1. Normality is the number of equivalents of solute per liter of solution. II. The equivalent weight is equal to the proportion of molecular weight and n, where n is the number of the number of replaceable H* and OH per molecule. Evaluate whether the statements above are correct or incorrect, and type the letter of your answer on the space provided. Write the capital letter corresponding to your choice. A- Only the first statement is correct. B- Only the second statement is correct. C- Both the first and the second statements are correct. D- Both the first and second statements are incorrect.
1. Normality is the number of equivalents of solute per liter of solution. II. The equivalent weight is equal to the proportion of molecular weight and n, where n is the number of the number of replaceable H* and OH per molecule. Evaluate whether the statements above are correct or incorrect, and type the letter of your answer on the space provided. Write the capital letter corresponding to your choice. A- Only the first statement is correct. B- Only the second statement is correct. C- Both the first and the second statements are correct. D- Both the first and second statements are incorrect.
Introductory Chemistry: An Active Learning Approach
6th Edition
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Mark S. Cracolice, Ed Peters
Chapter16: Solutions
Section: Chapter Questions
Problem 9E: What happens if you add a very small amount of solid salt (NaCl) to each beaker described below?...
Related questions
Question
6
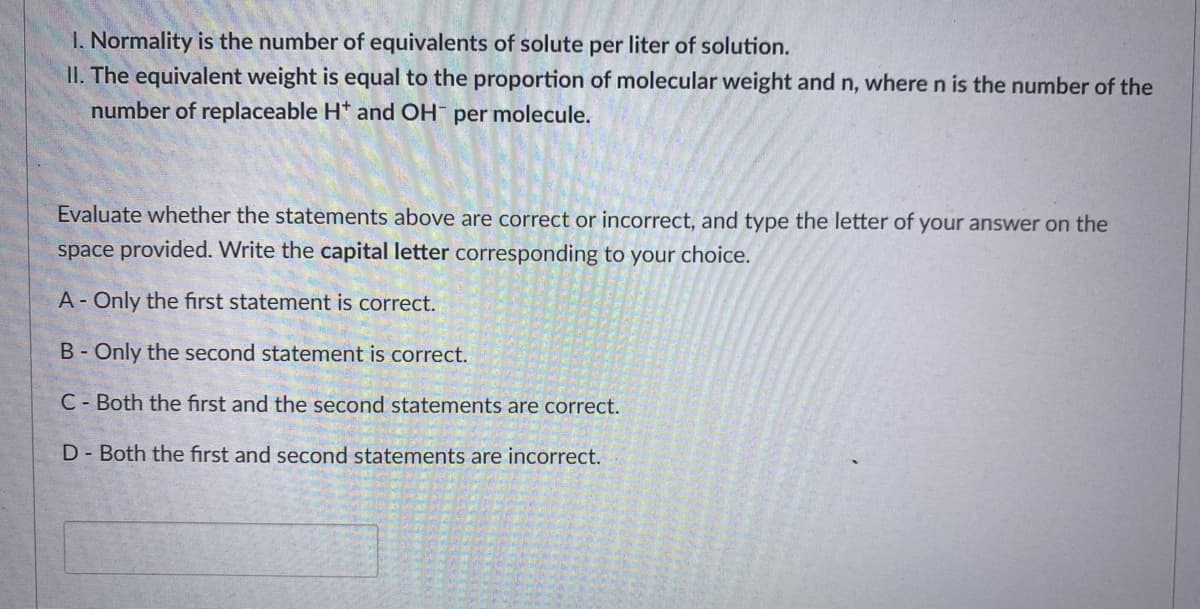
Transcribed Image Text:1. Normality is the number of equivalents of solute per liter of solution.
II. The equivalent weight is equal to the proportion of molecular weight and n, where n is the number of the
number of replaceable H* and OH per molecule.
Evaluate whether the statements above are correct or incorrect, and type the letter of your answer on the
space provided. Write the capital letter corresponding to your choice.
A- Only the first statement is correct.
B- Only the second statement is correct.
C- Both the first and the second statements are correct.
D- Both the first and second statements are incorrect.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT


Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning
