1. Using the solute formulas provided, convert grams of solute to moles of solute. For the hydrated salts be sure to include the waters of hydration in the molar mass. 2. It is reasonable to assume that the total mass of solvent includes the mass of both the water and the ice. If you used exactly 50.0 g of each, then you would have used 100.0 g or 0.100 kg of solvent. Use the actual masses of water and ice recorded in your lab notebook to calculate the mass of solvent. 3. Calculate the molality of the solution using the moles of solute (from Step 1) and the mass of solvent (from Step 2): moles of solute mass of solvent, kg molality, m =
1. Using the solute formulas provided, convert grams of solute to moles of solute. For the hydrated salts be sure to include the waters of hydration in the molar mass. 2. It is reasonable to assume that the total mass of solvent includes the mass of both the water and the ice. If you used exactly 50.0 g of each, then you would have used 100.0 g or 0.100 kg of solvent. Use the actual masses of water and ice recorded in your lab notebook to calculate the mass of solvent. 3. Calculate the molality of the solution using the moles of solute (from Step 1) and the mass of solvent (from Step 2): moles of solute mass of solvent, kg molality, m =
Macroscale and Microscale Organic Experiments
7th Edition
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Kenneth L. Williamson, Katherine M. Masters
Chapter6: Steam Distillation, Vacuum Distillation, And Sublimation
Section: Chapter Questions
Problem 6Q: A mixture of toluene (bp110.8C) and water is steam distilled. Visual inspection of the distillate...
Related questions
Question
The initial temperature for all 3 solutions was 0.9 degrees C.
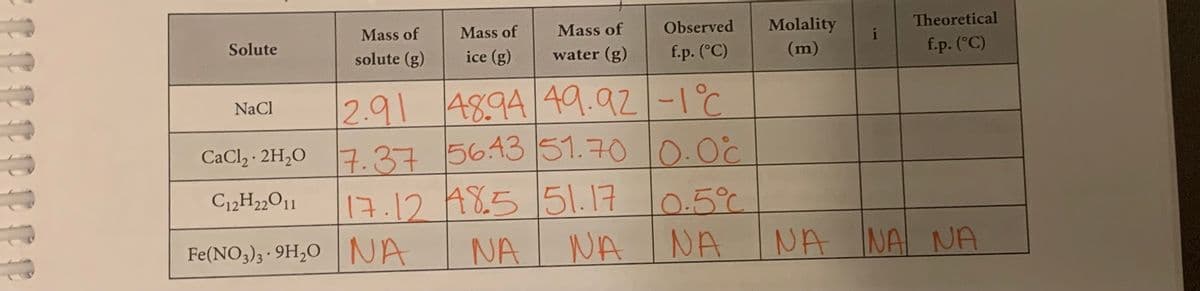
Transcribed Image Text:Observed
Molality
i
Theoretical
Mass of
Mass of
Mass of
Solute
solute (g)
ice (g)
water (g)
f.p. (°C)
(m)
f.p. (°C)
4894 49.92-1°C
2.91
CaClz - 2H,0 0.00
NaCl
7.37564351.70
17.12 A8.5 51.17
NA INA
C12H2„O11
0.5°C
Fe(NO,)3 · 9H,O|NA
NA
NA NA NA

Transcribed Image Text:Calculations
1. Using the solute formulas provided, convert grams of solute to moles of solute. For the hydrated
salts be sure to include the waters of hydration in the molar mass.
2. It is reasonable to assume that the total mass of solvent includes the mass of both the water
and the ice. If you used exactly 50.0 g of each, then you would have used 100.0 g or 0.100 kg of
solvent. Use the actual masses of water and ice recorded in your lab notebook to calculate the
mass of solvent.
3. Calculate the molality of the solution using the moles of solute (from Step 1) and the mass of
solvent (from Step 2):
moles of solute
mass of solvent, kg
molality, m =
4. Determine the value for the van't Hoff factor (i) for each of your solutes. For molecular solutes
the value is i = 1. For ionic solutes, you can determine the value of i by examining the molecular
formula. The value will be a small whole number; partial ionization is not expected, and waters
of hydration are not included.
5. Calculate the theoretical freezing point depression for each of the solutions in °C. The k, for
water is 1.86°C/m.
6. Calculate the theoretical freezing point of each solution, and compare it with the
experimentally observed freezing point for your solutes.
7. Comment in your lab notebook about the relationship between the observed freezing points
and the number of particles in solution (i) for each solute.
14
Colligative Pr o perties of Solutions- Freezing Point Depression | Labor atory 2
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Recommended textbooks for you

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole

Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole

Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning