Q: Write the products of the reaction below and in
A: SN1 reaction- AcOH is a polar protic solvent always favours SN1 reaction. step.1 Loss of leaving…
Q: 5.) For the following molecules V-Z, please circle which organic molecules would function well as…
A: Dienophiles- The alkene which has an electron-withdrawing group is known as Dienophiles. The…
Q: Rank the following dienophiles (from least reactive to most reactive) in terms of reactivity in a…
A:
Q: Explain how and why rearrangements occur during Friedel-Crafts alkylation reactions forming more…
A: Concept - In Friedel-Crafts alkylation reaction is an electrophilic aromatic substitution reaction…
Q: Rank the indicated hydrogen atoms in order of increasing ease of abstraction in a radical…
A: The ease of abstraction of hydrogen in a radical halogenation reaction depends on the stability of…
Q: A student was about to turn in the products he had obtained from the reaction of HI with…
A: Markovnikov’s rule: unsymmetrical alkene reacts with hydrogen halide, halide ions goes to the more…
Q: provide the proper diene and dienophile that will result in generating the products below. Thank…
A: All given products can be form through diels - alder reaction. It is the reaction between a…
Q: How does doubling [B:] affect the rate of an E1 reaction?
A: E1 reactions are the unimolecular elimination reaction. This is a two step elimination reaction, in…
Q: How does changing the solvent from CH3OH to DMSO affect the rate of an E2 reaction?
A: The reaction in which one molecule is eliminated from reactant compound, such reaction is called…
Q: What is the major product when the methoxy substituent in the preceding reaction is bonded to C-2 of…
A: The major product of the given reaction depends on the charge distribution in the diene and…
Q: What is the major product of the reaction shown below?
A: Answer: (D) Nucleophilic addition takes place at aldehyde carbonyl carbon. Nucleophile is alcohol…
Q: Explain why methyl vinyl ether (CH2 = CHOCH3) is not a reactive dienophile in the Diels–Alder…
A: Diels-alder reaction is a cycloaddition reaction in which two molecules combine to form a new ring.…
Q: What can cause an exception to Zaitsev's rule during E2 dehydrohalogenations? OA steric hindrance O…
A: To identify: What can cause an exception to Zaitsev's rule during E2 dehydrohalogenations.
Q: Rank the primary, secondary, tertiary, and aryl halides in terms of reactivity with the nucleophile.…
A: Nucleophilic substitution biomolecular reaction (SN2): The bimolecular nucleophilic substitution…
Q: 5.) For the following molecules V-Z, please circle which organic molecules would function well as…
A: Diels Alder reaction is between diene or conjugated diene and alkene.
Q: 9. How do Lewis acids play a role in generation of the electrophile? A mechanism will be needed to…
A: We are authorized to answer one question at a time. Since you have not mentioned which question you…
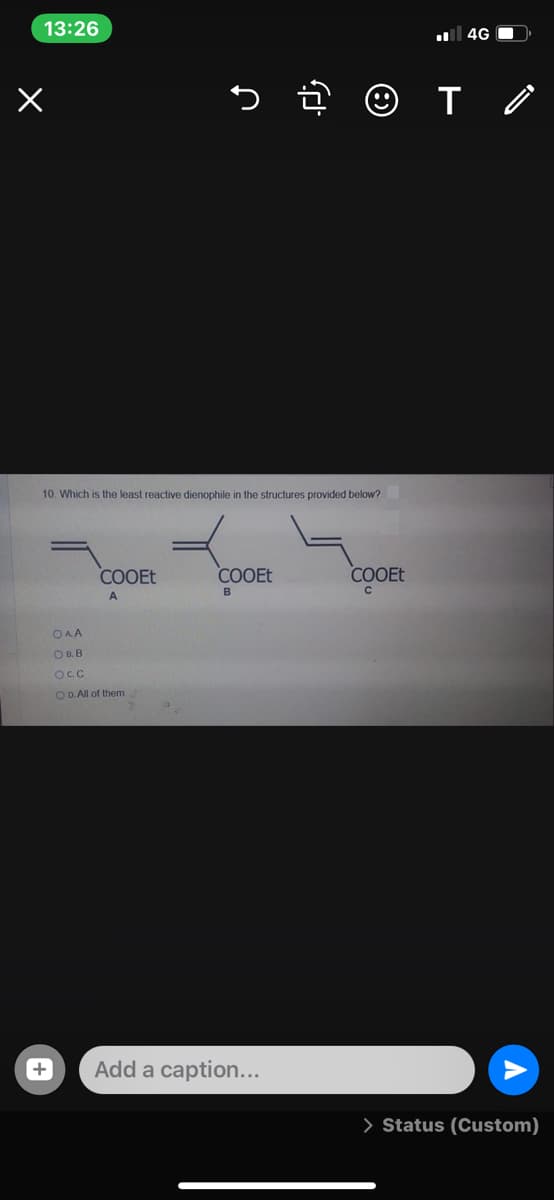
Q: Which is the least reactive dienophile in the structures provided below? COOET COOEt COOET OAA OBB…
A: Electron withdrawing group attached to the dienophile increases its reactivity. Electron releasing…
Q: Why does the incoming nitro group go para and not ortho?
A:
Q: 5.) For the following molecules V-Z, please circle which organic molecules would function well as…
A: Dienophiles- The alkene which has an electron-withdrawing group is known as Dienophiles. The…
Q: the dehydrohalogenation (E2) reaction, d
A:
Q: CH3 NC. NC A C B
A: In the Diels-alder reaction, the diene and dienophile react with each other to form the new product.…
Q: 6.) For the following reactions, please draw the structures of both the diene and dienophile that…
A:
Q: 3. Please rank the following dienes in their rate of reaction with the same dienophile in a…
A:
Q: see the attached question and out of these 3 comounds, which one is the best dienophile ?
A:
Q: 12) Show the formation of benzylic radical from toluene, NBS and light.
A: N-Bromosuccinimide (NBS) is used as oxidizing and brominating agent, which used as source for…
Q: Which of the following two compounds do you expect to be a better electrophile, and why?
A: The electrophiles are species, which are electron deficient and it can readily accept electrons. The…
Q: Rank the following dienophiles in order of increasing reactivity.
A:
Q: Rank the following dienophiles in order of increasing reactivity.
A:
Q: How does doubling [RX] affect the rate of an E1 reaction?
A: The removal of hydrogen halides in a two-step mechanism where the rate of the reaction depends on…
Q: Which of these would be the best electrophile for an Sy1 reaction? A) CH;Br B) (CH3);CBr C)…
A:
Q: Which is the major product of the reaction below?
A:
Q: Epoxide formation Radical bromination Br H.
A:
Q: What diene and dienophiles are used to make the following compounds? b. :a.
A: Answer:- This question is answered by using the simple concept of Diels -Alder reaction in which…
Q: Rank the following dienophiles in order of increasing reactivity. Be sure to answer all parts. A) OH…
A: Given Dienophiles
Q: COOH COOH
A:
Q: 5. Provide the missing reagents and product in the boxes below. Br2 HO
A: The solution is given below
Q: Which of the following statements is correct regarding SN2 reactions? SN2 reactions require a strong…
A: Given statements, 1. SN2 reactions require a strong nucleophile 2. SN2 reactions require a primary…
Q: What is the enol tautomer of the molecule below?
A:
Q: What two sets of a conjugated diene and a dienophile could be used to prepare the following…
A: Diels-Alder reaction: A conjugated diene reacts with a compound containing a carbon-carbon double…
Q: Acetoor
A: Nucleophilic Substitution Reaction: In the nucleophilic substitution reaction, one nucleophile…
Q: What is the major product for the reaction above?
A: Hi, as the complete question is 15 and question (16) is incomplete because options are not visible.…
Q: Step 3: Now let's focus on the dienophile. The dienophile is the electrophile in the Diels-Alder…
A: Diels Elder reaction is proceed by the reaction between diene (compound with double bond) and…
Q: ozonolysis
A: Ozonolysis is defined as a method of oxidatively cleaving of alkenes or alkynes using O3(ozone).
Q: 200 +
A:
Q: Which diene from the list below would be expected to react most rapidly with the dienophile?
A: Diene of Diels-Alder reaction are electron rich and dienophile are electron deficient.
Q: what reason is given on why the dienophile likes the diene?
A: The word dienophile means compound which shows attraction with diene . Phile means love and diene is…
Step by step
Solved in 3 steps with 3 images

- Predict the majpr product(s) of this radical halogenation reaction. I, II, III, IV.cmplete the reacting by adding necessrt reagents. write in thapce provided. make sure to write regents in chemical formula . n separate regents with comma & space (ex. h2so6, h202)I’m currently trying to write a lab report for the synthesis of dimolybdenum tetraacetate [Mo2(O2CCH3)4] from the reaction of molybdenum hexacarbonyl, Mo(CO)6 in glacial acetic acid and acetic anhydride under a nitrogen atmosphere, involving the difficult formation of a quadruple bond and requires high heat and long reaction time (approximately 20 hours). But these are the question I’m stumped on: 1. Why need the reaction be done under nitrogen? We also added dichlorobenzene and hexanes during the reaction. 2. Explain the purpose of dichlorobenzene and hexanes. 3. Why does the reaction take 20 hrs?
- Please explian the reasoning and priorities when ranking dienes and dienophiles, thank youChemistry Give the products of the reaction of 1 mole of 2-methy1-1,3-pentadiene with 1 mole of HBr. Whichproduct(s) will predominate if the reaction is under kinetic control? Which products) will predominateif the reaction is under thermodynamic control?In light of your answer to Problem 30-40, explain why a mixture of products occurs in the following reaction:
- please provide the machanisms of 1a, 1e, 1f! (plz give proper explanation)Weight of crude/pure product: 0.819/0.047g Percent yield: Theoretical Yield: Melting Point crude/pure:195/190 Q: What would the theoretical yield froms the Diels-Alder Adduct with your product. Also, what is the actual yield? How do i calculate those? i used anthracene 200mg, maleic anhydride 110 mg and 6ml of xylene



