1. Which of the following energy flow dia- gram correctly describes a heat engine that op- erates by taking one mole of a diatomic ideal gas through the cycle 1-2-3-4-1 as shown in the pV diagram below. Notr: 1+2 nd 3+4 (7, = 600K) diabatie processes (T.- 400X) (7= 300K) 1 (T, - 200K) Volume (m') A. В. с. D. Hot Reservoir Hot Reservoir Hot Reservoir Hot Reservoir 6.24k 6.24 k 8.73 8.73 5.82 k 2.08 k 4.16 k 2.91 k 4.16k 2.08 2.91 k 5.82 k Cold Reservoir Cold Reservoir Cold Reservoir Cold Reservoir 2. The figure shows a cyclic process where a diatomic ideal gas undergoes an isochoric pro- cess from a to b, an isobaric process from 6 to c, an iso- choric process from e to d and finally another isobaric process from d back to a. Let p = 2po. V = 2V. What is the efficiency of the cycle? Vo. Po A. 75.0% В. 37.1% C. 15.4% Volume D. 10.5% Pressure
1. Which of the following energy flow dia- gram correctly describes a heat engine that op- erates by taking one mole of a diatomic ideal gas through the cycle 1-2-3-4-1 as shown in the pV diagram below. Notr: 1+2 nd 3+4 (7, = 600K) diabatie processes (T.- 400X) (7= 300K) 1 (T, - 200K) Volume (m') A. В. с. D. Hot Reservoir Hot Reservoir Hot Reservoir Hot Reservoir 6.24k 6.24 k 8.73 8.73 5.82 k 2.08 k 4.16 k 2.91 k 4.16k 2.08 2.91 k 5.82 k Cold Reservoir Cold Reservoir Cold Reservoir Cold Reservoir 2. The figure shows a cyclic process where a diatomic ideal gas undergoes an isochoric pro- cess from a to b, an isobaric process from 6 to c, an iso- choric process from e to d and finally another isobaric process from d back to a. Let p = 2po. V = 2V. What is the efficiency of the cycle? Vo. Po A. 75.0% В. 37.1% C. 15.4% Volume D. 10.5% Pressure
College Physics
1st Edition
ISBN:9781938168000
Author:Paul Peter Urone, Roger Hinrichs
Publisher:Paul Peter Urone, Roger Hinrichs
Chapter15: Thermodynamics
Section: Chapter Questions
Problem 13CQ: Which cyclical process represented by the two closed loops, ABCFA end ABDEA, on the PV diagram in...
Related questions
Question
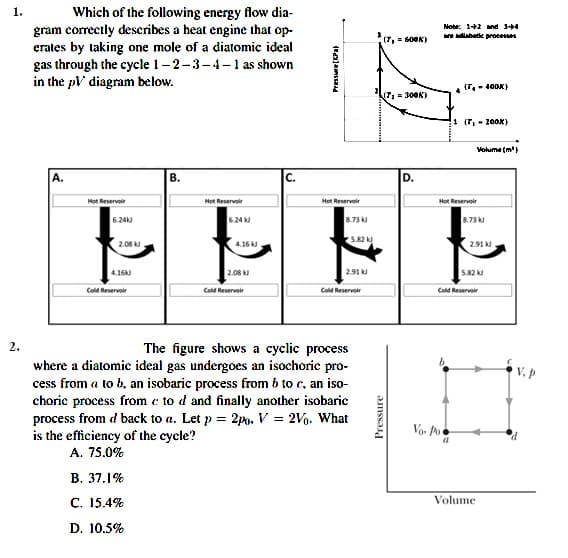
Transcribed Image Text:1.
Which of the following energy flow dia-
gram correctly describes a heat engine that op-
erates by taking one mole of a diatomic ideal
gas through the cycle 1-2-3-4-1 as shown
in the pV diagram below.
Note: 1+2 nd 3+4
diabatie processes
(T, = 60OK)
(T,- 400K)
7, = 300K)
1 (T, - 200K)
Volume (m')
A.
В.
C.
D.
Hot Reservoir
Hot Reservoir
Hot Reservoir
Hot Reservoir
6.24k
6.24 k
8.73
8.73
5.82 k
2.08
4.16 k
2.91 k.
4.16k
2.08
2.91 k
5.82 k
Cold Reservoir
Cold Reservoir
Cold Reservoir
Cold Reservoir
2.
The figure shows a cyclic process
where a diatomic ideal gas undergoes an isochoric pro-
cess from a to b, an isobaric process from 6 to c, an iso-
choric process from e to d and finally another isobaric
process from d back to a. Let p = 2po. V = 2V. What
is the efficiency of the cycle?
V. p
Vo. Po
A. 75.0%
B. 37.1%
C. 15.4%
Volume
D. 10.5%
Pressure
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Recommended textbooks for you

College Physics
Physics
ISBN:
9781938168000
Author:
Paul Peter Urone, Roger Hinrichs
Publisher:
OpenStax College


Physics for Scientists and Engineers, Technology …
Physics
ISBN:
9781305116399
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

College Physics
Physics
ISBN:
9781938168000
Author:
Paul Peter Urone, Roger Hinrichs
Publisher:
OpenStax College


Physics for Scientists and Engineers, Technology …
Physics
ISBN:
9781305116399
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

Principles of Physics: A Calculus-Based Text
Physics
ISBN:
9781133104261
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

College Physics
Physics
ISBN:
9781305952300
Author:
Raymond A. Serway, Chris Vuille
Publisher:
Cengage Learning

College Physics
Physics
ISBN:
9781285737027
Author:
Raymond A. Serway, Chris Vuille
Publisher:
Cengage Learning