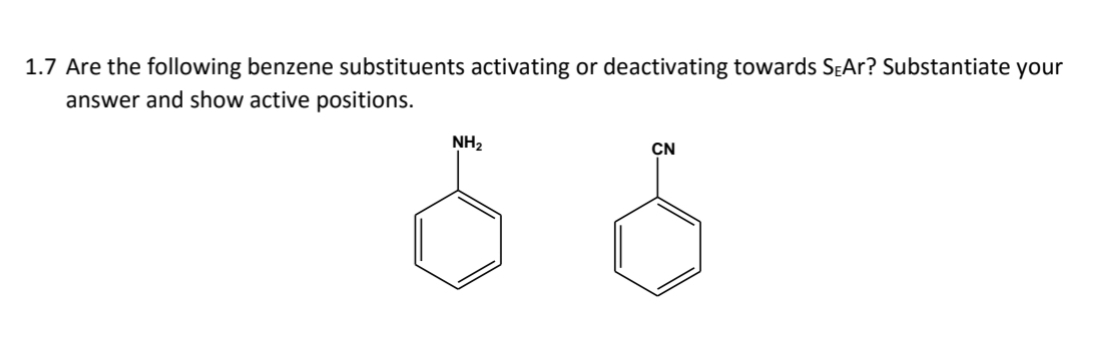
1.7 Are the following benzene substituents activating or deactivating towards SEAr? Substantiate your answer and show active positions. NH₂ CN 6 6
Q: a. How many grams are needed to prepare a 1 L stock solution of 10.0 M (Fe(II)) b. Using the…
A: The molarity of a solution is equal to the number of moles of solute present in 1 L of a solution.…
Q: O₂(g) + 4H*(aq) + 4e → 2H₂O Fe²+ (aq) + 2e → Fe(s) E₂ +1.229 V -0.440 V
A:
Q: The specific rotation of L-dopa in water is -39.5. The specific rotation of a solution of L-dopa and…
A:
Q: A compound is 80.0% carbon and 20.0% hydrogen by mass. Assume a 100.-g sample of this compound.
A: Stoichiometry allows us to make predictions about the outcomes of chemical reactions. Making useful…
Q: Calculate the percent by mass of water in the following compounds. (a) Cr(NO3)3 • 9H2O (b) AsO2 •…
A: Here we have to calculate the percentage by mass of water present in the following given compounds.
Q: Compare the boiling points of the various isomeric hydrocarbons shown in the table below. Notice the…
A: The boiling point of an alkane depends on the surface area of the molecule. So, higher the surface…
Q: If experimental determinations of Kp at different temperatures for a given equilibrium in the…
A: Given: ln Kp = a T-2 + c ln(T) and a = 123.5 K2 c = 23.6 To Calculate: The value of ΔHº and ΔSº at…
Q: When two gases are mixed, the pressure of the mixture is: O given by Gay-Lussac's Law of Combining…
A: Matter have generally three phase, i)Solid state ii). Liquid State iii) Gas state Gas : Gas is the…
Q: Module 5 Homework 1. Find the mole fraction of sodium acetate when 60g of sodium acetate are…
A:
Q: for each of the molecules below, indicate the pH range where the molecule would be ionized. لامارة…
A: Ph range depends upon the Type of Group present in the compound.
Q: 2. Complete the table by determining the molecular formula of each compound. Empirical Formula…
A:
Q: Relative Intensity 100 80 60 40 20- 0 10 20 30 40 50 60 m/z 70 80 90 100 110
A:
Q: Hydrogen peroxide, H₂O₂ (aq), decomposes to H₂0 (1) and O₂ (g) in a reaction that is first order in…
A: Given: The decomposition of H2O2 is first order in H2O2. Rate constant, k = 1.06 × 10-3 min-1 We…
Q: Problem 3.28 Label the electrophilic and nucleophilic sites in each molecule. a. -Br b. C. H H
A:
Q: compound will be more likely to exist as a hydr
A: Inorganic chemistry is a branch of chemistry in which we deal with transition metal compounds.…
Q: Which of the following statements is not correct? O The effusion of H₂ is faster than the effusion…
A: Effusion depend on pressure and temperature and the root mean square speed. At constant temperature…
Q: Why should a TLC plate be removed from the solvent before the solvent front reaches the top of the…
A: TLC plate: For both quantitative and qualitative analysis, thin layer chromatography (TLC) would be…
Q: How many balloons of 4.00-L capacity can be filled with hydrogen at a pressure of 1.00 atm and 27°C…
A: The number of balloons can be calculated the number of moles of gas filled in one balloon and the…
Q: (5) A new element, element Jn, is discovered on a separate planet. The energy requires to ionize one…
A:
Q: Write the Proton Balance Equation for the following solutions: 1) H2SO3
A: Proton balance equation also known as proton condition, which is a mathematical equation to describe…
Q: Chemistry 5. Ted figured out that the hydrogenation reaction C2H2(g) + H2(g) → C₂H4(g) has a…
A:
Q: By closest packing 1.00 mole of ping-pong balls in a circle for one layer, calculate the diameter of…
A: The problem consists of 2 individual parts . The first part need the concept of Quantum chemistry.…
Q: 3. In solution, sodium methoxide is a weaker base than sodium tert-butoxide because..... A.…
A: Answer: These are the questions based on chemical properties of organic compounds.
Q: Complex or Compound Name Metal Metal d Charge electrons Molecular Geometry Obeys 18- electron rule?
A: Coordination chemistry is branch of chemistry in which we deal with coordination compound.…
Q: o 50₂ CO₂
A: Lewis dot structures (or electron dot formulas) are the diagrams in which the valence electrons of…
Q: At 1 atm, how much energy is required to heat 87.0 g H₂O(s) at -18.0 °C to H₂O(g) at 135.0 °C?
A: We are asked to calculate energy to convert 87.0g H2O(s) at -18.0°C to H2O(g) at 135.0°C
Q: What would the name of this molecule be
A:
Q: Give the proper formulas for the following
A: To solve this problem we have to write the formula of the given compound . The formula of the…
Q: Final Burette Reading Initial Burette Reading Mass of flask + water (after adding aliquot) Mass of…
A: Density of a substance can be defined as the mass per unit volume. It is a property of substance…
Q: CHALLENGE PROBLEMS 1. You are approaching a traffic light. If the frequency what is the wavelength…
A: The relation between wavelength and frequency of light is : V= C/λ Here C is the speed of…
Q: Spell out the full name of the compound.
A:
Q: Cr(H2O)5Br]2+
A: Coordination chemistry is branch of chemistry in which we deal with coordination compound.…
Q: Draw all the resonance forms of the sigma complex formed by nitration of p-xylene. Draw the…
A: Sigma complex is formed by para xylene when it attacks on the electrophile, NO2+ ion. It is a…
Q: 19. If 500 mL of mineral oil are used to prepare a liter of mineral oil emulsion, how many grams of…
A: 19. 500 mL of mineral oil having a specific gravity of 0.87 are used to prepare a liter of mineral…
Q: 6. What is the frequency of a light source that has a wavelength of 620 nm? What is the energy this…
A: First we would convert wavelength from nm to m . Then using relation between frequency and…
Q: (a) Relative Abundance 100 -08 10 (b) 20 30 교 40 예 50 60 (d) 70 80
A: We are given the mass spectrum of an unknown molecule and we have to identify the unknown molecule…
Q: student forgets to sand the magnesium metal before the experiment . would his percent yield be…
A: The atomic number of magnesium metal is 12. It belongs to 's' block element. It is denoted by the…
Q: Balance the equation by inserting coefficients as needed. equation: Li,N+ H₂O- -> LiOH + NH3
A: Here we are required to balance the given reaction
Q: 3. Names of molecular compounds Formula Name CIFS CS₂ PCI Formula Name SF6 N₂0₁ SeF
A: Naming of the compound: • First you have to decide whether the compound is ionic or covalent. •…
Q: 73- 70- 65- 60- 55 50- 45- 40- 35- 30- 25- 20 15- 10- 5- 14 4000 2958.19cm-1 2931.95cm-1 3500 3000…
A: IR spectra provides the information of possible functional groups in the unknown compound. IR works…
Q: Calculate the overall net charge of the amino acid Lys at pH 10.4. (use the pK₂ values given earlier…
A: Overall charge on Lysine amino acid can be calculated by drawing its structure at given pH.
Q: You need to make an aqueous solution of 0.204 M magnesium acetate for an experiment in lab, using a…
A: Given, volume = 250 mL molarity of magnesium acetate = 0.204 M we have to calculate the mass of…
Q: How many milliliters of 6.0 M H₂SO4 (aq) would it take to dissolve 1.530 g of CuO precipitate? Show…
A: Molarity = moles of solute÷Volume of solution Moles of solute = mass of solute÷molar mass of solute…
Q: Consider the reaction: 2Na(s) + Cl₂(g) → 2NaCl(s) A student is making sodium chloride (NaCl(s)) by…
A: we have to calculate the percentage yield
Q: Calculate the ionic strength of a solution made by mixing 100.0 mL of 0.0200 M strontium nitrate…
A:
Q: Choose any EAS reaction. For your choice show an arrow-pushing mechanism using the compound below…
A: Electrophilic aromatic substitution(EAS): The aromatic compounds do not undergo addition reactions…
Q: Iron (II) can be precipitated from neutral solutions by oxidizing it with MnO4": MnO4 (aq) + 3Fe(OH)…
A:
Q: Chemistry 2. Prove that (-º), = for an ideal gas. = (F),
A: Here we are required to prove the above relation .
Q: D D Question 3 Phosgene decomposes by the reaction: COC₂ (g) CO (g) + Cl₂ (g) Kc- 8.3 x 10-4 (at…
A: Equilibrium concentration of COCl2 =? Equilibrium concentration of H2S =?
Q: Examining ammonia (NH3) in a laboratory you find that compressing 90.4 g of that substance to a…
A:
Step by step
Solved in 2 steps with 2 images

- In the following three compounds(1,2,3) arrange their relative reactivity towards the reagent CH3Cl / AlCl3. and justify it.Provide a detailed , stepwise mechanism for the following transformation . Use the curved arrow formalism to show the flow of electrons . Show all lone pairs , formal charges , and pertinent resonance structures .Answer the following Compounds CORRECTLY by giving the IUPAC NAME OF IT. •Label with proper notations (like hyphen, commas, periods, and grouping symbols) thank u❤️
- Provide an IUPAC name for the structures below. Pay attention to absolute stereochemistry.How many hydrogens are implied at the indicated carbons?Draw a diastereomer for each of the following compounds: Draw the molecule on the canvas by choosing buttons from the Tools (for bonds), Atoms, and Advanced Template toolbars. The single bond is active by default. Show the appropriate stereochemistry by choosing the dashed or wedged buttons and then clicking a bond on the canvas.
- Below are two potential methods for preparing the same ether (Option A and B), but only one of them is actually successful. Identify the successful approach (A or B)Provide the IUPAC name of the compound shown below please don’t forget to include the stereocenters.Consider the E2 elimination of 3‑bromopentane with hydroxide. The starting material consists of a chiral carbon with an in plane bond to bromine pointing to the upper left, an in plane bond to ethyl pointing to the right, a wedged bond to ethyl pointing to the lower left and a dashed bond to hydrogen pointing to the lower left. This reacts with hydroxide to form the product, water and bromide ion. Complete the curved arrow electron-pushing. Use three curved arrows to show the conversion to the product. Draw the organic product.
- Below is a schematic representation of possible reactions that Compound X can undergo. Use the scheme to answer the following questions. What is the IUPAC name for Compound X? What type of reaction (s) is/are represented by (i) and (ii)? Compound X undergo transitions through either [A] or [B] to produce compounds [1], [2], [3] and [4]. Draw the structures of [A] and [B].Determine whether the following molecules are aromatic (A), non-aromatic (NA), or anti-aromatic (AA), and show the use of a Frost circle in determing aromaticity. You may assume planarity.1.State the effects of substituents on a benzene derivative towards further aromatic substitution.

