Q: The evaporation of water is endothermic:H2O(l) → H2O( g) ∆Hrxn = +44.01 kJWhat minimum mass of water…
A: Determination of no. of mol of H2O that will evaporate from 175 kJ/mol: n=175 kJ44.01 kJ/mol=3.9764…
Q: e enthalpy change fo
A:
Q: A sample of sodium undergoes a temperature change of +11.9 °C while absorbing 549.7 joules of 7.…
A:
Q: Please help me show all the work as I will be using this to study, double and triple check your…
A: Temperature change can be calculated as follows:
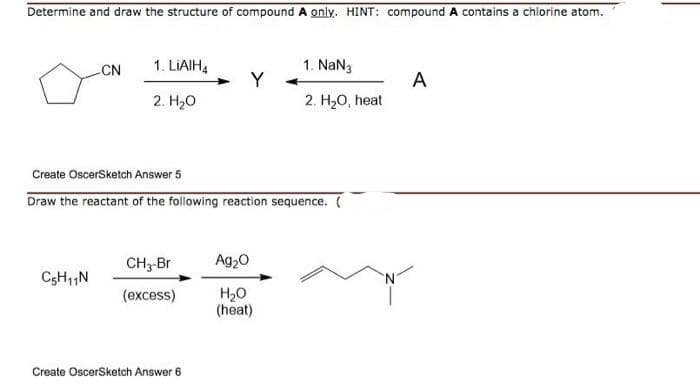
Q: 9. OH 1) PCC 2) NH,NH2, KOH, (HOCH,CH2)20, heat 10. CHO 1) HCN, NaCN 2) H20, H30*, heat 11. 1)…
A: Given ; reactants
Q: With reference to enthalpy changes, the term standard conditions means (a) P = 1 atm (b) some common…
A: Given that : We have to identify which of the following applies to the term standard conditions with…
Q: A 87.3 g chunk of concrete (specific heat: 0.85 J/g°C) is heated to 371.2 °C and dropped into a…
A: Using the heat capacity formula we can find final temperature , q = m x Cs x (Tf-Ti) m = mass Cs=…
Q: How much heat is released when 4.50 g of methane gas is burned in a constant-pressure system?
A: Given: Mass of methane = 4.50g To find: The amount of heat released
Q: || The temperature of a sample of copper increased by 25.0 °C Substance Specific heat J/(g °C) when…
A:
Q: What is the specific heat and average heat of unknown metal? Mass of metal: 10.52 Mass of water:…
A:
Q: Calorimeter mass 162.2 g Mass of calorimeter + cold water 226.6 g Initial temperature of cold water…
A: Note - Since you have posted a question with multiple sub-parts, we are bound to solve only the…
Q: How much heat (in J) is evolved or absorbed when a 35.1 g sample of ethanol (specific heat = 2.46…
A: specific heat capacity is the amount of heat required to raise the temperature of 1 Kg of a…
Q: When 47.1 J of heat is added to 13.2 g of a liquid, its temperature rises by 1.75 ° C. You may want…
A: The heat capacity of a substance is defined as the heat required to raise the temperature of one…
Q: A 37.70 g sample of a substance is initially at 28.8 °C. After absorbing 2571 J of heat, the…
A: The given problem can be solved by using the formula given below as; Q=m c ∆T…
Q: The specific heat capacity of methane gas is 2.20 J g¹ K-1. How many joules of heat are needed to…
A:
Q: 1) NAOH, heat Br d) 2) MCPBA Br 1) KOCCH,), heat 2) H,0
A: Elimination can occur via two elimination mechanisms - E1 and E2 Strong base like NaOH prefers E1…
Q: What does C represent in the change in heat formula? q= AH = mx C × AT O specific heat capacity O…
A: In given formula C is specific heat capacity.
Q: How much heat is required to raise the temperature of 1.5 x 103 g of water from 45'F to 130.F? The…
A:
Q: An ideal gas in a cylinder fitted with a piston expands at constant temperature from a pressure of 9…
A: note : as per bartleby guidelines i can provide you a solution of only first question in case of…
Q: 2080 J of energy are transferred into a 805.9-g chunk of copper (specific heat: 0.387 J/g K). By how…
A: Q = m . s . ∆T Where, Q = energy transfer in Joule ; m = mass of material in gram; s = specific heat…
Q: The specific heat of water is 4.18 Jg1°C1.Calculate the molar heat capacity of water. -1o
A: Step : 1 Calculate the molar heat capacity of water.
Q: In the laboratory a student finds that it takes 19.7 Joules to increase the temperature of 10.3…
A:
Q: The specific heat of a certain type of cooking oil is 1.75 J/(g⋅°C).1.75 J/(g⋅°C). How much heat…
A: Heat of reaction is difference of energy of product and energy of Reactant.
Q: How much heat is transferred from a 4.00 g gold nugget (specific heat = 0.129 J/g-°C) initially at…
A: Given : Mass of water = 5 Kg = 5000 g…
Q: HO HO HO Oa. 1) H2SO4/heat, 2) KMNO4/ H2O, H;O", heat Ob. 1) Ag20, 2) H:о* 1) NACN, 2) H3O+
A: Dehydration reaction takes place in the presence of protic acid and heat. Furthermore, alkene can be…
Q: What mass of natural gas (CH4) must burn to emit 265 kJ of heat? CH4(g)+2O2(g)→CO2(g)+2H2O(g)…
A: To solve this problem we have to consider the given thermochemical equation . According to this…
Q: What is the heat capacity of a substance if it takes 6.9 kJ of heat to warm 396.1g by 30.6 C?
A: Heat capacity ( c ) is the quantity of heat energy required to increase the temperature of a…
Q: A 125 g sample of liquid water has a temperature of 32.5 oC. What is the temperature of the water…
A: Given Mass of liquid Water = M = 125 g Temperature of water = T1 = 32.5 °C Heat Added to sample =q…
Q: 1. Given the specific heat (J/g∙°C) of the following metals: Al = 0.900 Au=0.129 Cu=0.385 Fe=0.444…
A: To solve this problem we will use the formula Q = m • c •∆T Where , Q = heat released or…
Q: 1. NaOMe, HOME Br 2. H,о", heat Br Meo OMe 3. SOC, heat 4. H2N-iPr (2 equiv)
A:
Q: 35 g of aluminum (specific heat 0.88 J/g °C) at 24 °C are placed in 180 mL (180 g) of coffee at 85…
A: Given : Mass of Aluminium = 35 g Specific heat = c = 0.88 J/g0C…
Q: hwquiz6-1 EP Immersive Reader Open in Desktc 12. How much heat is released when 3.42 g of nitrogen…
A:
Q: What is the heat capacity of 265 grams of water? The specific heat of water is 4.184 J/g °C. O 1105…
A:
Q: How much heat is required to raise the temperature of 854 g of H2O from 23.5 °C to 85.0 °C?
A: 4. Given : Mass of H2O = 854 g Initial temperature = 23.5 °C Final temperature = 85.0 °C To…
Q: NaOH N: КОН H,O, heat D,0 NEt,, heat
A:
Q: KH :2. NEt,, O C, hv heat
A:
Q: HNO, 1. HCI, Fe NANO, 1. HBF. 2. NaOH H,SO, 2. heat .F
A: When benzene is treated with Mixture of concentrated nitric acid and Sulfuric acid, then nitro…
Q: The specific heat of toluene (C7H8), is 1.13 J/g•K. How many kJ of heat are necessary to raise the…
A:
Q: 1. HNO,, H,SO4 2. H, Ni 3. NANO,, HCI 4.CuCl 1. Xs CH,I 2. Ag:0, Heat NH2
A: We have to find the product of these reactions.
Q: The specific heat of octane, C8H18(l), is 2.22 J/g-K. (a) Howmany J of heat are needed to raise the…
A: Molar mass of octane: 114 g/molSpecific heat: 2.22 J/g-K Molar mass of water: 18 g/molSpecific heat:…
Q: 1. PhMgBr, THF 2. PCC, CH2CI2 3. Os04, TBHP THF/H2O 4. Теон, PhН, heat
A: Ans
Q: 1. NaOEt, EIOH Br EI0 1 eg 2. H,0", H,0, heat
A: abstration of acidic proton by base attack of nucleophile to the alkyl halide hydrolysis of ester…
Q: The specific heat of aluminum is .900J/g degree C, what is its molar specific heat?
A: The specific heat of aluminium=0.900 J/goC Mass of 1 mol of aluminium= 26.98 g Molar heat capacity…
Q: Charcoal is primarily carbon. Determine the mass of CO2 produced by burning enough carbon (in the…
A: Given ∆H0rxn = -393.5 kJ | unit of ∆H0rxn = kJ not k Amount of heat energy produced = 5*102…
Q: A 27.7 g sample of ethylene glycol, car radiator coolant,loses 688 J of heat. What is the initial…
A:
Q: in H,0* CH NH, H,O* Он H. но HCI heat
A: The first reaction is a nucleophilic acyl substitution. Here nucleophilic amine is being acylated…
Q: How much energy is evolved as heat when 0.1 L of water at 0 degrees celcius solidifies to ice. (The…
A: The density of water at 0oC is 0.99987 g/mL. The mass of 0.1 L of water at 0oC is; 1.0 L=1000.0…
Q: O2 Mg(s) + 0,(g) → 2 MgO(s) + heat O NH, (g) + HCI(g) NH,Cl(s) + heat AgCl(s) + heat → Ag*(aq) + CI…
A: Exothermic reactions are reactions or processes that release energy, usually in the form of heat or…
Q: The combustion of 2.41 g of butane (C4H10) in a calorimeter causes a rise in temperature of 12.1°C.…
A:
Step by step
Solved in 4 steps with 3 images

- Give minor or major product?Which or which of the statements given below is correct. I) Maleic anhydride is a carboxylic acid derivative and its reaction with water is a reduction reaction. II) Fumaric acid and maleic acid are stereoisomers of each other III) Since fumaric acid has a more stable structure than maleic acid, its boiling point is higher. A. Solo I B. I and III C. II and III D. I, II, III E. Solo IIIFind a naturally occurring-chalcone and answer the following questions:a. Draw its structureb. Does it have biological activity? If so, explain what it doesc. Draw the two starting carbonyl compounds that could be used to make this naturally occurring chalconed. List references used. At the very minimum, include a reference for where you found the chalcone.
- 1. Fill in the blanks: (Refer to picture 1) In the space provided, give the starting materials or reagents (A to D) needed to produce the compounds shown in each step. (Refer to picture 2)Give the structures of compounds E to I.Supply the missing reagent in the following reaction: (see attachment) A. Lindlar, H2, mCPBA B. O3 or KMnO4 C. fused KOH D. NH3 E. NaNH2COMPLETE THE TWO REACTIONS BELOW
- Complete the reaction belowWhat is going on from compound B to compound C? Can you please select all the options that applyWhat is the objective of this experiment in a few sentences? PART C & D (couldn't post picture) c. Acid HydrolysisIn a small erlenmeyer flask place 25 mL of the stock starch solution and 5 mL of 10%HCI and mix. Place in boiling water bath. After 30 minutes of heating, use a plasticpipette to withdraw a few drops (aliquot) and place them on a spot plate. Allow to coolfor a few seconds and add 2 drops of iodine solution. Observe color and interpret theresults. Withdraw and test an aliquot every 5 minutes until the test with iodine does not produce ablue color. Keep track of the total elapsed time. Interpret the results. Do not discardsample and proceed to part d. d. Fehlings Test on hydrolyzed sampleUpon completing part c. above and the test no longer gives a blue color, withdraw a 5 mLsample and neutralize with 10% NaOH until no longer acidic to litmus. Add 1 mL ofFehlings solution (A+B), place in boiling water bath, and time any color change asbefore. Compare this result with your…
- Based on the image attached, explain the step-by-step mechanism reaction of reduction for methyl-2-hydroxybenzoate that formed 2-hydroxymethyl phenol as a product.1.Explain why pyridine ( Kb=2.3x10-9) is a much stronger base than pyrrole (Kb=2.5x10-14), 2.Explain and illustrate, why it is difficult to perfom Friedel-Craft reactions on unactivated pyridine.Can you help with part b. Thank you!

