1. This question is about nuclear chemisty. a) What is the age of a mummy, given a 14C activity of 8.5 min-1 g-1, The activity just before the 14C equilibrium was destroyed was 15 dis min-1 g-1 and the half-life of 14C is 5730 y. b) What should be the current activity, in dis min-1 g-1 of a wooden object believed to be 1100 years old? The activity just before the 14C equilibrium was destroyed was 15 dis min-1 g-1 and the half-life of 14C is 5730 y. c) What is the energy, in joules, associated with the α decay of 238U? (equation in picture
1. This question is about nuclear chemisty. a) What is the age of a mummy, given a 14C activity of 8.5 min-1 g-1, The activity just before the 14C equilibrium was destroyed was 15 dis min-1 g-1 and the half-life of 14C is 5730 y. b) What should be the current activity, in dis min-1 g-1 of a wooden object believed to be 1100 years old? The activity just before the 14C equilibrium was destroyed was 15 dis min-1 g-1 and the half-life of 14C is 5730 y. c) What is the energy, in joules, associated with the α decay of 238U? (equation in picture
Chapter3: Mechanisms
Section: Chapter Questions
Problem 107EQ
Related questions
Question
1. This question is about nuclear chemisty.
a) What is the age of a mummy, given a 14C activity of 8.5 min-1 g-1, The activity just before the 14C equilibrium was destroyed was 15 dis min-1 g-1 and the half-life of 14C is 5730 y.
b) What should be the current activity, in dis min-1 g-1 of a wooden object believed to be 1100 years old? The activity just before the 14C equilibrium was destroyed was 15 dis min-1 g-1 and the half-life of 14C is 5730 y.
c) What is the energy, in joules, associated with the α decay of 238U? (equation in picture)
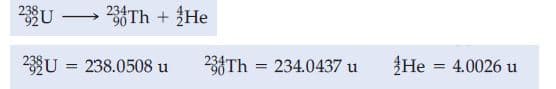
Transcribed Image Text:238
234Th + He
90
23U = 238.0508 u
234Th = 234.0437 u
He = 4.0026 u
%3D
%3D
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 6 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning


Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning