1.Calcluate the theoretical yield of the coumerin prodcut from the reaction below. 2. Given an actual yield of 4.1 grams of coumarin. Calculate the percent yield of coumerin.
1.Calcluate the theoretical yield of the coumerin prodcut from the reaction below. 2. Given an actual yield of 4.1 grams of coumarin. Calculate the percent yield of coumerin.
Chapter2: Crystallization
Section: Chapter Questions
Problem 3Q
Related questions
Question
100%
1.Calcluate the theoretical yield of the coumerin prodcut from the reaction below.
2. Given an actual yield of 4.1 grams of coumarin. Calculate the percent yield of coumerin.
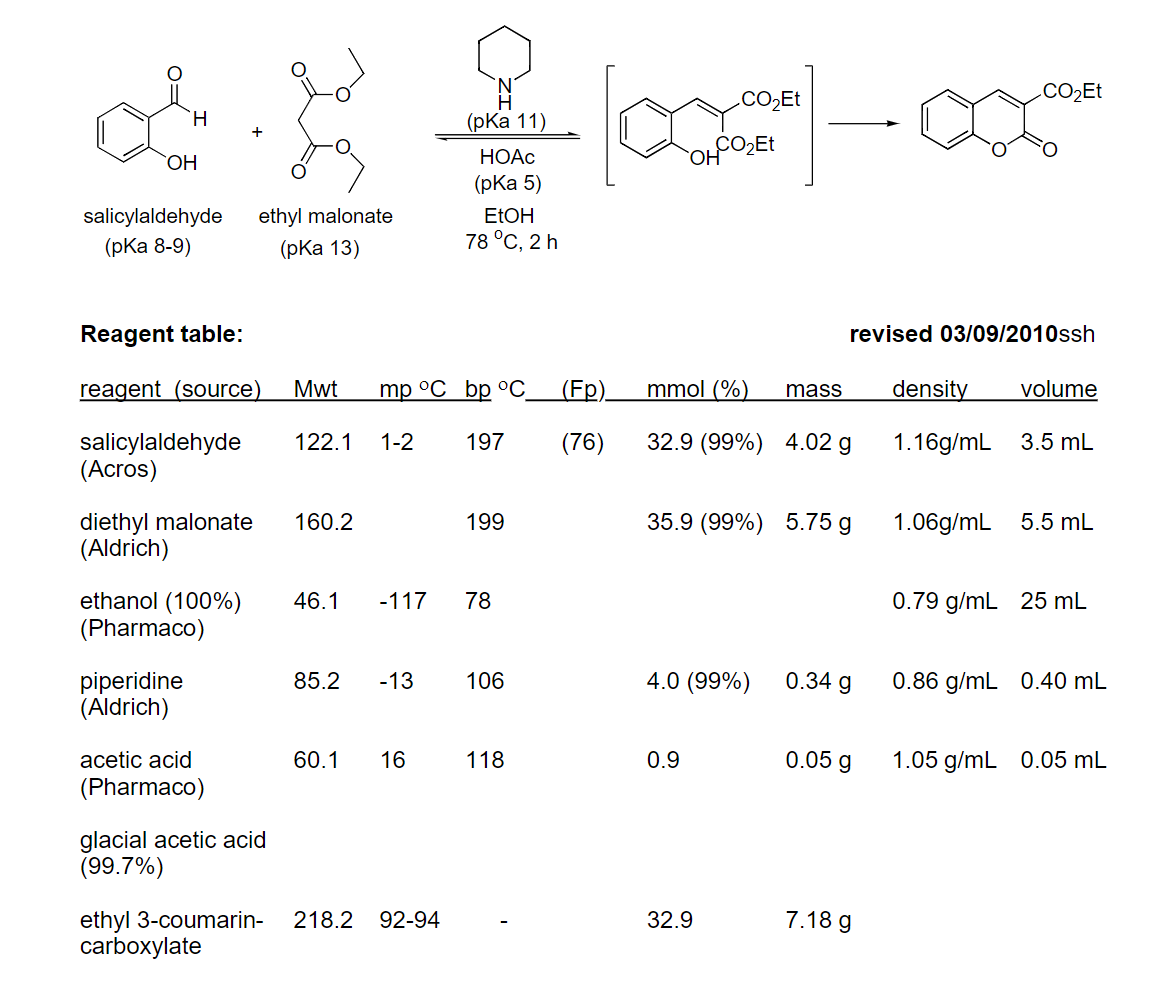
Transcribed Image Text:.CO2Et
.CO2E
(pКa 11)
ОН
НОАС
(pКa 5)
salicylaldehyde
ethyl malonate
EEOH
(pКa 8-9)
(РКa 13)
78°С, 2 h
Reagent table:
revised 03/09/2010ssh
reagent (source)
Mwt
mp °C bp OС
(Fp)
mmol (%)
mass
density
volume
salicylaldehyde
(Acros)
122.1
1-2
197
(76)
32.9 (99%) 4.02 g
1.16g/mL
3.5 mL
diethyl malonate
(Aldrich)
160.2
199
35.9 (99%) 5.75 g
1.06g/mL
5.5 mL
ethanol (100%)
(Pharmaco)
46.1
-117
78
0.79 g/mL 25 mL
piperidine
(Aldrich)
4.0 (99%)
0.34 g
85.2
-13
106
0.86 g/mL 0.40 mL
acetic acid
60.1
16
118
0.9
0.05 g
1.05 g/mL 0.05 mL
(Pharmaco)
glacial acetic acid
(99.7%)
7.18 g
ethyl 3-coumarin-
carboxylate
218.2
92-94
32.9
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

Recommended textbooks for you

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT


Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT


Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning