Q: The sublimation of carbon dioxide at -78°C is Calculate ASsub when 24.8 g of CO2 sublimes at this…
A: Chvvvvv
Q: HNO, H,SO, KMno, H (b) 1. Fe, HCI 2. NaOH NaNO, HCI (4) HO, heat (e) (a) CHOH, H mehybaraben
A: Toulene upon nitration p-nitro toulene is formed Alkyl benzene upon oxidation with KMnO4 gives…
Q: How many moles of potassium oxide (K,O) will be formed when 1.52 moles of potassium rea vith oxygen…
A:
Q: Chlorine monofluoride can react with fluorine to form chlorine trifluoride:(i) ClF(g) + F2(g) ⟶…
A:
Q: Balanced Chemical Equation Reaction Type: At completion of reactions: Grams of calcium hydroxide…
A: The answer are as follows:
Q: Stored chemical energy E) How does the diagram below show that the reaction is endothermic? Products…
A:
Q: Consider these reactions, where M represents a generic metal.…
A: The given equations are as follows. 2M(s)+6HCl(aq)⟶2MCl3(aq)+3H2(g)…
Q: Consider these reactions: Reaction 1: H2 (g) + Cl2 (g) → 2HC1(g) AH = -184.6 kJ Reaction 2: 20F2 (g)…
A: -> If ∆H =+ve => reaction is endothermic reaction. -> if ∆H = -ve => reaction is…
Q: __ Cu(NO3)2 (aq) + __ NaOH (aq) → Type of Reaction:
A: Given:
Q: Name the type of reaction SO3 S OH H,SO4
A: To find: The type of reaction
Q: Given these reactions, X(s)+1/2O2(g)⟶XO(s) Δ?=−887.7kJ/mol XCO3(s)⟶XO(s)+CO2(g)…
A: X(s)+1/2O2(g) ⟶ XO(s) ΔH1=−887.7kJ/mol…
Q: Classify these reactions as exothermic of endothermic: a. energy + SO2 (g) b. C3H18 (g) + O2 (g) c.…
A: The answer of the above question is given in following steps
Q: Which of the following reactant make gas products? O All of these are examples will form a gas O Hl…
A: When two or more Substances react with each other, they produce different type of compounds. Like -…
Q: 2. a) Write a formation equation for CaS04 including states and a AH term after the equation. b)…
A:
Q: 41.56 g of CaCO3(s). CaO(s) CO2(g) -> CACO3(s)
A: Applying mole concept and gas equation formula P1V1/ T1= P2V2/ T2
Q: Use the following equation to answer the question. PCI5 PCI3 + Cl2 + Heat ---> Which of the…
A: As per Le Chatelier's principle, if any change is made in a chemical reaction like concentration of…
Q: Consider these reactions, where M represents a generic metal.…
A: Given reactions: 2M(s)+6HCl(aq)→2MCl3(aq)+3H2(g) ∆H1=-669.0 kJ----eqn(1)HCl(g)→HCl(aq)…
Q: Which of the following reactions are endothermic, and which are exothermic? a) N2 + 3H2 - 2NH3 +…
A:
Q: Consider these reactions, where M represents a generic metal.…
A: Answer: We have to calculate enthalpy of formation of the given reaction.
Q: The amphoteric reaction between two water molecules is energy + H20 + H20 <-» H3O* + OH". Is this…
A: Two water molecules dissociate into H3O+ and OH- is the reaction of self ionization of water or auto…
Q: Consider the following reaction Ca(s) + 2HCI(aq) --> CaCl2(aq) + H2(g) How many moles of CaCl, can…
A: 2 moles
Q: The reusable booster rockets of the space shuttle use a mixture of aluminum and ammonium perchlorate…
A: Here we are required to find the Enthalpy change for the reaction.
Q: Consider this reaction: Mg + 2HCI --> H2 + MgCl2 How many moles of H2 will form from 26 moles of…
A: From the reaction it is clear that one mole of Mg reacts with two moles of HCl to form two moles of…
Q: Consider the following reaction. How many moles of oxygen are required to produce 4.00 moles of…
A: The given equation is already a balanced equation. Fr the equation we can infer that 6 moles of…
Q: 3. Calculate reaction AH°rxn and reaction AU°rxn for the reaction: TiCl4(1) + 2H2O(1) → TIO2(s) +…
A: Given reaction is : TiCl4 (l) + 2H2O (l) ------> TiO2 (s) + 4HCl (g) Calculate the standard…
Q: 9. BaCl (aq) + K,O (aq) BaO + KCl) (s) (aq) Balanced equation: Classification: 10. Mg + Og) -→ MgO,…
A: Balanced chemical reaction is the reaction in which elements are present in product are equal to the…
Q: Ipy of the reaction: N2 (g) + O2 (g) → 2NO (g), given the following reaction N2 (g) + 202 (g) → 2NO2…
A: Given : We have to calculate the enthalpy of given reaction.
Q: Consider the following reaction Ca(s) + 2HCI(aq) --> CaCl,(aq) + H2(8) How many moles of HCl are…
A: Calcium metal reacts with hydrogen chloride to form calcium chloride and hydrogen gas. The equation…
Q: Consider these reactions: Reaction 1: H2 (g) + Cl2 (9) → 2HC1(g) AH = -184.6 kJ Reaction 2: 20F2 (g)…
A: Reaction 2 is considered as exothermic reaction. Because, the delta H value is negative. That means…
Q: mol H2 react). g reactant and calculate the p Hrate of hydrogen n (kals) reou
A: The balanced reaction as follows: C2H2 + 2H2 → C2H6
Q: Aqueous hydrochloric acid (HCI) will react with solid sodium hydroxide (NaOH) to produce aqueous…
A: HCl (aq) + NaOH (s) ---> NaCl (aq) + H2O (l)
Q: Reactants Products Br,0) Reactants Products H30 Bal,(s) Reactants Products H20 Baso,(s)
A: Note: According to our guidelines we are supposed to answer only first three subparts. Kindly repost…
Q: Type of reaction? ___________________: Hydrochloric Acid and Potassium Carbonate ___HCl(aq) +___…
A: This reaction is acid base neutralisation reaction. here we have to balance the reaction. K2CO3aq +…
Q: A particular reaction was performed in a test tube. The student observed that the sides of the test…
A: We have to predict correct the reason for cooling of test tube.
Q: Consider these reactions, where M represents a generic metal.…
A:
Q: what type of reaction woudl the following equation represent?
A: Given :- KOH(aq) + HBr(aq) ––––> KBr(aq) + H2O(l) To determine :- Type of reaction
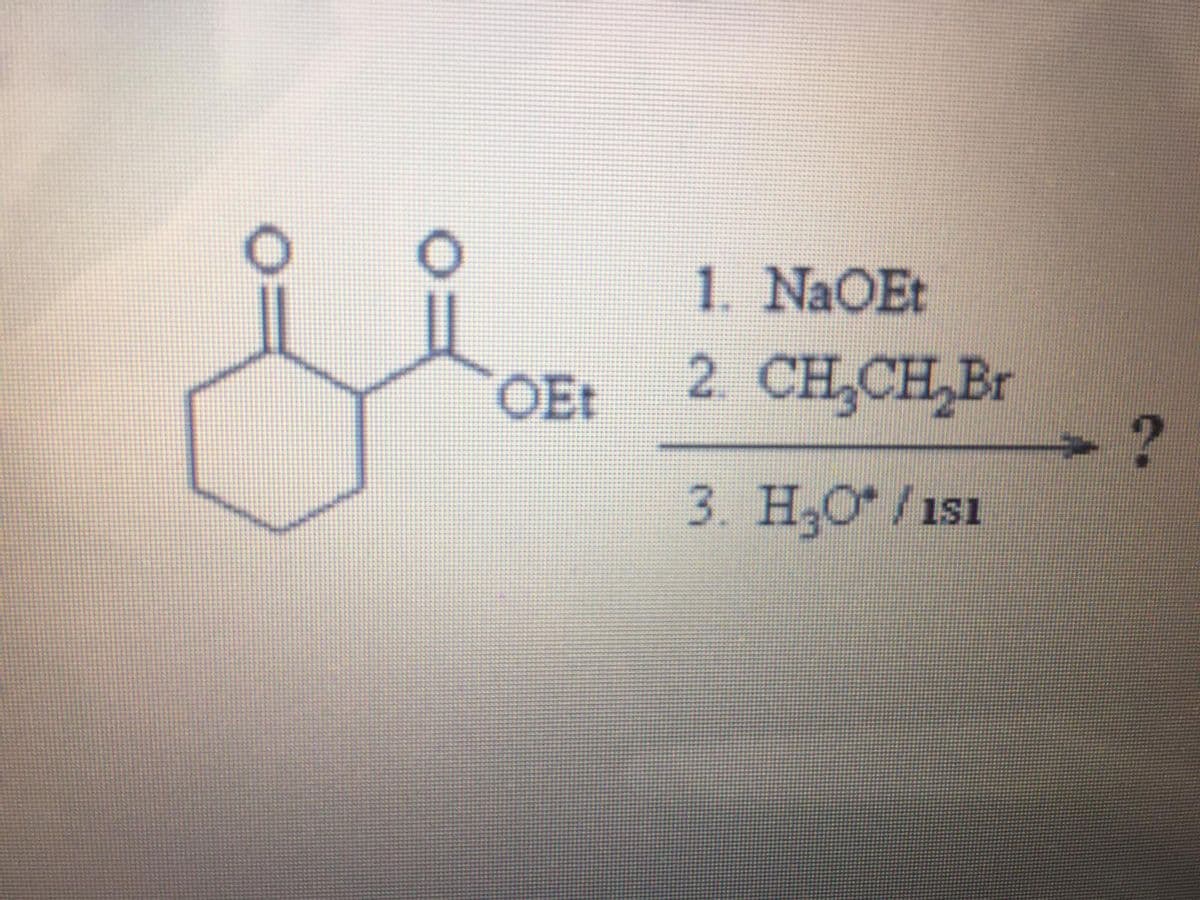
Q: COOH `NEt2 но (а) (b) ELOH, H* (а) NAOH, (d) (c) (f) HBr, H2O2 (d) `NEt2 H*, (f) (h) (g) NABH, (b)…
A:
Q: The chemical reaction 2 H2O + energy → 2 H2 + O2 is 1. an exothermic reaction. 2. a synthesis…
A: 1 Exothormic reaction :-The reaction in which energy releaseExample :- CS+O2g→CO2g+Energy heat2…
Q: Use the given information to determine the enthalpy of the reaction
A:
Q: 4. The reaction: 4 Al (s) + 3 02 (g) → 2 Al203 (s) ; AH° = -3351 kJ Is and therefore heat is by the…
A:
Q: The following diagram represents a high-temperaturereaction between CH4 and H2O. Based on this…
A: Limiting reagent is the reactant that limits the chemical reaction and determines the amount of the…
Q: Consider the following graph of the reaction 2 SO3(g) 2 2 SO2(g) + O2(g) AH = +197.8 kJ 60 503 502…
A: Actually when the volume of the system is decreased, the concentration of all the gas increases…
Q: Reaction 1: H₂(g) + Cl₂(g) → 2HCl(g) AH = -184.6 kJ Reaction 2: 20F2(g) O2(g) + 2 F2 (g) AH = -49.4…
A:
Q: Hlendmo 7. Writing and Balancing Reactions. Write and Balance the following reactions with the…
A:
Q: Consider these reactions: Reaction 1: H2(g) + Cl2(g) 2HC1(g) AH = –184.6 kJ Reaction 2: 20F2(g) →…
A: In given question we have three reactions and we need to tell which one of these reaction is…
Q: Consider a reaction between: AgClO4 and NaCl. A: NaClO4(s) B: AgCl(s) C: AgClO4 (s), Cl2(g) D: no…
A:
Q: (1/8)S8(s) + H2(g) →H2S(g) ΔHrxn = −20.2 kJ What is ΔH when 4.90 mol of S8 reacts?
A: Introduction For a chemical reaction, The amount of energy per mole released or produced at constant…
Q: Solu bility nles, leadl) indicde (P.blz) is an inseluble Comppard. Camplete the folloning reaction…
A: The chemical equations should be balanced in accordance with the law of conservation of mass. This…
Q: f) An reaction is one that gives off heat. g) The amount of energy required to raise the temperature…
A: " Since you have posted a question with multiple sub-parts , we will solve first three sub-parts for…
Q: Given the reaction 2 CO CO2(g) What is AHO of this reaction? AHO CO (g) = -111 kJ/mol AHO 2H₂ (g) =…
A:
what occurs as a result of the reaction
Step by step
Solved in 4 steps with 3 images

- 2N2O5-------> ?(A) 4NO2{g) + O2 (g)(B) 2NO2 + O6(C) N2O6 + O4how do you solve ______C10H14N2(s)+ ______O2(g)---->____CO2(g)+____H2O(l)+_____NO2(g)?A major component of honey is fructose (C6H12O6) with a molar mass of 180.16 g/mol. A tablespoon (15.0 mL) of honey is considered one serving. How many tablespoons must be eaten to consume 0.1700 moles of fructose? Assume honey is 100% fructose. The density of honey is 1.45 g/mL.
- H2S (g) + 3/2O2 (g) ----> H2O (g) + SO2 (g) ΔS° =______J/KCocaine(C17H21O4N) is a natural substance found in the leaves of the coca plant, which has been used for centuries as a local anesthetic and stimulant. Illegal cocaine arrives in the USA either as a pure compound or as the hydrochloride salt(C7H21O4NHCl). At 25 degrees celsius, the salt is very soluble in water(2.59kg/L), but pure cocaine is much less so(1.70g/L) a. What is the maximum amount of salt in grams that can dissolve in 50.0mL of water? b. When a 25mL aqueous solution that contains 62.50 g of the salt is treated with NaOH, the salt is converted to pure cocaine. How much water in liters is needed additionally to dissolve the formed pure cocaine,3. Please complete all parts to the following table:
- In one sentence explain what is wrong with them, and rewrite them so that the coefficients are correct. 2 Mg(OH)2(s)+4HCl(aq)->2MgCl2(aq)+4H2O(l)1. In the image name 3 molecular components of the molecule ( A-C) 2. Why are some of the atoms numbered as 1’ and some are not?What can be concluded from this table?
- 2SO2 (g) + O2 (g) ------> 2SO3 (g) ΔS° = _____J/KCalculate how many water molecules there are for 1000g of water, H2O. A. 3.34 x 1025 water molecules B. 6.02 x 1023 water molecules C. 3.34 x 10-26 water molecules D. 6.68 x 1025 water moleculesPlease, please explain the part I have circled in the pic--I don't understand why the mols of H2SO4 have to be multiplied by 2!

