: Acid which release H+ and base which gain of H+. Conjugate acid is produce from base when base in…
Q: e acid (A), the base (B), the conjugate acid (CA
A:
Q: 2. One mole of H* is always neutralized by mole of OH.
A: Neutralization occurs when moles of acid are equal to moles of base and reacts with each other to…
Q: According to the Bronsted theory, which of the following statements is true?
A: According to the Brønsted-Lowry concept of acids and bases :- an acid is a proton (H+) donor and a…
Q: The ph of a solution is 12.65 what is the [H+
A:
Q: pH pOH H*] [OH"] Acidic, Basic or Neutral? Solution a 9.61 M M [H*] [OH"] M 3.6 x 10-6 M| pH pOH…
A: pH = -log[H+] pOH = -log[OH- ]
Q: In each equation, label the acids, bases, and conjugate pairs: a.) NH3 + H3PO4 ⥫⥬ NH4 + + H2PO4 b.)…
A:
Q: Identify each of the substances in the reaction as acid, base, conjugate acid, or conjugate base.…
A:
Q: 4. Write the formula of the conjugate base of each acid. H2SO4 HI NH4+ HPO42- HCO3-
A:
Q: 1. Classify each reactant as an acid or base. a. HC1 + NH3 Cl + NH4+ b. CH3COOH + H₂O C. OH + HSO4…
A:
6
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

- The Ka of acetic acid (CH3CO2H) is 1.8x10-5 and the Kb of methylamine (CH3NH2) is 4.4x10-4. Complete the following equilibrium reaction (acid-base reaction) equation and predict the side of the equilibrium that is favored. Explain. Show all your work.Organic Chemistry II: How do we know which direct the reaction? Why Methoxy Functional Group direct the reaction instead of fluorine? Is it something to do with electronegativity ? Or is it something to do with withdrawing group?? How do we know if a gourp is more withdrawing ??????How is nucleophilicity (nucleophile strength) related to basicity?
- Calculate the heat of reaction deltaH for the following reaction: 2HCl(g)+Br2(g)——> 2HBr2(g)+ Cl2(g) Round your answer to the nearest kJ/mol.1 example of reaction for each of the types of organic reactions: substitution, elimination, addition, and rearrangement. Show:a. the overall reaction (reactants --> products)b. the reaction mechanism (indicate intermediate product)c. indicate which is the reactive species or intermediate in the reaction (radical? electrophile? nucleophile?)d. overall description of the reaction eg., radical substitution or SROrgonic Chemistry II: The answer is writtten as followed. But I Need Explanation. My question is that: Can I siwtch reagent 2 to reagent 1? for example reagent 1 is Cl2,Fecl3, can I changed it to reagent 2??? Why and why not???
- Tunicates are marine animals that are called "sea squirts" because when they are taken out of water, they tend to contract and expel seawater. Lepadiformine is a cytotoxic agent (toxic to cells) isolated from a marine tunicate. During a recent synthesis of lepadiformine, the investigators observed the formation of an interesting by-product (3) while treating diol 1 with a reagent similar in function to PBr3 (J. Org. Chem. 2012, 77, 3390–3400):Organic Chemistry 1. Can you please explain all three answer choices ? Why is it ! and III, and not II ? Thank youChecking my answer, there was difference with solution. (organic chemistry smith 5th ed 2.47P (d)) Solution said 'the acid base reaction between pentyne and ammonia is product way because pka of pentyne is 25, ammonia is 38 so pentyne will be deprotanted by ammonia.' but I think (and as written in the book) NH4+ is stronger acid than pentyne and acid-base reaction works when reactant is stronger acid than product conjugate acid. It isn't match that pentyne makes NH4+. who is incorrect?
- When water is the solvent, the pKa of acetic acid (CH3CO2H) is 4.75, but when DMSO is the solvent, the pKa is 12.6.Explain why. Hint: Review Section 2.9 and consider the ability of each solvent to solvate cations and anions.Show the steps necessary to transform the acid on the left into the compound on the right. Be sure to use SOCl2The SN2 reaction of (S)-2-brombutan with cyanide (-CN) with the others from the server.

