Q: Take a look at this molecule, and then answer the questions in the table below it. CH₂OH CH₂OH H H H…
A: Step 1: Step 2:Step 3: Step 4:
Q: A small portion (2.22 mL) of ascorbic acid solution (0.2310 mg/L) was transferred to a volumetric…
A: We can calculate the concentration of the diluted ascorbic acid solution using the following…
Q: Which of the following would be the best method to make meta-bromopropylbenzene from benzene? a.)…
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: Solid silver acetate is slowly added to 50.0 mL of a 0.0620 M sodium sulfite solution. The…
A: The objective of the question is to find the concentration of silver ion required to just initiate…
Q: Without doing any calculations, match the following thermodynamic properties with their appropriate…
A: The objective of the question is to match the thermodynamic properties with their appropriate…
Q: Determine the pH of a solution by constructing a BCA table, constructing an ICE table, writing the…
A: From the ICE table we can now have the expression of Kb in order to determine the unknown. Kb =…
Q: Draw the Lewis structure and describe the bonding in Cr2(CH3COO)4(H2O)2 using MO theory. In the…
A: Step 1: To start, let's draw the Lewis structure for Cr2(CH3COO)4(H2O)2. Since we're using…
Q: 3. Calculate the Packing Factor of the Zinc Blenda structure to know the degree of occupancy of the…
A: ### Steps to Calculate the Packing Factor1. **Determine the Number of Atoms per Unit Cell:** - In…
Q: anode of an electrolytic cell must be connected to which terminal, positive or negative, of the…
A: The objective of the question is to understand the connection of anode in an electrolytic cell to…
Q: Question 5 0.2/0.2 pts Calculate the molar volume (V/n) of argon gas at a temperature of 298 K and a…
A: Given: T=298K;P=0.400atm;R=0.08206Latm/molK;V/n=???L/molStep 1: Write the ideal gas equation.PV=nRT…
Q: CONCEPT QUESTION 1. Retrosynthesis reaction. For the product given below, show the retrosynthesis…
A: Step 1:Step 2:The retrosynthesis and the forward synthesis of the target molecule are given. Step 3:…
Q: Give the complete common name, including anomer and stereochemistry labels, of the following…
A: Step 1: The given structures of monosaccharides are in cyclic form. First thing to do is to project…
Q: Question 10. Propose a synthesis for this compound. CH3 N H₂N-CH3 N-CH3 + H2O
A: Step 1: Step 2: Step 3: Step 4:
Q: Please correct answer and don't use hend raiting
A: Step 1: Step 2: Step 3: Step 4:
Q: Show calculations to prepare 100 mL of 0.1 M CH3CO₂H solution from the 1 M CH,CO solution. Table…
A: The objective of the question is to prepare a 0.1 M CH3CO2H solution from a 1 M CH3CO2H solution and…
Q: What is the molarity, M, of ions in a 0.60 M aqueous solution of sodium sulfate, Na2SO4? 0.30 M 0.20…
A: Step 1: SolutionGiven,molarity of sodium sulfate (Na2SO4) = 0.60 M we need to calculate the molarity…
Q: Draw the structure of all products of the mechanism below. Click and drag to start drawing a…
A:
Q: what did i do wrong here. , it seems to be marking it wrong but i thought it needed to be protonated
A:
Q: At the end of the experiment you will be assessing the purity of your aspirin by measuring its…
A: Step :
Q: An analytical chemist is titrating 247.8 mL of a 1.100M solution of acetic acid (HCH, CO₂) with a…
A:
Q: Cengage Learning OWLv2 | Online teaching and C…
A: The objective of this question is to calculate the entropy change for the system when 2.46 moles of…
Q: For the reaction 2CO(g) + 2NO(g) → 2CO2 (g) + N2 (9) AH°=-746.6 kJ and AS° = -198.0 J/K The standard…
A: Step 1: Step 2: Step 3: Step 4:
Q: (b) EtO 0 OEt 1) NaOEt, EtOH 2) 3) H3O*, heat Br
A: Step 1: Step 2: Step 3: Step 4:
Q: Does this method in the picture below resolve the components of the drug mixture well?
A: Based on the information provided in the method, the method appears to be well-optimized for…
Q: None
A:
Q: complete the following condensation reaction
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: The beautiful expert Hand written solution is not allowed.
A: Now ∆G = - RTlnK = - nFE⁰cellThus E⁰cell = RT lnK / nF = [8.314 × 298 × ln (5.65 ×…
Q: I need answer expert solutions please give me fast answer
A: Approach to solving the question: Detailed explanation:IUPAC rule: 1. Choose the longest continuous…
Q: (3 points) Using the dissociation constant(ka), calculate the equilibrium concentrations of Cd2+ and…
A: The complex ion Cd(CN)42- dissociates into its component ions when dissolved in aqueous solution:…
Q: 24. Water plays an important role in traditional ceramics shaping processes. With the help of a…
A: Step 1:The effect of water volume on the aggregate volume of clay-based ceramics is crucial in…
Q: Polymers can be cross-linked using a variety of reactions that create connections between different…
A: To cross-link the polystyrene derivative with terephthaloyl chloride and AlCl3, one repeat unit of…
Q: Disparlure, 7,8-cis-epoxy-2-methyloctadecane, is an attractant for the Gypsy Moth. Disparlurecan be…
A: Step 1: The objective is to provide the synthesis of the specified…
Q: Show the steps necessary to transform the acid on the left into the compound on the right. Be sure…
A: Answer:
Q: The correct lewis structure fir carbonate acid H2CO3
A: Step 1: Step 2: Step 3: Step 4:
Q: Using the aromacy rules determine if the following compounds A ,B are aromatic and explain why
A: Step 1The 1st one is antiaromatic because it has got 4 pie electron and due to huckel rule ie 4n+2…
Q: I need help with part b
A: 2. After adding 15 mL of KOH: At this point, 15 mL of 0.100 M KOH reacts with the barbituric acid.…
Q: Provide the reagents and solvents necessary to perform the indicated transformation. More than one…
A: Step 1: Step 2: Step 3: Step 4:
Q: help 10
A: Step 1: Sketch the pyridine's structure. The six-membered ring of pyridine is made up of one…
Q: 5- In the Wittig reaction, the lide reacts with an aldehyde to form an alkene. Which of the…
A: For the Wittig reaction stereochemical outcomes:- Stabilized ylides, which are usually formed from…
Q: Use the data given in the table to calculate the value of AGxn at 25° C for the reaction described…
A: For #1: The Gibbs free energy change (ΔG0) of reaction can be calculated using the Gibbs free…
Q: None
A: Please see the attached image for the solution.If there are queries please do not hesitate to ask.…
Q: Can you explain the mechanism for this question HO OH H2SO4 cat. H₂O
A:
Q: • Provide conditions for one of these transformations OCH3 a) LOCH3 H3CO H3CO SO3H OCH3 b) OCH3 H3CO…
A: 1) Electrophilic aromatic substitution reaction in which methoxy groups are ortho, para directing…
Q: Can you please answer this question with steps. Thanks.
A: The objective of the first part of the question is to determine which ligand, X or Y, is the…
Q: By the action of a solution of hydrogen chloride in nitromethane, 3-methybut-1 - ene gives a mixture…
A: The question is asking to explain the results of the reaction of 3-methylbut-1-ene and…
Q: For the following equilibrium reaction, determine the expression for the concentration of NO,Cl(g)…
A:
Q: why is it wrong?
A: Step 1: Step 2:Mechanism Step 3: Step 4:
Q: (7 points) Calculate the silver ion concentration, [Ag*], of a solution prepared by dissolving 8.25…
A: Step 1:To find the silver ion concentration [Ag+],in the solution after dissolving 8.15g of AgNO3…
Q: Which product is most likely to form in the reaction series depicted below? Br O₂N 1. HNO3. H2SO4 2.…
A: Step 1: Step 2: Step 3: Step 4:
Q: Payalben
A: The objective of this question is to calculate the concentration of Ag+ in a voltaic cell using the…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

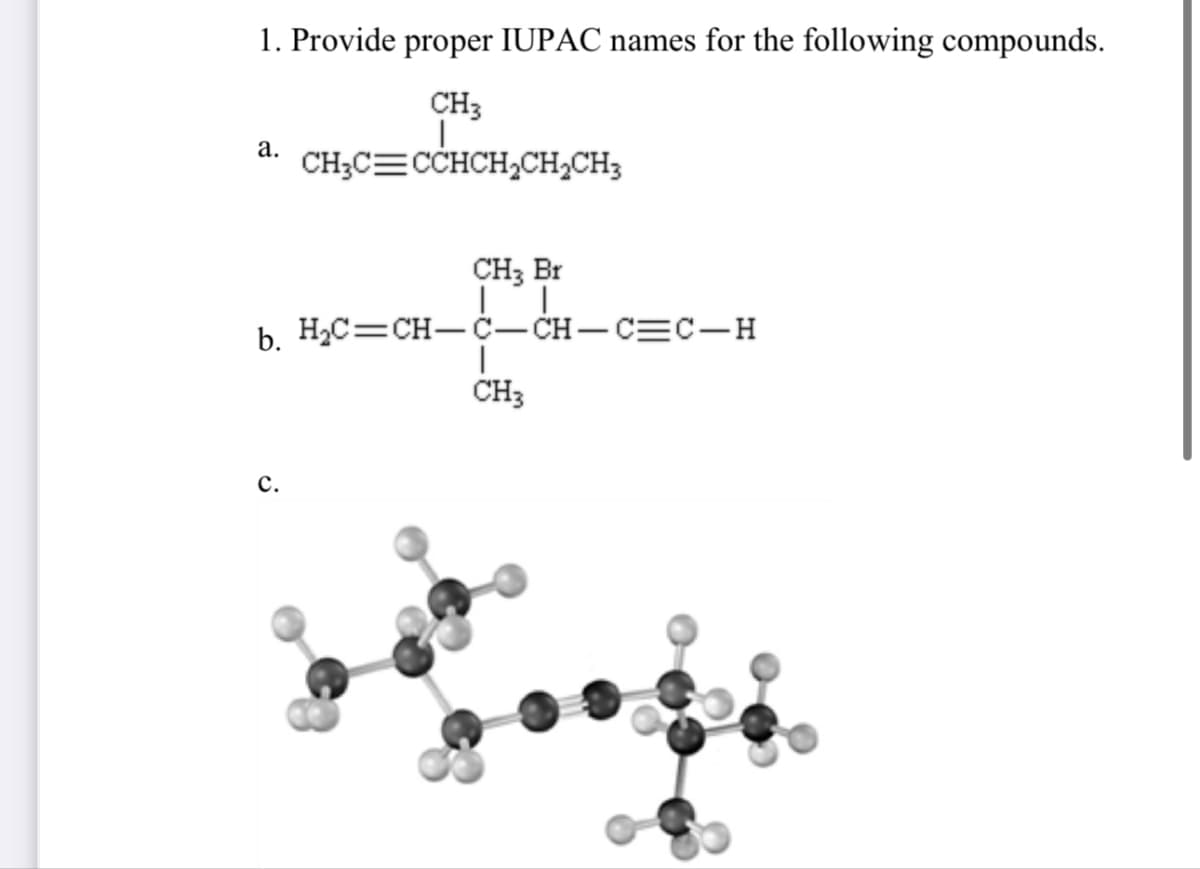
- what is the IUPAC name for this? step by step plsCH3-CH-O+CH3MgClIn the next chapter we'll look at cycloalkanes—saturated cyclic hydrocarbons—and we’ll see that the molecules generally adopt puckered, nonplanar conformations. Cyclohexane, for instance, has a puckered shape like a lounge chair rather than a flat shape. Why?
- 3. a. What is the chemical structure of benzoic acid, circle functional groups different than alkane,alkene, alkyne? b. Is it polar or nonpolar? _______________________ c. What is its water solubility in g/L? __________________________here's the reaction to be used for reference: C2H4 + H2 --> C2H6 1. What type of reaction is involved in C2H4 + H2 --> C2H6? 2. What is C2H4 in the reaction? 3. What is the name of the product in the reaction? a. ethane b. ethene c. ethyneWrite the IUPAC name of the following: CH3— CH2 — CHO
- Draw all constitutional isomers formed by monochlorination of each alkane.The skeletal line formula for a branched alkene is shown below. (i) What is the molecular formula of this compound? (ii) How many carbon atoms are in the longest chain, ignoring the double bond? (iii) What is the longest chain incorporating both carbons of the double bond? (iv) How many substituents are on this chain? (v) Give the IUPAC name for this compound. [6]Draw all constitutional isomers formed by monochlorination of each alkane with Cl2 and hv.

