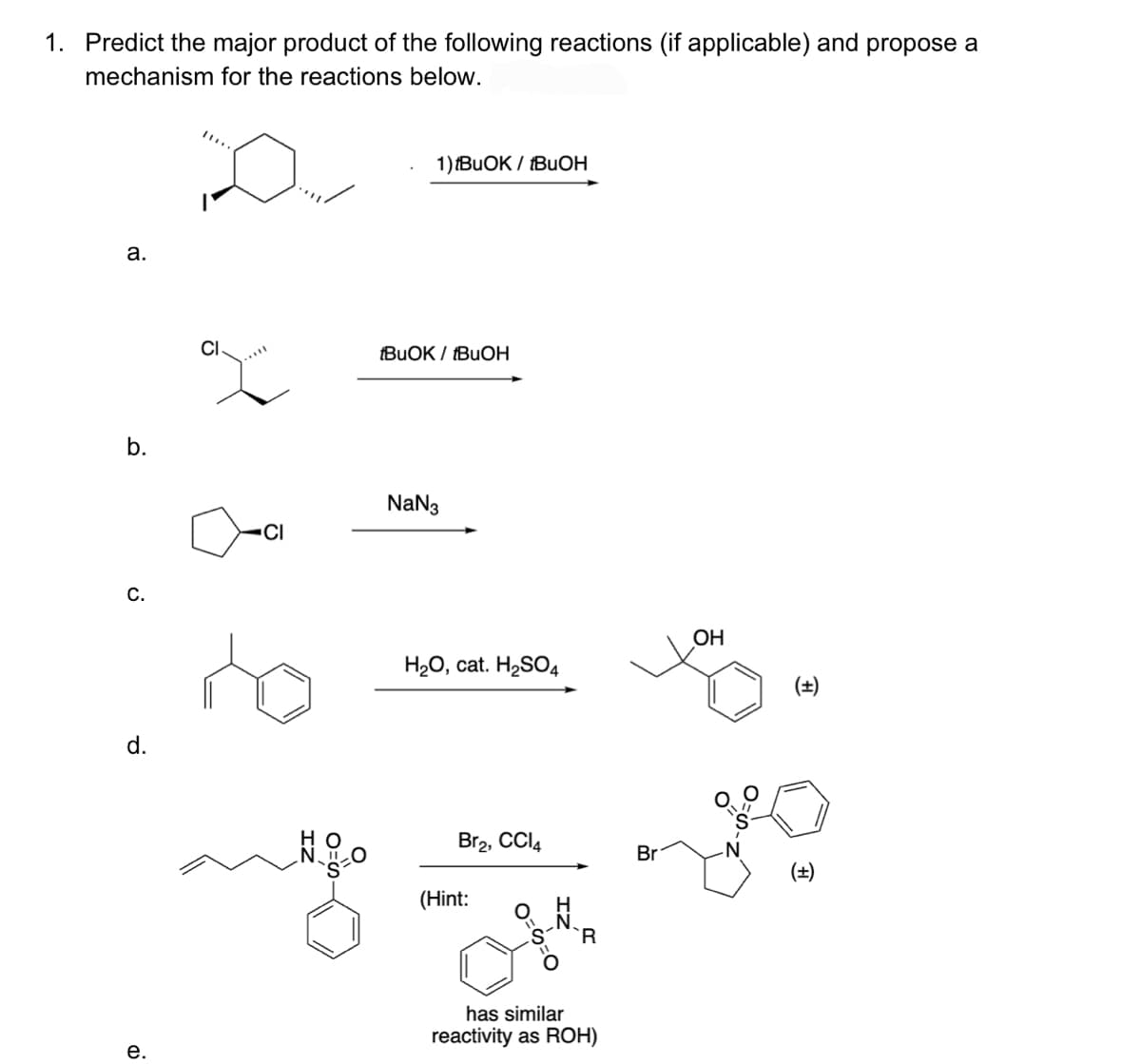
1. Predict the major product of the following reactions (if applicable) and propose a mechanism for the reactions below. a. b. C. d. e. CI CI IZ O 1) IBUOK / IBUOH tBuOK / tBuOH NaN3 H₂O, cat. H₂SO4 Br2, CCI4 (Hint: has similar reactivity as ROH) Br OH (±)
Q: Draw the structure of the product that results from the treatment of (E)-oct-4-ene with D2 in the…
A: The question is based on organic reactions.We need to identify the product and explain its…
Q: A student prepares aspirin using 2.56 grams of salicylic acid (MW = 138 g/mol) and 4.85 mL of acetic…
A: The chemical equation for the formation of aspirin is given below:
Q: Predict the product of each reaction. a) b) >==< HI (1 equiv.) 1. H₂, Lindlar's catalyst 2. RCO₂H 3.…
A: Organic Reactions are given.We have to identify the products formed in the reactions.
Q: Draw the product of the reaction shown below at physiological pH (pH = 7.4). Ignore inorganic…
A: While making peptide bonds, the amino group of an amino acid is protected by different groups to…
Q: Propose an efficient synthesis for the following transformation: The transformations above can be…
A: Conversation of alkene into aldehyde.find out suitable reagent
Q: Please explain and solve very correctly
A: The given amine is a tertiary amine. One of the largest alkyl groups attached to the nitrogen atom…
Q: What is the IUPAC name of the following compound? & a. (R)-1-chloro-3-methyl-2-cyclohexene b.…
A: The given compounds contain an alkene functional group. For the IUPAC name of an alkene, first of…
Q: The concentration of the hydronium ion in an aqueous solution at 25 °C is 0.0090 M. What is the…
A:
Q: Predict the products of the following biochemical reaction: CH,−O CH O CH2-O O O + 3 KOH P In…
A: Saponification is a process involving the hydrolysis of fats with its reaction with alkali, and…
Q: The total pressure in a tire is 1.80 atm at 23.0∘C. If the temperature is increased to 39.0∘C, and…
A:
Q: Consider the equilibrium A + 2B=AB2 and the two reaction systems shown below. The system on the left…
A: The direction in which the reaction proceeds to reach equilibrium can be predicted by determining…
Q: 1. In a gravimetric analysis of a chloride salt, the following data were obtained. Mass of original…
A: The objective of the question is to understand the impact of sunlight exposure on the results of a…
Q: What is/are the product(s) from the following reaction? 11. Br Br Br Br₂/CH₂Cl₂ O II and IV OI and…
A: Information of question
Q: 1. Zn | Zn²+(1.0 M) || Cu²+(1.0 M) | Cu 2. Zn | Zn²+(1.0 M) || Cu²+(0.1 M) | Cu 3. Zn | Zn²+ (0.1 M)…
A: “Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: Question 8: Select the letter that best identifies the major product of the reaction shown. HO H A.…
A: Na2CrO7 oxidises secondary alcohol to ketone and primary alcohol to carboxylic acid.
Q: Predict the product(s) for the reaction shown. OH + IV excess HI heat OH
A: Ether react with hydroiodic acid to form alcohols and corresponding haloalkanes.When present in…
Q: 15.) The following sequence of amino acids occurs in a globular protein and forms an a-helix. Would…
A: α-helices are a type of secondary structure in proteins where amino acids are arranged in a helical…
Q: A 870. mL solution was made by mixing 419. mL of hexane, which has a density of 0.66 g/mL, and 469.…
A: Given ,Volume of solution = 870. mLvolume of hexane = 419. mLvolume of toluene = 469. mL
Q: A certain reaction, 2A + BC, has a standard enthalpy of reaction of AH = -40.4 kJ/mol. What would be…
A: Given reaction with the value: ... (1)
Q: Circle the product in each of the following dehydration reactions. Can you also name the reactant…
A: We have to identify the product of the reactions.
Q: 2. Name, give, or draw the structural formula for each of the following coordination compounds using…
A: There are some rules for naming a coordination compound.The following are the rules :-Rule 1- While…
Q: stereochemistry when applicable. Â "Give the major organic product(s) of the following. Include e of…
A: As per the question given there are many subparts given and it is asked that first e should be…
Q: In ClF3 the two lone pairs of electrons can be placed one axial and one equatorial, both equatorial,…
A: The objective of the question is to determine the placement of the two lone pairs of electrons in…
Q: An aqueous solution containing both Sr2+ and CO32- ions is mixed in order to precipitate SrCO3(s).…
A: Given net ionic equation is Sr2+(aq) + CO32-(aq) ---------> SrCO3(s) Molarity of Sr2+ ions in the…
Q: 3) **The phase diagram for an unknown compound shown here. What is the approximate, normal melting…
A: A question based on phase diagram. The most appropriate choice among the 5 given options is…
Q: After performing a reaction a TLC plate was run to identify the product. Below is the completed TLC…
A: TLC is used to check reaction's progress.Let's assume we put three reagents as evident from lane A…
Q: Provide the correct molecular structure (3D structure with wedges and dashes) below before the…
A: The given is a ligand substitution reaction of coordination compounds.To find the original reactant…
Q: What is the expected product from the following reaction sequence? года OI Oll O III ONV OV Oi…
A: Find out product of reaction DIBAL - it is reducing agent. It reduces ester into aldehydeNaOEt act…
Q: 2. - Draw a detailed mechanism for the following reactions. You do not need to draw arrow steps for…
A: “Since you have asked multiple questions, we will solve the first question for you according to our…
Q: Solution X has a pH of 9.80, and solution Y has a pH of 7.2. a.) Which solution is more acidic?…
A:
Q: Along which line would a sample of pure X be a mixture of solid and gas? choose a letter Which is…
A:
Q: reaction H₂SO₂() + H₂O() → H₂O* (aq) + HSO₂ (aq) N₂(g) + 3H₂(g) → 2N H₂ (g) BaCO,(s) → BaO(s) +…
A: Entropy is a measure of the amount of energy in a system that is not available to do work. It is…
Q: 18. Which of the following is not a classical isostere for a chloride residue? A F A. A B. B C. C D.…
A: Two molecules or ions with the same number of atoms and the same number of valence electrons are…
Q: Draw the oxidative addition product formed in the first step of the Heck reaction. PdL₂+RX- The…
A: In oxidative addition step of Heck reaction, oxidation state as well as coordination number of…
Q: Give the major organic product(s) of the following. Include stereochemistry when applicable. 6 S of…
A: The question is based on organic reactions.We need to identify the product and explain its…
Q: Question 2: lodine monochloride (ICI) is an electrophilic halogenating agent with similar properties…
A: The given reaction is addition reaction to double bond but the catche is that there is addition of…
Q: Calculate Ecell at 298 K for the voltaic cell diagrammed below Cr(s) | Cr²+ (aq, 0.015 M) || Fe³+…
A:
Q: Br ||| Based on resonance and inductive effects, identify the strongest base among the aniline…
A: Aniline is basic in nature due to the presence of a lone pair of electrons on the nitrogen atom. But…
Q: Use the information in the ALEKS Data tab to sort the following chemical species by reducing power.…
A: Determine the order of reducing power.The tendency of the chemical species to reduce another element…
Q: How many grams of Co are there in a sample of Co that contains 4.58×1023 atoms? _____grams
A: Number of atoms=.58×1023 atomsFind out mass of Co
Q: Balance the following redox reaction in acidic solution. 2+ Zn(s) + O₂(g) Zn(aq) + H₂O(1) - 2+ Zn…
A:
Q: a. b. C. Write an IUPAC name for each of the following molecules: NH₂ u ye NH₂ OH HN á Br
A:
Q: How many different molecules are drawn below? 0 X Ś
A: To identify the different types of molecules, give the IUPAC names of all the given structures. If…
Q: CH3 (Et),NH H₂O* 1) LDA,-78ºC 2) Br Н OH H OH H3O* NaOH 25%
A: According to Bartleby Q&A guidelines we are supposed to answer only three sub parts of question.…
Q: Calculate the following quantity. 0.02123 m + 1.12 m + 0.00123 m 1.1424 m 1.14246 m 1.1 m 1.14 m…
A: The significant figure refers to particular digits in a number. According to the measurement rule,…
Q: + The pH of an acidic solution is 2.95. What is [H*]? 1 4 7 +/- 2 5 8 . M 3 6 9 0 C x 100
A: Given pH of acidic solution =2.95To find[H+]
Q: Draw the two chair conformations of β-D-galactopyranose and Determine which is the most stable..
A: Given compound is beta-D-galactose.Pyranose is six member cyclic structure of carbohydrate and beta…
Q: 4.25 kg of solution A (2.7% salt) is mixed with 2.96 kg of solution B (4.5% salt). What is the final…
A: Mass of solution A = 4.25 kg Mass percent of salt in solution A = 2.7% Mass of solution B = 2.96 kg…
Q: What is the major organic product from the reaction of 1-pentyne with 1 mole of HBr? Please type the…
A: The addition of two or more equivalents of HCl or HBr converts alkynes to geminal dihalides through…
Q: Indicate the number of signals of the following molecule in 1H-NMR and the multiplicity of eacu…
A: Proton-NMR spectroscopy is mainly used for the identification of the complete structure of an…
Step by step
Solved in 4 steps with 1 images

- Describe how the product is purified. 4,4'-DIBROMOBIPHENYL [Biphenyl, 4,4'-dibromo-] Submitted by Robert E. Buckles and Norris G. Wheeler1. Checked by R. S. Schreiber, Wm. Bradley Reid, Jr., and Robert W. Jackson. 1. Procedure In a 15-cm. evaporating dish is placed 15.4 g. (0.10 mole) of finely powdered biphenyl (Note 1). The dish is set on a porcelain rack in a 30-cm. desiccator with a 10-cm. evaporating dish under the rack containing 39 g. (12 ml., 0.24 mole) of bromine. The desiccator is closed, but a very small opening is provided for the escape of hydrogen bromide (Note 2). The biphenyl is left in contact with the bromine vapor for 8 hours (or overnight). The orange solid is then removed from the desiccator and allowed to stand in the air under a hood for at least 4 hours (Note 3). At this point, the product weighs about 30 g. and has a melting point in the neighborhood of 152°. The crude 4,4'-dibromobiphenyl is dissolved in 75 ml. of benzene, filtered, and cooled to 15°. The…(a) Illustrate the following name reactions giving suitable example in each case :(i) Clemmensen reduction (ii) Hell-Volhard-Zelinsky reaction(b) How are the following conversions carried out?(i) Ethylcyanide to ethanoic acid (ii) Butan-l-ol to butanoic acid(iii) Benzoic acid to m-bromobenzoic acidcan i get help drawing out actual structures including the nucleophilic addition of Cy2NH to parafomaldehyde and its hemiaminal intermidiate and the condensation step when it is displaced by terminal alkyne forming allene, also what is dioxane getting rid of as the solvent, thanks
- Can someone explain the answers to 28, 29, and 30? The answers are B, A, A. I understand lindar catalyst makes cis alkenes and Na/NH3 makes trans alkenes but I dont understand how the second step of Br2, OsO4 influences the stereochemistry between the 2 . Will rate quickly if helpful. Thanks!Suggest an explanation for the observed order of SN1 reactivity of the following compounds. Want solution ASApWhy cis-Ru(II)Cl2(DMSO)4 reacts with pyridine, et cetera, to give substitution of the DMSO but not the chloride ligands, but trans-Ru(II)Cl2(py)4 react with suitable Na+ and K+ salts in aqueous pyridine to afford chloride-substituted derivatives. write the reactions equations.
- EXPLAIN BY WORDS IN ORDER FOR ME TO UNDERSTAND THE GENERAL REACTION SCHEME. IN THIS CROSS-COUPLING REACTION WHICH ORGANOHALIDE (R-X) REACTS WITH ORGANOBORANE [R1B(OR)2].Explain the following terms, make sure to include chemical structures as examples. a.) Syn-addition b.)Zaitsev product c.)Concerted(relating to hydroborations or SN2 reactions)Please propose a synthesis of the target molecule using as many steps or reagents and answer these questions. 1. Why use chemoselectivity as the functional group and not another? 2. Why regioselectivity? 3. Why stereoselectivity? 4. What are the changes in the oxidation state?
- 1. In which of the following tests/reagents would cyclohexene and hexane both yield a positive result? I. t-BuCl in AlCl3 II. Br2 in light III. Halogenation by I2 in KI IV. KMnO4 2. Which of the following is statements is/are TRUE about the experiment on the relative rates of electrophilic aromatic substitution?I. The experiment must be performed in dark conditions. II. Using the same solvents, a faster decolorization will be observed in aniline as compared to ethylbenzene. III. Chlorobenzene will react faster than methoxybenzene. IV. The use of AlCl3 in the halogenation of aromatic compounds using elemental bromine could hasten the reaction.For butenafine, develop a synthesis that starts with napthalene and benzene. (aromatic methyl groups can be converted to alkyl bromides using Br2/light, and aldehydes by heating the alkyl bromide in DMSO, eg. ArCH3 to ArCH2Br then to ArCHO).Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides. One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(C2H5)3). 3SnCl4 + 4AI(C2H5)3 →3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.160 L of SnCl4 (d = 2.226 g/mL) was treated with 0.346 L of triethylaluminum (Al(C2H5)3): d = 0.835 g/mL). What is the theoretical yield in this experiment (mass of tetraethylstannane, Sn(C2H5)4)? If 0.257 L of tetraethylstannane (d= 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?



