Q: A gas mixture contains 0.35 moles of Ne, 0.10 moles of He, and 0.39 moles of Ar. If the total…
A:
Q: Consider the insoluble compound aluminum phosphate , AlPO4 . The aluminum ion also forms a complex…
A: Given , Insoluble compound Aluminium phosphate (AIPO4)
Q: Suppose i want to prepare or manufacture Bike Polish, Now what is the Master formulae for preparing…
A: The formulation of a bike polish can vary depending on the desired properties such as shine,…
Q: Calculate the pH of the following solutions prepared by: Part A Dissolving 4.7 g of lithium…
A: Dear student since you have posted multiple parts question, As per our company QnA guidelines We are…
Q: What are the special rules or laws to predict predominant products of alcohol and ether (organic…
A: There are some general reaction of common functional group in organic chemistry. They are frequently…
Q: Part C Write a balanced redox equation for the reaction that occurs. Express your answer as a…
A: So Au (gold) does not dissolved. But Al (aluminum) got dissolved
Q: hen 12.0 mL of a 8.00 x 10^ -5 M potassium sulfate solution is combined with 18.0 mL of a 4.49 x 10^…
A: Solubility product quotient, Qsp is the product of the concentrations of the products, each raised…
Q: Complete the following table by calculating the missing entries and indicating whether the solution…
A:
Q: Consider the following system at equilibrium where AH = 108 kJ/mol, and Ke = 1.29x10-2, at 600 K.…
A: This whole process problem is based on the concept of Le Chatelier's principle. Equilibrium constant…
Q: A solution is prepared by dissolving 0.20 mol of propanoic acid and 0.20 mol of ammonium chloride in…
A: 0.20 mol of propanoic acid and 0.20 mol of ammonium chloride are dissolved in enough water to make…
Q: Which of the following scenarios would have a greater osmotic pressure? a) 0.250 M lithium bromide…
A: Osmotic pressure - The external pressure applied on solution so the process of osmosis stops called…
Q: Which of the following bonds is the most polarized without actually being ionic? C-H O-H S-H Na-F…
A: The covalent bond is formed by the sharing of electrons between the atoms.
Q: Which of the following is formed by arranging its components in a repeating pattern? A.…
A: Crystalline materials have a highly ordered and repeating pattern of atoms, molecules, or ions,…
Q: Could someone please Create 2 different acids Models I wrote the formula But do not know how to turn…
A:
Q: Using the graphic below which elements would demonstrate more metallic character? A. Elements in the…
A: Metallic property: Metals are electropositive and they easily lose electrons to form cations. The…
Q: 1) Provide the major organic product in the reaction below. 1. Na CH3CH2CH2OH Answer: 2. PhCH₂Br
A: We have given the organic reaction and we have to find out the major product of the reaction.
Q: Consider a container that is divided by a semipermeable membrane. A sodium chloride (NaCl) solution…
A: Answer: When a solution and solvent are separated by a semipermeable membrane than solvent particles…
Q: Answer the questions below about the highlighted atom in this Lewis structure: H 4 HIC C=C-H I H In…
A:
Q: Consider the following reaction: 2 A2B5(g) → 4AB2(g) + B2(g) The rate constant is…
A: We know for a first order reaction , [A]t = [A]oe-kt where k = first order rate constant
Q: Which of the following is the best choice for a buffer system? 1.equal volumes of 0.1 M ammonia…
A: A buffer is a solution which resist any change in pH on adding a small amount of acid or base . it…
Q: How is C2H6 classified? A. both an Arrhenius acid and Bronsted-Lowry acid B. neither an acid, nor a…
A: Arrhenius theory and Lowry-Bronsted theory are two different models used to describe acid-base…
Q: How do you test to be sure there is a release of oxygen in a mercury oxide lab experiment. Is the…
A: Mercuric oxide decomposes to mercury on being heated which condenses and form a silver mirror .…
Q: y-glutamyltranspeptidase Cysteinylglycinase N-acetylase H Acrolein Å GSH GSH
A: The enzyme involved in the formation of mercapturic acid is called gamma glutamyltranspeptidase…
Q: Consider the following reaction to form the anionic metal complex [CuCl2]- Cu+ (aq) + 2…
A: Since,Equilibrium constant or Kc is the ratio of the equilibrium concentrations of product over…
Q: An unstable isotope of Re-191 is a beta producer. What is the other product of the reaction? A)…
A: The term isotopes can be defined as the variants of a given chemical element that differ in neutron…
Q: alculate the concentration of IO3 in a 7.45 mM Pb(NO3)₂ solution saturated with Pb(IO3)2. The K sp…
A:
Q: What is the solubility, in mol/ L, of AB2 in pure water? Ksp = 10.52 x 10-8 for AB2 Report your…
A:
Q: The below synthesis was designed using the Organic Chemistry Roadmaps in the appendix of your…
A: Na in NH3 reduce alkyne to trans or E-alkene. The alkene gets controlled ozonolysis with O3/(CH3)2S…
Q: Predict the major product(s) you would obtain from sulfonation of fluoroenzene. On a benzene ring
A: We have to give the major product of the sulfonation reaction of fluorobenzene. Introduction:…
Q: How many grams of Br2 are needed to completely convert 15.0 g Al to AlBr3?
A:
Q: CH3 C1₂ CC14
A: solving only the first question as per guidelines. To draw the product and mechanism of the given…
Q: 3) Consider the galvanic cell whose cell diagram is given below. Ni(s) | Ni** (aq, M=0.500 M) || Ag…
A: We will use the following formulas-
Q: 0.8 moles of NaCIO is added to 1.0 liters of water. What is the pH of this salt solution, assuming…
A:
Q: 11. Carbonate buffers are important in regulating the pH of blood at 7.40. What is the concentration…
A:
Q: 5. Zn(s) + NO 3(aq) 2+ (aq) Zn + 4(aq) + NH (acidic)
A: Since you have asked multiple type questions, we will solve only first questions for you. If you…
Q: a) b) 9 c) Br Br Br. Na₂S Li CN acetone OH 1. Na OH 2. CH₂l
A: The product of given reactions are shown below;
Q: Consider the following rection: 2 A(g) + B2(g) ⇌ 2 AB(s) If Kc for the…
A: Kc is equilibrium constant
Q: The enthalpy change for the formation of 1 mole of CH, from its elements in their standard states is…
A:
Q: > MISSED THIS? Watch KCV 11.4, IWE 11.6; Read Section 11.8. You can click on the Review link to…
A: The question is based on the concept of ideal gas equation. we need to calculate temperature for…
Q: How many non-bonding electrons are on the central atom in the optimized Lewis structure of NBr3, in…
A: Lewis structure is a simplified way to depict the electrons and bond of a molecule.
Q: Consider the Arrhenius equation: k=Ae^(Ea/RT) Which of the following statements is not correct?…
A: The objective of the question is to identify the statement which is not correct :
Q: Explain the observed reactivity rate of the following alkyl halides in an S№1 reaction. -CI slowest…
A:
Q: If a sample of pure X is a mixture of liquid and gas, which point marks the highest possible…
A: Phase diagram: The phase diagram represents the different phases of the substance and the pressure…
Q: Calculate the pH and pOH of the given solutions at 25 -C from the concentration of H3O+ ions. Answer…
A:
Q: solution contains 5.72×10-3 M potassium sulfate and 6.78×10-3 M ammonium fluoride. Solid lead…
A: The question is based on the concept of chemical equilibrium and solubility product expression. we…
Q: 916 JOY 19V9 11 An aqueous solution has A hydroxine ion concentration of 1.0 x 10-7 m. a) hip nila…
A: Answer:- This question is answered by using the simple concept of calculation of hydronium ion…
Q: Consider the following system at equilibrium where AH° = 108 kJ/mol, and Ke = 1.29x10-2, at 600 K.…
A: This whole process problem is based on the concept of Le Chatelier's principle. Le Chatelier's…
Q: What is the pH of a buffer solution where [HA] = 1.5[A-] and Ka = 4.35 x 10-5? Report your answer…
A: Given -> [HA] = 1.5[A-] Ka = 4.35 × 10-5 -> pH of buffer solution can be determined by using…
Q: Calculate the amount of heat needed to boil 19.8 g of acetic acid (HCH3CO₂), beginning from a…
A:
Q: Consider Following reaction: CH4(g) + 4 NO(g) → 2 N2(g) + CO2(g) + 2 H2O(g) The concentration of…
A:
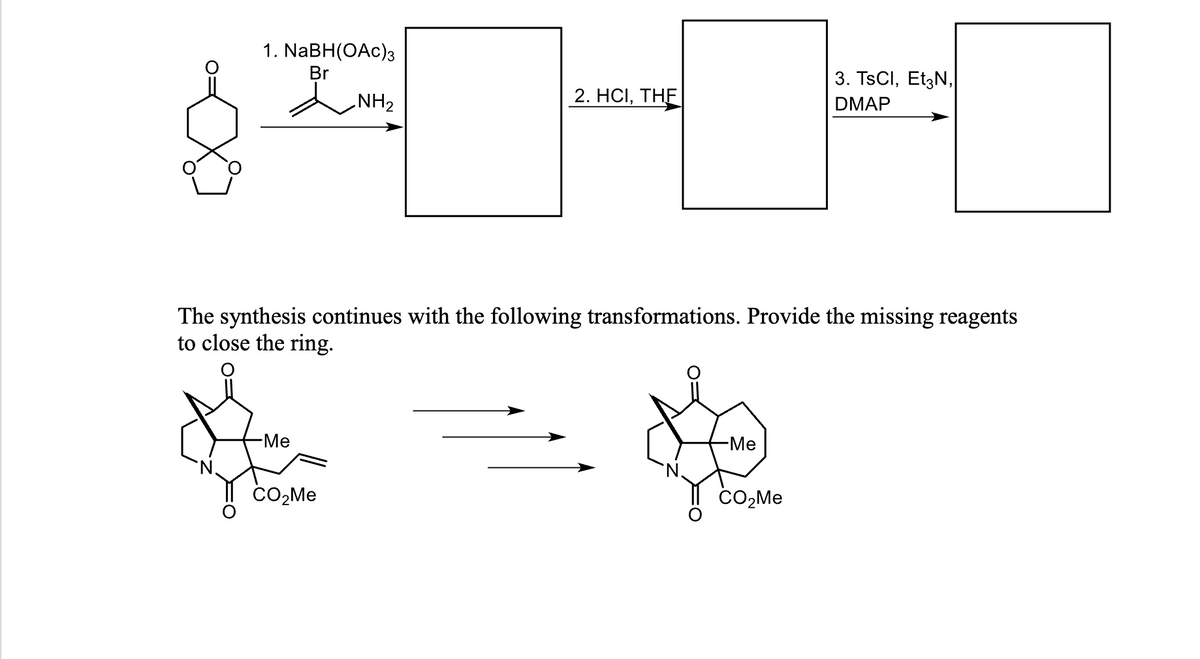
Provide the intermediates for the reactions below. Thank you !
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- These reagents can produce ketones with alkynes A. BH3, THF, H2O2 B. KMnO4 C. O3 D. H2SO4, H2O, HgSO4Which of the following reagent best accomplish this transformation below? a. BH3, THF, H2O2 b. NaNH2, NH3 c. H2SO4, H2O, HgSO4 d. H2, LindlarChoose the best reagents from the list provided below for carrying out the following conversion. Match the reagent with the step number. HCl (aq), Zn(Hg) Br2, FeBr3 Na/NH3, -33 degrees C NBS, light KMnO4, H3O+ Mg metal, ether KOH, EtOH, heat
- Triethyloxonium tetrafluoroborate, (CH3CH2)3O+ BF4 −, is a solid with melting point 91–92 °C. Show how this reagent can transfer an ethyl group to a nucleophile (Nuc:−) in an SN2 reaction. What is the leaving group? Why might this reagent be preferred to using an ethyl halide?When 3-methyl-1-butene is reacted with 9-borabicyclo[3.3.1]nonane, the "1-ol" product is formed. What is the detailed reactin scheme for the transformation? Describe the purification procedure.Explain how the reaction of (CH3)2CHCH(Cl)CH3 with H2O yields two substitutionproducts, (CH3)2CHCH(OH)CH3 and (CH3)2C(OH)CH2CH3
- Rank the following in increasing order of reactivity towards nucleophilic acyl substitutiona. CH3COCl, CH3COOCH3, CH3CONH2b. CH3COOCH3, CH3COOCH2CCl3, CH3COOCH(CF)3In which compound is the halogen substituted most rapidly by aq hydroxide ions? a. CH3CH2CH2CH2Cl b. (CH3)3CI c. (CH3)3CCl d. CH3CH2CH2CH2IPredict the major products of the following reactions.(a) 1@ethylcycloheptene + ozone, then (CH3)2S(b) 1@ethylcycloheptene + warm, concentrated KMnO4(c) 1@ethylcycloheptene + cold, dilute KMnO4
- What product(s) would you expect to obtain from reaction of 1, 3-cyclohexadiene witheach of the following?(a) 1 mol Br2in CH2Cl2(b) O3followed by Zn(c) 1 mol HCl in ether(d) 1 mol DCl in ether(e) 3-Buten-2-one (H2C = CHCOCH3)(f) Excess OsO4, followed by NaHSO3Reaction of (CH3)3CCHO with (C6H5)3P=C(CH3)OCH3, followed by treatment with aqueous acid, affords R (C7H14O). R has a strong absorption in its IR spectrum at 1717 cm−1 and three singlets in its 1H NMR spectrum at 1.02 (9 H), 2.13 (3 H), and 2.33 (2 H) ppm. What is the structure of R? We will learn about this reaction in Chapter 18.Determine the reagents A, B, C, D, E and the structure of compounds I, II, II in the reaction below



