Q: The vapor pressures of CCl4 (A) and C₂HC 3 (B) between T = 350 and 360 K, can be determined…
A: The objective of the question is to calculate-The mole fraction of in Liquid PhaseThe mole fraction…
Q: You want to determine Fe in a solution via manganometric titration. To determine the exact…
A: Relevant Reaction Equations:Standardization Reaction:Balanced equation: Na2C2O4 (aq) + 2KMnO4 (aq) +…
Q: Draw the major product of this reaction. Ignore inorganic byproducts. Drawing SOCl2 о OH Q
A: Please comment down for any doubt. I hope my answer helps you.
Q: An analytical chemist is titrating 228.8 mL of a 0.6100M solution of hydrazoic acid (HN3) with a…
A:
Q: Does this method in the picture below resolve the components of the drug mixture well of a paper…
A: Gas Chromatography/Mass Spectrometry (GC/MS) and US EPA Method 8081 for Chlorinated Pesticides (GC)…
Q: Classify each of the following cycloadditions and explain why orbital symmetry conservation rules…
A: The objective of the question is to classify cycloaddition reactions and explain why orbital…
Q: Based on the data shown in the figure on the side, knowing that the nominal volumes for the flask,…
A: The objective of the question is to determine the most accurate analytical instrument from the given…
Q: Predicting qualitatively how entropy changes with mixing and separation For each system listed in…
A: Entropy is the measure of disorder of a system. It depends on volume, temperature, pressure. 1)…
Q: None
A: Note that if there were acetal present with oxygen atoms in place of the sulphur, then the…
Q: Starting from cyclohexanone, show how to make the two given products, include conditions to use in…
A:
Q: I completed an experiment "Iron contenr of fruit and vegetable through complex ion formation" How…
A: The number of moles of a substance is the amount of a substance in which there are 6.022×1023…
Q: Decrease volume Add Heat Left Left END Left Remove HCl(aq) Right Right Left Right Decrease pressure…
A:
Q: I need expert Drow the mechanism answer solutions
A: Step 1: Explanation: Carbonyl compounds react with primary amines to give us imines.Carbonyl…
Q: 4. Thermodynamic data for C(graphite) and C(diamond) at 298 K is given in the table below. 6) Will…
A: The objective of the question is to calculate the change in enthalpy (ΔH°), entropy (ΔS°) and Gibbs…
Q: (i) What is the difference among 1, 2, and 3?
A: The dissociation constant is the equilibrium constant for the dissociation of the complex. The…
Q: Based on your ICE table (Part 1) and the definition of Ka, set up the expression for Ka in order to…
A:
Q: - 3 J ☐ Counted 4 possible signals in 2-pentanone 2 3 Counted 3 possible signals in xylene Noted…
A: Step 1: Explanation: Number of signals = number of different types of H-atoms.H-atoms in different…
Q: None
A:
Q: Which gas will diffuse the FASTEST under the same conditions? CO C3H8 Cl2 H₂S
A: Step1: The rate of diffusion of a gas is inversely proportional to the square root of its molar…
Q: ) Give the major organic product(s 2. CH CH₂OH, H
A:
Q: Q-1: A mixture of ideal gases possesses the properties listed in table below whe examined at 2 bar…
A: The objective of this question is to determine the composition and properties of a gas mixture,…
Q: help 12
A: Step 1: The hydroxide ion (OH-) from LiOH attacks the carbonyl carbon atom of the starting ester…
Q: how can i plot this data with a regression line and including r^2 term?
A: Approach to solving the question:Here's the break down of solution step by step:Import Necessary…
Q: Table NU.1 lists some sources of household background radiation, all of which emit gamma radiation…
A: The objective of the question is to write nuclear equations for the decay of various radioactive…
Q: The IUPAC name for the compound shown is Save for Later Submit Answer
A:
Q: Propose an efficient synthesis for the following transformation: Br The transformation above can be…
A: Step 1: Step 2: Step 3: Step 4:
Q: Ascorbic acid, H2C6H6O6, is a diprotic acid usually known as vitamin C. For this acid, pKa1 is 4.10…
A: The objective of the question is to find the molar concentration of ascorbic acid in the solution…
Q: Based on your ICE table (Part 1) and the definition of Kb, set up the expression for Kb in order to…
A:
Q: Mechanism Ke KOH, CH3CH2OH reflux 0: (This proton is Acidic Hence of (from KOH) which is a strong…
A: The objective of the question is to understand the mechanism of the reaction between KOH (Potassium…
Q: None
A: ### Calculating pH1. **Write the equilibrium expression for hydrolysis:** The equilibrium constant…
Q: 22.35 Propose an efficient synthesis for each of the following compounds using the malonic ester…
A: The methylene protons of diethyl malonate (DEM) are sufficiently acidic. A base NaOEt is used in…
Q: Claisen Schmidt Condensation Unknown Product C19H180 Unknown IR and NMR Spectrums 100 50 D 4000 3000…
A: The objective of the question is to determine the product of a Claisen Schmidt Condensation reaction…
Q: This question has multiple parts. Work all the parts to get the most points. For the reaction below…
A: Step 1:In this reaction, aldehyde and alcohol are reacting When an alcohol reacts with an aldehyde,…
Q: Experiment4: Solubility Product In the experiment to determine the solubility of KI04, in water,…
A: The objective of the question is to calculate the for the reaction given.
Q: None
A: Step 1:Proton NMR spectroscopy gives information about types of protons their signals, splitting…
Q: None
A: Reaction: Answer:
Q: The van der Waals equation of state was designed to predict the relationship between pressure p,…
A: Step :
Q: An aqueous solution contains 0.25 M HF and 0.20 M NaF . The pKa for HF is 3.45. What is the pH of…
A: The objective of this question is to calculate the pH of an aqueous solution containing 0.25 M HF…
Q: Sn2 and N (starting concentration 10 mol/L) should be separated by fractionated precipitation using…
A:
Q: Rank the following correlation coefficients (r) in order of their strength from strongest to…
A: The Correlation takes values between -1 to 1 both inclusive. Now the more the value of Correlation…
Q: None
A: Step 1: Step 2: Step 3: Step 4:
Q: Calculate the pH of solutions of the following weak acids. This one is bit tricky. Think carefully…
A: Calculate the pH of a solution of a weak acid, using the dissociation of acid and equilibrium…
Q: Calculate the volume in of a 0.606M copper(II) sulfate solution that contains 500. mmol of copper 2…
A: The objective of this question is to calculate the volume of a 0.606M copper(II) sulfate solution…
Q: By titration, it is found that 62.5 mL of 0.176 M NaOH(aq) is needed to neutralize 25.0 mL of…
A: Step 1:To find the concentration of the HCl solution, we can use the equation for neutralization…
Q: A 1.00 liter solution contains 0.45M hydrocyanic acid and 0.58M sodium cyanide. If 0.15 moles of…
A: The objective of the question is to determine the truthfulness of the given statements after the…
Q: Which of the following carbocations would you expect to rearrange? If you expect rearrangement, draw…
A: Step 1: Step 2: Step 3: Step 4:
Q: Use acid-base titration to determine the concentration of: – A strong acid: HCl
A: The pH of an acid varies when a base is added to it. At the equivalence point, a sudden change in pH…
Q: P3.1: In the atmosphere, molecular oxygen is photochemically decomposed as follows: Oz + hư →…
A: “Since you have posted a question with multiplesub-parts, we will provide the solution only to the…
Q: Select the best answer for the product of the reaction below. င်း H a.) d.) 1.) HCI, EtOH COཉྫEt 2.)…
A: Option E: This option is correctSolution:
Q: The rate constant for this first-order reaction is 0.720 s -1 at 400 °C. A → products How long, in…
A: Detailed explanation:Given the first-order rate law, the time t can be calculated using the…
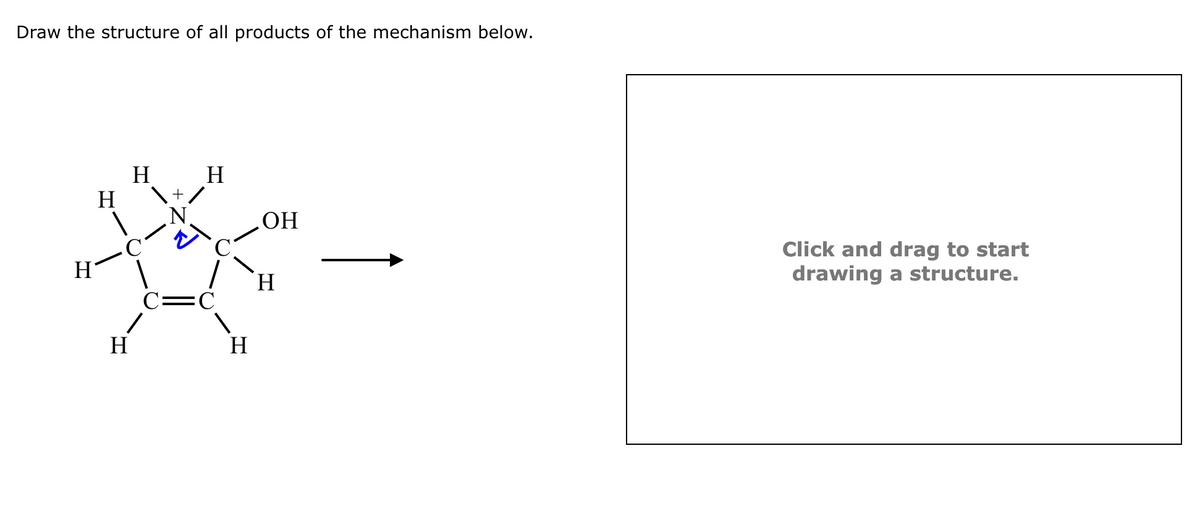
Draw the structure of all products of the mechanism below. Click and drag to start drawing a structure.
Step by step
Solved in 2 steps with 1 images

- Draw the Mechanism of Ether Cleavage in Strong Acid—(CH3)3COCH3 + HI → (CH3)3CI + CH3I + H2OUse Newman’s projection to predict the major product for an E2 reaction of the substrate shown below, and also draw out how the final product will look liDraw the mechanism ffrom benzaldehyde to this using: i)NaBH4 ii)TsCl, py iii)NaCN iiii)H+, H2O
- Show me the mechanism of this reaction using only 1 bromination. *R listed is NHCOCH3.In the box to the left of each reaction below, write the mechanism by which it occurs (could be SN1, SN2, or E1, or even 2 of them). Then draw the product(s).From each pair, select the stronger nucleophile. Q.) CH3OCH3 or CH3SCH3
- Supply the missing reagent in the following reaction: (see attachment) A. Lindlar, H2, mCPBA B. O3 or KMnO4 C. fused KOH D. NH3 E. NaNH2Provide complete mechanism of but-2-yne with HCl. Which is the best representation?propose a mechanism for this transformation. pay attention to the labelled carbon.

