Q: What is the expression for the change in concentration of A if the initial concentrations of A and B…
A:
Q: ulate the pH of a solution prepared by dissolving 1.90 g of sodium acetate, CH,COONA, in 85.0 ml of…
A: Given, Mass of Sodium Acetate = 1.90 g Moles of Sodium Acetate = 1.90/82 = 0.0232 mol Moles…
Q: t is desired to prepare cupric sulfate solution by dissolving 250 grams of cupric sulfate in enough…
A: Since you have posted a question with multiple sub-parts, we will solve the first three subparts for…
Q: What is the difference between the enthalpies of the products and the reactants? AH O AG O Am O AS…
A: Which one of the following is correct
Q: CH3CCH (propyne or methylacetylene)
A:
Q: Find the theoretical density of aluminum given the table below and atomic mass of aluminum of 26.98…
A: Answer: Aluminum has FCC crystal packing in which atoms are present at the vertices and center of…
Q: One gram of sodium hydroxide pellets is dissolved in two hundred-fifty milliliters water. What is…
A:
Q: A chemist fills a reaction vessel with 5.97 atm chlorine (Cl,) gas, 6.65 atm phosphorus (P.) gas,…
A: ∆G for reaction can be calculated using formula ∆G = ∆G° + RTlnQ So, first we need to calculate ∆G°…
Q: 1- Calculate the rate law equation. Report the rate constant with its unit. What is the overall…
A:
Q: ion 3 or the molecule below, the s bond between the carbons marked I and II is formed by…
A:
Q: The following skeletal oxidation-reduction reaction occurs under basic conditions. Write the…
A: Consider the given redox reaction, answer the following questions---
Q: Calculate the pH of a 0.350 M solution of benzoic acid. Benzoic acid is a weak monoprotic acid with…
A:
Q: 2On heating N,O, the following data obtained Pressure(mm) 360290 278 139 114 52 t, (Sec) 212255 300…
A: 2) To calculate order of the reaction and half life period for heating of N2O5. Please post other…
Q: Give a specific application of Lechatelier's principle in our daily lives with its concept…
A: As per the Lechatelier's principle, any system in equilibrium when subjected to any external change…
Q: An aqueous solution of ferric hydroxide has a specific gravity of 1.25 and a weight fraction of…
A: Given , Specific gravity or density = 1.25 Weight fraction of solute = 0.15g To calculate…
Q: A supply of fluorodeoxyglucose (FDG) arrives at a positron emission tomography clinic at 8 am. At 2…
A:
Q: What is the solubility of argon (in units of grams per liter) in water at 25 °C, when the Ar gas…
A:
Q: According to the collision theory of kinetics, All of the options describe the collision theory of…
A: Rate of reaction :- The decrease in concentration of reactants with time or.increase in…
Q: • When 235U is bombarded with one neutron, fission occurs and the products are three neutrons, 94K…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: 80 CO AS = ΔΗ ||
A: Entropy is degree of randomness or disorderness in system. Order of Entropy of different phases…
Q: A bomb calorimeter, or constant volume calorimeter, is a device often used to determine the heat of…
A: Heat capacity of calorimeter is calculated by using formula : Heat capacity = Heat absorbed by…
Q: 1- Calculate the rate law equation. Report the rate constant with its unit. What is the overall…
A: Given reaction: NH4+(aq) + NO2-(aq) → N2(g) + 2H2O(l) We have to find the rate law equation. Also,…
Q: R-CR R H 1 The following compound can be dassified as an acid NO to. Strong and a low pka value b.…
A:
Q: Chemistry Describe the complex fused ring system of cocaine, ester functionality, benzene ring, and…
A: Cocaine have molecular formula of C17H21NO4 . The systematic name(IUPAC) of Cocaine is…
Q: A weak acid is dissolved in water to generate a solution that is 0.100 M. The pH of this solution is…
A:
Q: If a rock sample was found to contain 1.27 x 10-7 mol of argon-40, how much potassium-40 would also…
A:
Q: 3. Assuming the presence of acids HA and bases (A) as needed, give a plausīble mechanism (think…
A:
Q: What is the molarity of a solution of calcium chloride which contains 33500 mg of solute in a total…
A: Molarity is calculated using formula : Molarity = Moles of soluteVolume of solution (L) Note : As…
Q: Which of the following substances has the highest boiling point? CH3CN CH3Cl HCOOH CH2O…
A: The boiling point compound is directly proportional to the strength of intermolecular forces of…
Q: Aluminum metal reacts with iron (II) sulfate to produce aluminum sulfate and iron metal. How many…
A:
Q: Given that the energy ordering of molecular orbitals was shown by calculation to be 1σ, 2σ, 1π, 3σ,…
A: Solutions Given that Given that the energy ordering of molecular orbitals…
Q: 9. Which of the following has the greatest number of particles? (A) 9.012 g of Be (B) 1 mole of Na…
A: The no of moles is the ratio of given mass to that of molar mass of that substance. For more than…
Q: When a 19.34 gram piece of lithium is placed in a sample of water, how many grams of hydrogen gas…
A:
Q: 8. The relative mass of carbon is 3/8 that of an oxygen molecule. How many grams of carbon are…
A: 1 mole of any substance contains 6.022 × 1023 particles. This means same no of moles of any…
Q: Consider the reaction data. A → products T (K) k (s-1) 225 0.372 625 0.611 What two points should be…
A: Here we are required to find the activation energy of the reaction
Q: Given the following formation reactions of carbon dioxide, water, and ethanol, compute for the total…
A: Given-> C2H5OH(l) + 3O2(g) ---> 2CO2(g) + 3H2O(g)
Q: The reaction of 14.4 g of CIO2 with excess H,O yields 14.0 g of HCIO3. What is the percent yield? 6…
A: Which one of the following is correct answer
Q: a. How many moles of acetic acid and sodium acetate are present in 50.00 mL of the buffer? 2. A…
A:
Q: The reaction of 2-methylpropene with HBr in ether is described below. Answer the following questions…
A: 1.) In this reaction , HBr adds on double bond to give a single bond . In this reaction , first H+…
Q: 28.2 mL sample of 0.103 M formic acid (HCH02) is titrated with 0 103 V/ NaOH. Calculate the pH after…
A:
Q: What is the IUPAC name of the following compound: ICH3 (R)-2-hexen-5-ol (S)-2-hexen-5-ol…
A:
Q: What. is the addit.on the pH of 25.0mLof resulting from e solution with pH 3,550 I1.25e? (10.9) of…
A:
Q: 1. O3 14. 2. (work-up) OH C. A. B. 15. A. В. C. O, NaHCO, NaHco,
A: -> ozonolysis of alkene produce carbonyl compounds. ->In presence of peroxy acid epoxidation…
Q: A solution of Nickel sulfate (II) (MW: 154.75 g/mol) has a concentration of 500mM. Give the…
A: Answer: In this question we have to convert molar concentration of the NiSO4 solution into (w/v)% of…
Q: 2Al(s)+3CuCl2(aq)→3Cu(s)+2AlCl3(aq) A piece of aluminum metal is placed in an aqueous solution of…
A:
Q: A saturated solution of Pb(OH)2 was found to contain 4.1x10-8 mol OH in 100.0 mL. Calculate the Kep…
A: 1.) To calculate Ksp, we would write dissociation equation of Pb(OH)2 and write it's ksp expression.…
Q: Which of the following amino acids is expected to be the LEAST soluble in water and why? CH3 H3N CO2…
A: First question : LEAST soluble in water : All three are amino acids having different side chain…
Q: What is the irreducible representation for the a-bonding situation of an homolepetic octahedral…
A:
Q: What is the major organic product obtained from the following synthesis of a halohydrin: CH3 Br CH3…
A: In this question we have to tell the major organic product of the reaction.
Q: Provide the correct IUPAC name for the compound shown here. H3C H H CH-CH3 CH3
A:
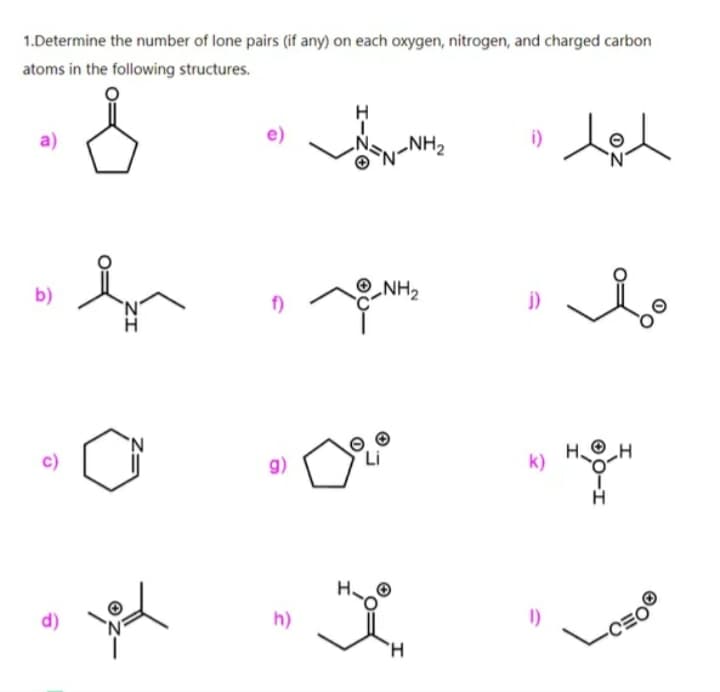
Step by step
Solved in 2 steps with 2 images

- Select all of the following that have permanent dipole moments (i.e., they are polar). -BCl3 -PCl3 -CH3OCH3 (an ether) -C3H8pls help ASAP! “which term best describes the relationship between the following two molecules?”Make a continuous model for C4H10 by using 4 black 4 hole carbon atoms, 10 white one hole hydrogen atoms, and 13 pink bonds. Then write in wedge dash notation C4H10 and add in missing hydrogen atoms.
- Can someone please help me label the localized and delocalized lone pairs and pi bonds in this structure? Thank you so much!1. Convert the following condensed structures into Bond-line structures: a) CH3CONHCH2OCH3 b) O(CH2)3CHCH3 c) CH3CHCHN(CH3)CH2CH3 d) (CH3)3CCH2CH2COOCH2CH3 e) CH3CH2CCCH2CO(CH2)2N(CH3)2 f) CH3CH2C(CH3)CHCH(CH2)4 g) (CH3)2CCHCH2OCH(CH3)CH2CN h) CH3CH2CHClCHBrCH2CONHCH(CH3)2 i) (CH3)2CHCCCH2OCH(CH2Br)CH2CH3 j) CH3NHCHCCH3CHClCON(C2H5)2Consider the compound in Figure 3. Which types of orbitals overlap to make the bond indicated in red? * A- sp³ – sp³ B- sp² – sp² C- sp³ – sp² D- sp² – sp³ E- None of these options is correct
- Organic chemistry is a bit like cooking. Later in this course we will study “recipes” for preparingnew molecules. A common instruction in such recipes is “add a strong acid” or “add a strongbase” (rarely are they added at the same time since a violent reaction ensues). You will see theseterms frequently from now on, and are expected to be able to recognize them or draw theirstructures from memory. Memorize them before next class.A)mark any formal charges on the structures below, and provide the hybridization for any atom with an arrow pointed to it. B) below draw the various Newman projections listed if looking down the C2--> C3 bondthe context is "Cosmone is a molecule used by fragrance manufacturers to provide a rich and elegant musky essence to many perfumes. Cosmone has the molecular formula C15H26O." I need help on part (e) and (f).
- Polarity (a 3-d representation with bond and molecular dipoles annotated) of ClO4- , PF3, H2O, PF5 , SF4What are all of the types of orbital overlaps that occur in the above structure. p-p overlap sp²-sp overlap s-sp² overlap sp²-sp² overlap s-sp overlap sp-sp overlap s-s overlap ---------- In cumulene, what are the C=C=C and H−C−H ideal bond angles, respectively?Enter the C=C=C bond angle followed by the H−C−H bond angle separated by a comma (no spaces, no º symbol required).Possible Newman projection for following?

