
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 30.SE, Problem 41AP
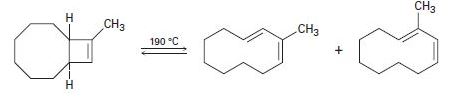
In light of your answer to Problem 30-40, explain why a mixture of products occurs in the following reaction:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
The following reaction has a ΔSsystem < 0
O2(g) → 2O(g)
T or F
can you explain why this is false?
Answer Nos. 7-12 Only.
Write only the product and the type of reaction it undergone.
Thank you!
I need help with this problem.
how how the forward and reverse reaction rates respond to having some HOCl suddenly removed from the following system at t1.
H2O(g) + Cl2O(g) ⇌ 2 HOCl(g)
Chapter 30 Solutions
Organic Chemistry
Ch. 30.1 - Prob. 1PCh. 30.3 - Prob. 2PCh. 30.3 - Prob. 3PCh. 30.4 - Prob. 4PCh. 30.6 - What stereochemistry would you expect for the...Ch. 30.6 - Prob. 6PCh. 30.7 - Prob. 7PCh. 30.8 - Propose a mechanism to account for the fact that...Ch. 30.8 - When a 2, 6-disubstituted allyl phenyl ether is...Ch. 30.9 - Prob. 10P
Ch. 30.SE - Predict the product obtained when the following...Ch. 30.SE - Prob. 12VCCh. 30.SE - The following rearrangement of N-allyl-N,...Ch. 30.SE - Plastic photochromic sunglasses are based on the...Ch. 30.SE - Prob. 15MPCh. 30.SE - Prob. 16MPCh. 30.SE - Prob. 17MPCh. 30.SE - Prob. 18APCh. 30.SE - Prob. 19APCh. 30.SE - Prob. 20APCh. 30.SE - Prob. 21APCh. 30.SE - Prob. 22APCh. 30.SE - Prob. 23APCh. 30.SE - Prob. 24APCh. 30.SE - Prob. 25APCh. 30.SE - Prob. 26APCh. 30.SE - Prob. 27APCh. 30.SE - Prob. 28APCh. 30.SE - Propose a pericyclic mechanism to account for the...Ch. 30.SE - Prob. 30APCh. 30.SE - Prob. 31APCh. 30.SE - Prob. 32APCh. 30.SE - Prob. 33APCh. 30.SE - Bicyclohexadiene, also known as Dewar benzene, is...Ch. 30.SE - Prob. 35APCh. 30.SE - Prob. 36APCh. 30.SE - The 1H NMR spectrum of bullvalene at 100 C...Ch. 30.SE - Prob. 38APCh. 30.SE - Prob. 39APCh. 30.SE - Prob. 40APCh. 30.SE - In light of your answer to Problem 30-40, explain...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- On which do you expect to have a more intense and concentrated “hotspot” of negative charge: methoxideion or bicarbonate ion?arrow_forwardWhich of the following reactions result in a positive ∆ Ssys? A. Pb(NO3)2 (aq) +KCl (aq) ---> PbCl2 (s)+ KNO3 (aq) B. HCl (g) + H2O (l) ---> HCl (aq) C. H2 (g) + I2 (g) ---> 2HI (g) D. 2H2O (g) ---> 2H2 (g) + O2 (g) E. H2O (g) ---> H2O (l)arrow_forwardThe following reaction scheme contains one or more flaws. Explain what is wrong in each of the following reaction schemes and suggest how you would correct the scheme.arrow_forward
- Through what mechanism does this reaction primarily proceed? a. SN2 b. E1 c. E2 d. SN1arrow_forwardWhich of the following reactions result in a positive ∆ Ssys? a. H2O(g) = H2O(l) b. H2(g) + I2(g) = 2 Hl(g) c. C2H2O2(g) = 2CO(g) + H2(g) d. H2O(g) + CO2 (g) = H2CO3(aq)arrow_forwardChoose the best reagents from the list provided below for carrying out the following conversion. Match the reagent with the step number. HCl (aq), Zn(Hg) KMnO4, H3O+ CH3Cl, AlCl3 HNO3, H2SO4 Cl2, FeCl3 fuming sulfuric acidarrow_forward
- Need your help for this: Show the reaction mechanism for the formation of a. RCONH2 from the reaction of RCOOR' with NH3 b. RCOOR" from the reaction of RCOOR' with R"OH in acidic mediumarrow_forwardThe reaction of one equivalent of hydrogen bromide with 1,3-butadiene gives product D and G via pathway 1 and 2 respectively. Which intermediate is more stable? (This diagram will be used for problem 27 - 32.)arrow_forwardIdentify which substitution mechanism best fits the following statement: The reaction proceeds through a concerted mechanism. A) SN1 B) SN2arrow_forward
- Solve the given problem 8 and explain the mechanismsarrow_forwardIn the reaction of NaBH4 with formaldehyde in methanol sovlent, the products are BH3, CH3OH and sodium methoxide. what is the transition state for the slow step of this reaction? please give structure.arrow_forward________ is reduced in the following reaction: Cr2O72- + 6S2O32- + 14H+ → 2Cr3+ + 3S4O62 + 7H2O O2- S4O62- Cr6+ H+ S2+arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you


 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning



Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
SAR of Anticancer(Antineoplastic) Drug/ Alkylating agents/ Nitrogen Mustard; Author: Pharmacy Lectures;https://www.youtube.com/watch?v=zrzyK3LhUXs;License: Standard YouTube License, CC-BY